Berries are red, berries are blue…I’ve got a berry surprise for you!

Berries are red, berries are blue…I’ve got a berry surprise for you!
DESCRIPTION: I observe a red to blue color change when I rinse my bowl after eating frozen blueberries. Sounds like an acid-base reaction, doesn’t it? Read on to learn about the blueberry surprise!
INTRODUCTION: I often eat frozen blueberries for breakfast. When I do so, I notice that the blueberries leave a red-purple residue in the bowl. This color of the frozen blueberry residue is due to the presence of anthocyanins and anthocyanidins (Figure 1)1-3, which impart red, violet, and blue colors to many parts of plants: stems, leaves, fruits, and flowers.1-7 (For the rest of this post, both anthocyanins and anthocyanidids will collectively be referred to as AC).
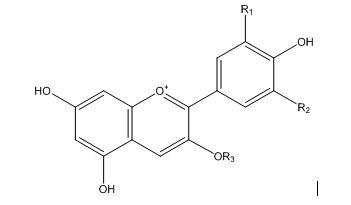
Figure 1: General structure of many anthocyanins (R3 = glucose) and anthocyanidins (R3 = OH). R1 and R2 = H, OH, or OCH3 for various compounds. For example, for the particular compound, delphindin (commonly found in blueberries),1-3 R1 = R2 = OH and R3 = glucose.
For quite some time now, I have noticed an interesting color change when I rinse my bowl with tap water after eating frozen blueberries. When doing so, the residue left from the frozen blueberries changes from red-violet to blue-violet (Figure 2).

Figure 2: Left - residue left in a bowl from frozen blueberries. Right - same bowl after rinsing with tap water.
When I first noticed this color change several years ago I reasoned that the blueberry residue changed color because of a change in pH upon addition of tap water. It is well known that AC molecules change color with pH4-7 so I simply assumed a pH change was occurring (Figure 3).
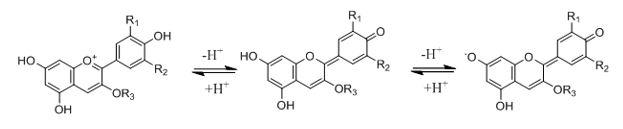
Figure 3: Simplified description of changes in structure in anthocyanins and anthocyanidids induced by increases in pH (indicated from left to right). These structures tend to impart (from left to right) red, violet, and blue colors. The full range of reactions involving pH-induced color changes is beyond the scope of this article.
Over the years I have become increasingly skeptical of the explanation that a shift in pH causes the color change I see upon rinsing my blueberry-stained bowl. That’s because blueberries tend to be acidic, so a mixture of blueberry residue and tap water is likely to be acidic. But AC molecules tend to be red or violet in solutions that are slightly acidic to acidic (Figure 2). How could blueberry residue turn blue in an acidic environment?
I decided to run several tests to try to figure out how the blueberry residue could be changing blue when rinsed with tap water. You can see the culmination of these investigations in the video below:
Video 1: Blueberries Are Red, Blueberries Are Blue..., Tommy Technetium YouTube Channel, 1/21/2020
PERFORMING THE EXPERIMENT WITH HOUSEHOLD ITEMS: You can perform the experiments seen in the video quite easily by using only items obtained around the home or at the grocery store. To do so, frozen blueberries are placed in a clear or white container. The container is shaken to deposit blueberry residue on it. After the blueberries are removed, a small amount of distilled water is added. Next, salts from various common sources (Table 1) are dissolved into the blueberry residue/water mixture. When doing these experiments I noticed that some of the metal ions from household sources induced color changes not observed in the experiments shown in the video.
Table 1: Descriptive results of experiments in which salts containing metal ions from various common sources were added to a mixture of frozen blueberry residue and distilled water
|
Metal ion |
Common source of metal ion |
Color observed |
|
None |
Distilled water |
Red-purple |
|
? |
Tap water |
Grey-blue |
|
Na+ |
Table salt |
Very similar to distilled water |
|
Mg2+ |
Epsom salts |
Blue-purple |
|
Al3+ |
Alum |
Deep violet |
|
K+ |
No Salt® Salt Substitute |
Very similar to distilled water |
|
Ca2+ |
Prestone® Driveway Heat® |
Blue-purple |
|
Cu2+ |
Powder from corrosion on old pennies |
Blue-purple |
DISCUSSION: In the video it is demonstrated that it is not only pH, but also metal ions that can cause AC molecules to change color. Thus, it could be that metal ions in my tap water bind with AC compounds to cause the blue color. I am not certain exactly which metal ion(s) and AC compounds interact to cause the blue color. Nevertheless, I did observe that copper ions induced a color change in blueberry residue strikingly similar to the color change caused by addition of tap water. I will note that some studies have indicated that when AC molecules bind to Cu2+, a shift to a bluer color appears.8-11 However, the blue shift seems to be more commonly observed upon interactions between AC molecules and Al3+, Mg2+, Ca2+, and/or Fe3+.4,5,12-14
The associations of metal ions with AC molecules often involve the formation of beautifully complex structures that involve as many as 12 individual AC molecules. In addition, molecules that are similar to AC, called flavones, are often involved in these intricate structures. The process of two or more AC molecules interacting is termed copigmentation, which is often accompanied by color change. Interactions leading to copigmentation can be mediated by metal ions and/or intermolecular forces between AC molecules (Figure 4).4-5,7,12-14

Figure 4: Highly simplified representations of some copigmentation structures involving AC molecules and flavones (adapted from references 4, 5, and 14). In the diagram, black bars represent AC molecules, red bars represent flavone molecules, and black spheres represent metal ions.
CONCLUSION: Who would have thought that the simple red to blue color change I observe when washing my dishes could involve such intricate chemistry? The beautiful nature of chemistry never ceases to amaze me. I hope you enjoyed learning about this “blueberry surprise” as much as I did. As always, feel free to leave a comment if you and your students try some of these experiments on your own – especially if you learn something new or observe something that makes you think my explanations might be wrong!
REFERENCES:
1. Soumalainen, H.; Keranen, A. J. A. The First Anthocyanins Appearing During the Ripening of Blueberries, Nature, 1961, 191, 498-499.
2. Francis, F. J.; Harborne, J. B.; Barker, W. G. Anthocyanins in the Lowbush Blueberry, Vaccinium angustifolium, Food Science, 1966, 31, 583-587.
3. Routray, W.; Orsat, V. Blueberries and Their Anthocyanins: Factors Affecting Biosynthesis and Properties, Comprehensive Reviews in Food and Science Safety, 2011, 10, 303-320.
4. Trouillas, P.; Sancho-García, J. C.; De Freitas, V.; Gierschner, J.; Otyepka, M.; Dangles, O. Stabilizing and Modulating Color by Copigmentation: Insights from Theory and Experiment, Chem. Rev. 2016, 116, 4937−4982
5. Kumi Yoshida,K.; Mori, M.; Kondo, T. Blue flower color development by anthocyanins: from chemical structure to cell physiology, Nat. Prod. Rep., 2009, 26, 884–915
6. Alkema, J.; Seager, S. L. The Chemical Pigments of Plants, J. Chem. Educ. 1982, 59, 183-186.
7. Khoo, H. E.; Azlan, A.; Tang, S. T.; Lim, S. M. Anthocyanidins and anthocyanins: colored pigments as food, pharmaceutical ingredients, and the potential health benefits Food & Nutrition Research, 2017, 61, 1-21.
8. Vestergaard, M.; Kerman, K.; Tamiya, E. An electrochemical approach for detecting copper-chelating properties of flavonoids using disposable pencil graphite electrodes: Possible implications in copper-mediated illnesses, Analytica Chimica Acta 2005, 538, 273-281.
9. Smyk, B.; Pliszka, B.; Drabent, R. Interaction between Cyanidin 3-glucoside and Cu(II) ions Food chemistry 2008, 107, 1616-1622.
10. Brown, J. E.; Khodr, H.; Hider, R. C.; Rice-Evans, C. A. Structural dependence of flavonoid interactions with Cu2+ ions: implications for their antioxidant properties, Biochem. J. 1998, 330, 1173–1178.
11. Mira, L.; Fernandez, M. T.; Santos, M.; Rocha, R.; Florêncio, M. H.; Jennings, K. R. Interactions of Flavonoids with Iron and Copper Ions: A Mechanism for their Antioxidant Activity, Free Radical Research 2002, 36, 1199–1208.
12. Kosaka Takeda, K. Blue metal complex pigments involved in blue flower color, Proc. Jpn. Acad. 2006, 82, 142-154.
13.Goto, T.; Tadao Kondo, T. Structure and Molecular Stacking of Anthocyanins – Flower Color Variation, Angew. C'hem. Int. Ed. Engl. 1991, 30, 17-33.
14. Shiono, M.; Matsugaki, N.; Takeda, K. Structure of the blue cornflower pigment, Nature 2005, 436, 791.
Comments
9Blueberries and Copper
Hi Tom,
I really enjoyed that discussion. I'm wondering where the copper ions are coming from? Are they leaching from the pipes, leaching from brass fittings? It would be interesting to check people's water from new houses where all of the plumbing is pex. (sp) Would that also have copper ions just because of the fixtures, or would the amount go down enough to have no effect on the blueberries?
In reply to Blueberries and Copper by Amiee Modic
I'm not 100% sure...
Hi Amiee:
Thank you for your comments and questions. I’ve not tested the water from my tap, but I will note that the water report for my township states that the water contains 100 ppb Cu2+. It is also the case that the water pipes in my home are made of copper metal.
Having said all this (and even though my experiments indicate Cu2+) I’m still not entirely sure the effect I’m observing is from Cu2+. First, I have a water softener which should remove a lot of the metal ions (including Cu2+) that could complex the anthocyanins. Also, several metal ions or mixtures of ions commonly found in tap water (Mg2+, Ca2+, Fe3+) can complex with anthocyanins and anthocyanidids. Thus, there are a variety of scenarios that could be causing what I’m seeing. You’ll note that in my experiments I only tested individual ions – but not mixtures of ions – with the blueberry mixture. You’ll also notice that I didn’t carefully control for added metal ion concentrations in my experiments.
So while it’s a fair explanation that Cu2+ is causing the effect, I’m not completely sold on the idea. Hopefully I’ll be able to carry out some more experiments to gain more insight into what is going on. Any suggestions you have for tests are welcomed!
Magnesium bisglycinate in blueberry smoothie
Hi Tom,
Nice article! While adding supplemental powders to blueberry smoothies, I was surprised to see a color change from the normal purple to a more turquoise blue. In changing the order in which I added them, I have narrowed down the culprit to the magnesium bisglycinate. It's a very fast change!
In reply to Magnesium bisglycinate in blueberry smoothie by Lynda Froese
Share video!!
Okay, you win the comments for this post. Can you share some video?
Blueberries and the like
Many of us in the UK occupied our spring 2020 lockdown doing these. See https://youtu.be/WX2dSdvVPe0 and later I did this https://youtu.be/tbR5dHcWWds . Almost any flowerhead, fruit skin and coloured leaf will yield something new. Of course I prefer to eat blueberries, so being a microchemist , I work with the skin peeled off by forceps from just two blueberries.
Thanks for the info from the States to use the Butterfly Pea plant. It might be worthwhile in lessons not to use its Latin name!!!! We can buy it online in the UK. Green Chemistry at its best, note https://www.beyondbenign.org/.
This is in a website you all may know; https://www.chemedx.org/article/aqueous-red-cabbage-extracts-more-just-…
In reply to Blueberries and the like by Bob Worley
Colorful
Beautiful work as usual, Bob. Thank you for sharing the videos and the link. I always enjoy hearing about what you are up to in the lab.
Share Your Thoughts