Aqueous red cabbage extracts: More than just a pH indicator

Co-Authored by Iain A. Smellie*, Iain L. J. Patterson*, Adrian Allan**, Bob Worley***
*University of St Andrews, School of Chemistry, North Haugh, St Andrews, United Kingdom, **Dornoch Academy, Evelix Road, Dornoch, Sutherland, United Kingdom, ***CLEAPSS, Brunel Science Park, Uxbridge, United Kingdom
The use of anthocyanins in red cabbage extracts (Brassica oleracea L. var. capitata L. f. rubra) as pH indicators has long been a popular classroom activity or outreach demonstration.1-4 The general structures of the red cabbage anthocyanins at different pHs are shown in scheme 1.5,6
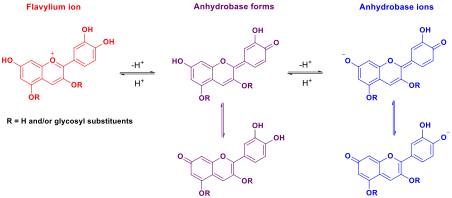
Scheme 1: Reaction scheme showing the structural changes in the anthocyanin chromophore (derived from red cabbage) in acid, neutral and weakly basic conditions.
Flowers, fruits and vegetables contain a diverse range of anthocyanins, commonly encountered examples are derived from the set of anthocyanidin aglycones shown in figure 1.5,6 Other aglycones are known, however the structural diversity observed in naturally occurring anthocyanins can also originate from O-glycosyl substituents. Glycosylation can occur at positions 3 and 5,6 although many familiar examples are 3-O-glucosides (some examples are provided in the supporting information document).
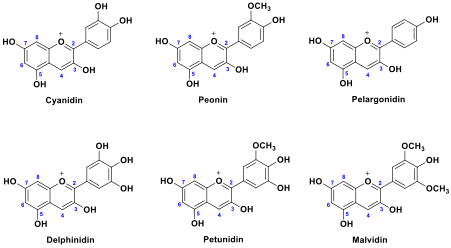
Figure 1: Key anthocyanidin aglycones (flavylium ion forms) observed in naturally occurring anthocyanins
Red cabbage is one of the most popular sources of anthocyanin indicators due to the ease of safe extraction of the dye (in this case derivatives of cyanin)7 and ready availability of cabbage from grocery stores. However, flower petals, such as those from roses8 and pansies,9 are also reported to be convenient anthocyanin sources too. More recently, the use of anthocyanin pH indicators has also proved to be a versatile activity to support home study tasks.9,10
In recent months, the response to the COVID-19 pandemic has led many educators being faced with restrictions on the delivery of laboratory teaching.11 As part of the teaching response, the use of plant-derived pH indicators has proven to be a versatile basis for practical tasks that can be completed at home. The vivid colours of anthocyanin pH indicators are engaging, and the variety of low hazard and inexpensive natural sources makes them easily accessible to educators in high schools and universities. These attributes led us to investigate further applications of plant-derived dyes in chemistry teaching and outreach activities. As part of our study, we sought to make use of materials that could be obtained from grocery stores, hardware stores or online retailers. It was reasoned that the use of commercially available materials would keep costs low and potentially allow the activities to be conducted as home study tasks if necessary. In this article we describe the use of plant derived-dyes in conjunction with household materials to illustrate colour changes that are not solely dependent on pH change. The first example is a reversible reaction based on oxidation/reduction chemistry. This process can be used to illustrate organic chemistry concepts (such as bisulfite addition, this is usually encountered in the context of addition to aldehydes12) and is relevant to important chemical principles applied in the production of fruit juices and wine. We also introduce very simple procedures that use plant dyes as a means to detect the presence of certain metal ions. These activities provide an opportunity to discuss the origin of the observed colours of flowers, fruits and vegetables.
Reversible decolorisation of red cabbage anthocyanin solution
Previous classroom activities have been used to show that anthocyanins extracted from grape juice13 and red cabbage14 are decolourised in the presence bisulfite ions at low pH. This effect is well known in the food industry, this is because the sulfites used to preserve certain fruit products and alcoholic drinks15 are known to react with anthocyanin chromophores16,17 (this process is illustrated in scheme 2). Under acidic conditions, flavylium ions (usually coloured red) derived from anthocyanins are susceptible to nucleophilic attack by bisulfite ions to afford the corresponding flavene-4-sulfonate (frequently referred to as a “bisulfite adduct”).17 the resulting sulfonates are colourless and explain the observed bleaching of wines, juices and other coloured plant-derived material on addition of sulfites or sulfur dioxide.16-18 This reaction is not desirable in red wines and coloured fruit juices, however, it can reduce undesirable “pinking” of white wines.18
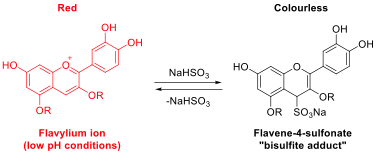
Scheme 2: Reaction scheme showing the reaction of cyanidin-derived anthocyanins with bisulfite ions.
Earlier reports have described classroom use of sodium bisulfite or sodium sulfite solutions for decolourisation of anthocyanin solutions. We have found that commercially available sodium or potassium metabisulfite tablets15 (“Campden tablets” 19) that are used in home brewing are also safe and convenient for effecting the same change. This observation is not surprising since metabisulfites dissolve in water to form bisulfite ions in solution. We found that addition of a drop of drain cleaner (that contains ca. 8% HCl) sourced from a grocery store was suitable for adjusting the aqueous solution of red cabbage extract to pH 1. Furthermore, we found that addition of dilute hydrogen peroxide (3%, available from online suppliers) was able to reverse the reaction with bisulfite. On addition of dilute peroxide, the solution returned to being red in colour, this observation is consistent with the flavylium species being reformed.
Example decolourisation procedure
In each trial, 5-10 drops of red cabbage extract was added to 25-30 mL of water. 1 or 2 drops of drain cleaner (containing 8% HCl) was then added to form a red solution. A commercially available “Campden” tablet was crushed with a spoon until a powder was obtained. Small portions of the crushed tablet were added to the solution with swirling and until the red coloured solution faded. To reverse the reaction, 3% hydrogen peroxide was added dropwise to the colourless solution with swirling. Peroxide addition and mixing were continued until the red colour of the flavylium ion was observed again (video 1). (A full procedure and additional pictures of this process are available in the supporting information document.)
Video 1: Bisulfite bleaching as recorded by the authors. Published by ChemEd X on Vimeo. (accessed 9/2/20)
Scheme 3 shows the reversible process with cyanidin-derived dyes extracted from red cabbage, however similar results were also obtained from anthocyanin extracts from rose petals, begonia petals, blue hydrangea sepals and aqueous black bean extract.

Scheme 3: Colour changes associated with the reversible reaction of anthocyanins with bisulfite ions in aqueous solution.
The observed colour changes can be explained by the fact that the formation of the colourless flavene bisulfite adduct is reversible,17,18 therefore addition of a suitable oxidant is able to reduce excess bisulfite in solution20 and shift the equilibrium in favour of the formation of the flavylium ion. This process is known to take place in white wines, in some cases, small quantities of anthocyanins in wine treated with sulfite can undergo oxidation by atmospheric oxygen.18 As a result, low concentrations of flavylium ions can then be formed and these introduce a pink hue to the wine. The same colour change has also been reported to occur on addition of dilute hydrogen peroxide to white wine at low pH.18
Colour changes from addition of metal ions
The variation of colour of anthocyanin solutions as a function of pH is very well understood,5,6 however this is not the sole environmental factor that determines the colour of plant materials that contain anthocyanins. The interaction between metal ions and anthocyanins is very important in the observed colours of flowers, one of the most studied examples of this is the origin of the colour of blue, violet and red hydrangeas (Hydrangea macrophylla).6 It has been known since the mid-20th century that control of soil pH determines the amount of Al3+ available to hydrangeas and that this balance has an effect on the observed colour of hydrangea sepals. This interaction is complicated, however, a significant finding is that in acidic soil (ca. pH 5) the hydrangea sepals have a tendency to be blue, this coloration being due to complexation of Al3+ to delphinidin 3-O-b-d-glucopyranoside (1). After much research, the structure of “hydrangea blue-complex” (2) was recently determined.21 The key component of the aluminium complex is the anhydrobase form of delphinidin, this allows binding of the anthocyanin to the Al3+ ion (see figure 2).

Figure 2: Anthocyanin dyes from hydrangeas (Hydrangea macrophylla)
The complexes formed between metal ions and anthocyanins can be very complicated structures since a variety of intramolecular and intermolecular copigmentation interactions can occur.5,6 A full description of these interactions is beyond the scope of this article, however, for clarity, figure 3 shows a simplified generic binding motif between metal ions and anthocyanins (figure 3, Motif A). A simplified intermolecular (π-π stacking) copigmentation interaction is also shown (figure 3, Motif B), in reality there is a rich array of potential non-covalent copigmentation interactions.

Figure 3: Representative anthocyanin/metal ion binding motifs
We reasoned that the underlying concepts relating to the role of metal binding to anthocyanins could make a very simple, but engaging, classroom or home study investigation for students. In this case, the effect of metal ions on the colour of anthocyanin solutions derived from different plant sources could be made. This year T. Kuntzleman published a lead study that investigated the effect of the addition of metal ions to solutions containing blueberry (Vaccinium angustifolium) dyes.22 The results from this work showed that addition of a variety of divalent and trivalent metal ions led to readily observable colour changes. Further investigations are now reported here, in this case using extracts from red cabbage, roses, and begonia has identified similar trends. We again focused on using materials that could be sourced easily from hardware stores or from online retailers. Iron(II) sulfate (FeSO4) and copper(II) sulfate (CuSO4) solutions are readily accessible in hundred gram quantities from hardware stores and online retailers since both compounds are sold for gardening purposes (see the supporting information document for further details). Potassium aluminium sulfate (KAl(SO4)2.12H2O) and magnesium sulfate (MgSO4.7H2O) are also available from various retailers as “alum” and “Epsom salts” respectively. The following section describes the general procedure and results for each trial, red cabbage extract was initially selected, although further results with flower, fruit and black turtle bean (Phaseolus vulgaris) extracts are provided in the supporting information document.
Addition of solutions containing Fe2+, Cu2+, Al3+ and Mg2+ to anthocyanin extracts from red cabbage
In each trial, a teaspoon (ca. 5-6 g) of the desired metal salt was dissolved in 100-150 mL of water to make a series of stock solutions. 5-6 drops of each stock solution were then added to anthocyanin mixtures extracted from plant material in water (ca. 5 mL). The solution was swirled for a few seconds and the resulting colours were observed (examples are illustrated in figure 4). A set of results that use red cabbage as a source of anthocyanins is presented here as an example, the supporting information outlines the anthocyanin extraction procedure.

Figure 4: Solutions of red cabbage extract mixed with Fe2+, Cu2+, Al3+ and Mg2+
The red cabbage extract became dark blue immediately after addition of iron(II) sulfate solution, this effect is consistent with previous observations.23 It has been reported that the anthocyanin/metal ion binding event leads to rapid autoxidation of bound Fe2+ to Fe3+.24 Similarly, addition of copper (II) sulfate afforded a dark blue solution. Addition of potassium alum or magnesium(II) sulfate resulted in purple coloured solutions, although the sample containing Al3+ exhibited a more obvious change since the solution was darker and slightly more blue/violet than the cabbage extract.24 In contrast, the sample containing Mg2+ was observed to be lighter and slightly more pink in colour (very similar to the original cabbage extract). Although not a specific objective of this activity, UV-visible absorption spectra of the sample solutions have been obtained and these are provided in the supplementary information. The spectra showed the solutions containing iron, copper and aluminium ions had absorptions at longer wavelengths than the cabbage extract, while the solutions containing magnesium ions showed very little difference. We have surveyed a variety of different flower and fruit extracts, the results of these are presented in the supporting information document. In many cases, sample solutions containing iron(II) sulfate and flower extracts, were violet or blue (red roses and red begonia for example). However, in some cases, flower extracts were also observed to be very dark (brown/black) after addition of iron(II) sulfate solution. It is difficult to say for certain what caused some samples to be very dark, however it is possible that complexes between tannins and iron (reminiscent of those found in gallotannate inks 25,26) could also be present.
Microscale addition of solutions containing Fe2+, Cu2+, Al3+ and Mg2+ to anthocyanin extracts from red cabbage
A microscale method for testing the effect of metal ions on the colour of anthocyanin solutions (extracted from plant material) was also tested. This approach has been found to be an effective way to demonstrate various reactions, such as precipitations and testing solution pH with indicators.27,28 Pleasingly, in this case, the colour changes observed in anthocyanin solutions due to the presence of metal ions were also clearly visible using microscale techniques (figure 5). The tests simply required 1 or 2 drops of extract to be spotted on a transparent plastic sheet, a small quantity of crystalline metal sulfate was then transferred to the spotted solution using a wooden cocktail stick and carefully mixed (a detailed procedure is outlined in the supporting information document). This approach allows many samples to be tested at the same time, once complete, the plastic sheet can be wiped clean using a paper towel or cloth.
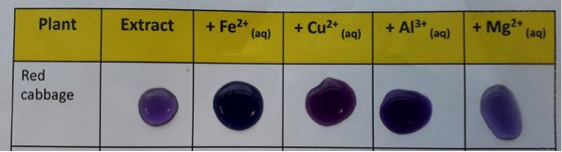
Figure 5: Microscale Solutions of red cabbage extract mixed with Fe2+, Cu2+, Al3+ and Mg2+
Hazards
Safety glasses are recommended for these activities. If performed as a home study activity, appropriate eye protection can be sourced from hardware stores or online retailers. Do not allow any of the substances in use to come into contact with skin or eyes. Care must be taken to avoid inhaling dusts when manipulating solid materials. All metal salt solutions should be disposed of according to local regulations. The packaging of compounds intended for garden use may also provide appropriate guidance.
Conclusion
In summary, we have examined reactions of aqueous anthocyanin extracts from red cabbage with stimuli other than varying pH. Instead, in this study, we have investigated the effect on the colour of red cabbage extracts when metal ions or bisulfite ions are added. The reported protocols can be implemented without the need for specialist equipment and the necessary chemicals can be obtained from grocery or hardware stores. We hope that the practical activities described will prove useful to educators as outreach or classroom activities, they may also prove useful as home study tasks.
Potential further activities - This report shows some preliminary results, however there should be plenty of scope to extend this activity further. Many flowers, fruits and vegetables contain anthocyanins, so there are many more opportunities for further testing. If accessible, other metal ions could also be examined. While it would not be possible to predict in all cases whether addition of metal ions would lead to a colour change, this activity could also be a good opportunity for students to plan and perform a short investigation that focused on observation skills, perhaps presenting their results in a short report, presentation or poster. As part of our investigation, we had access to a spectrophotometer, however in a school classroom or home study environment, a simpler alternative could be to use a free smartphone app to try and compare the colours of the solutions. A recent example of this type of analysis has been reported in the context of determining the concentration of a solution of copper(II) nitrate.29 There are also many recent developments that allow spectrophotometry experiments30 to be performed in the classroom or home without specialised equipment.
Acknowledgements – Firstly, we are very grateful to Prof. T. Kuntzleman for all the help and time he has given us in preparing this work, indeed his article this year on blueberries (ref 22) was one of the inspirations for the current study. We would also like to thank May Armstrong and Jessica Ross from the S6 class at Dornoch Academy for all of their efforts testing many of the procedures presented. Finally, we would like to thank Mrs G. C. Smellie and Mr B. F. Patterson for providing many of the flowers and gardening materials for the initial trials conducted in July/August 2020.
Supporting Information - Log into your ChemEd X account to access.
Experimental procedures are provided in addition to photographs from trials conducted with various flowers, fruit, vegetables and beans. A short film of the metabisulite/peroxide sequence is also provided.
References
1. Fortman, J. J.; Stubbs, K. M. Demonstrations with red cabbage indicator. J. Chem. Educ., 1992, 69 (1), 66.
2. Linder, J. L.; Aljic, S.; Shroof, H. M.; Di Giusto, Z. B;. Franklin, J. M.; Keaney, S.; Le, C. P.; George, O. K.; Castaneda, A. M.; Fisher, L. S.; Young, V. A.; Kiefer A. M. Exploring acid–base chemistry by making and monitoring red-cabbage sauerkraut: A fresh twist on the classic cabbage-indicator experiment. J. Chem. Educ., 2019, 96 (2), 304-307.
3. Stoddard, R. L.; McIndoe, J. S. The color-changing sports drink: An ingestible demonstration. J. Chem. Educ., 2013, 90 (8), 1032-1034.
4. Fogde, A.; Kurtén, B.; Sandberg, T.; Huynh, T. Colorimetric hydrogel from natural indicators: A tool for electrochemistry education. J. Chem. Educ., 2020, 97, ASAP
5. Trouillas, P.; Sancho-García, J. C.; De Freitas, V.; Gierschner, J.; Otyepka, M.; Dangles, O. Stabilizing and modulating color by codyeation: Insights from theory and experiment. Chem. Rev., 2016, 116, 4937-4982.
6. Yoshida, K.; Mori, M.; Kondo, T. Blue flower color development by anthocyanins: from chemical structure to cell physiology. Nat. Prod. Rep., 2009, 26, 884-915.
7. Chen, Y.; Wang Z.; Zhang, H.; Liu, Y.; Zhang, S.; Meng, Q.; Liu, W. Isolation of High Purity Anthocyanin Monomers from Red Cabbage with Recycling Preparative Liquid Chromatography and Their Photostability. Molecules, 2018, 23 (5), 991.
8. Dewprashad, B.; and Hadir, L. Developing an invisible message about relative acidities of alcohols in the natural products henna, turmeric, rose petals, and vitamin A. J. Chem. Educ., 2010, 87 (1), 36-39.
9. Selco, J. I. Using hands-on chemistry experiments while teaching online. J. Chem. Educ., 2020, 97, ASAP 10. Schultz, M.; Callahan, D. L.; Miltiadous, A. COVID-19 Development and use of kitchen chemistry home practical activities during unanticipated campus closures. J. Chem. Educ., 2020, 97, ASAP
11. Holme, T. A. Journal of Chemical Education Call for Papers: Special Issue on Insights Gained While Teaching Chemistry in the Time of COVID-19. J. Chem. Educ., 2020, 97, 1226-1227.
12. Boucher, M. M.; Furigay, M. H.; Quach, P. K.; Brindle, C. S. Liquid−Liquid extraction protocol for the removal of aldehydes and highly reactive ketones from mixtures. Org. Process Res. Dev., 2017, 21, 1394-1403.
13. Castka, J. F. Grape juice as an indicator. J. Chem. Educ., 1962, 39 (5), A431.
14. Solomon, S.; Oliver-Hoyo, M.; Hur, C. Generating water-soluble noxious gases: an overhead projector demonstration. J. Chem. Educ., 1998, 75 (12), 1581-1582.
15. McClure, D. W. The chemistry of winemaking and brewing. J. Chem. Educ., 1976, 53 (2), 70–73.
16. Sims, C. A.; Morris, J. R. Effects of pH, sulfur dioxide, storage time, and temperature on the color and stability of red muscadine grape wine. Am. J. Enol. Vitic., 1984, 35, 35-39.
17. Berké, B.; Chèze, C.; Vercauteren, J.; Deffieux, G. Bisulfite addition to anthocyanins: revisited structures of colourless adducts. Tetrahedron Lett., 1998, 39, 5771-5774.
18. Andrea-Silva, J.; Cosme, F.; Ribiero, L. F.; Moreiira, A. S. P.; Malheiro, A. C.; Coimbra, M. A.; Domingues, M. R. M.; Nunes, F. M. Origin of the pinking phenomenon of White Wines. J. Agric. Food Chem., 2014, 62 (24), 5651-5659.
19. “The Origin of the Campden Tablet”, http://cider.org.uk/campden.html (accessed 25th August 2020)
20. Hoffmann, M.; Edwards, J. Kinetics of the oxidation of sulfite by hydrogen peroxide in acidic solution. J. Phys. Chem., 1975, 79 (20), 2096−2098.
21. Ito, T.; Aoki, D.; Fukushima, K.; Yoshida, K. Direct mapping of hydrangea blue-complex in sepal tissues of Hydrangea macrophylla. Sci. Rep., 2019, 9, 5450-.
22. Kuntzleman, T. Berries are red, berries are blue...I’ve got a berry surprise for you!, Chemical Education Xchange (chemedx.org). https://www.chemedx.org/blog/berries-are-red-berries-are-blue (accessed 25th August 2020)
23. Fenger, J.; Moloney, M,; Robbins, R. J.; Collins, T. M.; Dangles, O. The influence of acylation, metal binding and natural antioxidants on the thermal stability of red cabbage anthocyanins in neutral solution. Food Funct., 2019, 10, 6740-6751.
24. Bernal, F. A.; Orduz-Diaz, L. L.; Coy-Barrera, E. Exploitation of the complexation reaction of ortho- dihydroxylated anthocyanins with aluminum(III) for their quantitative spectrophotometric determination in edible sources. Food Chemistry, 2015, 185, 84-89.
25. Martín-Gil, J.; Ramos-Sánchez, M. C.; Martín-Gil, F. J. Chemical composition of a fountain pen ink. J. Chem. Educ. 2006, 83 (10), 1476–1478.
26. Ponce, A.; Brostoff, L. B.; Gibbons, S. K.; Zavalij, P.; Viragh, C.; Hooper, J.; Alnemrat, S.; Gaskell, K. J.; Eichhorn, B. Elucidation of the Fe(III) gallate structure in historical iron gall ink. Anal. Chem. 2016, 88 (10), 5152– 5158.
27. Worley, B.; Villa, E. M.; Gunn, J. M.; Matteson, B. Visualizing dissolution, ion mobility and precipitation through a low-cost, rapid-reaction activity introducing microscale precipitation chemistry. J. Chem. Educ. 2019, 96 (5), 951–954.
28. “Chemistry on a plastic sheet”, https://microchemuk.weebly.com/8-chemistry-in-small-volumes.html (accessed 25th August 2020)
29. Montangero, M. Determining the amount of copper(II) ions in a solution using a smartphone. J. Chem. Educ. 2015, 92 (10), 1759–1762.
30. Michelle L. Kovarik*, Julia R. Clapis, and K. Ana Romano-Pringle. Review of student-built spectroscopy instrumentation projects. J. Chem. Educ. 2020, 97 (8), 2185–2195.