The Density Bottle is Red, the Density Bottle is Blue...

The Density Bottle experiment has inspired many colorful variations over the years.1-6 I recently came across yet another colorful variation that I’d like to discuss. Check it out in the video below (Video 1)
Video 1: Bi-Color Science Experiment, Tommy Technetium YouTube channel.
In this demonstration, neon purple food dye, water, acetone, and salt are mixed. The mixture separates into a two-layer system with a pink acetone layer on top and a blue saltwater layer on the bottom. This phenomenon, known as the “salting out” effect, commonly happens when salt is added to a mixture of water and an organic solvent, causing the layers to separate.
The pink-blue color separation observed here has been reported previously,2 but I would like to add some details on how the color separation occurs. The neon purple food dye used in the experiment (Video 1) contains a mixture of Blue Dye #1 (Figure 1, left) and Red Dye #3 (Figure 1, right).
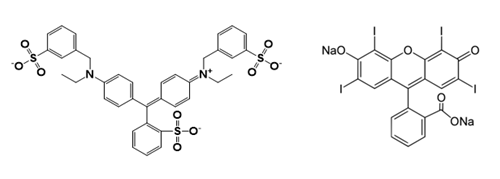
Figure 1: Chemical structures of (left) Blue Dye #1 (MW = 792 g mol-1) and (right) Red Dye #3 (MW = 880 g mol-1).
During the salting out process, the layer into which dyes dissolve depends on several factors, with the most important being the size of the dye.7-9 This is because for a dye molecule to dissolve, it must “make room” for itself by “pushing apart” water molecules. This process is endothermic because there is an energy cost for separating the hydrogen bonding between water molecules. Since larger molecules must separate more water molecules to dissolve, this energy cost increases with molecular size. However, this energy cost can be offset by the release of energy through hydrogen bonding interactions between the water and the dye molecule.
Now we are ready to understand how the blue dye ends up in the water layer, but the red dye does not. First, the blue dye (MW = 792 g mol-1) is 10% smaller than the red dye (MW = 880 g mol-1), so the energy cost for dissolving it in water is lower than that of the red dye. Second, the blue dye contains over twice as many atoms (11) capable of accepting hydrogen bonds from water, as compared to the red dye (5) (to see this, count the number of nitrogen and oxygen atoms on each molecule). So the blue dye can release more energy through hydrogen bonding than the red dye when it dissolves in the water layer.
It must be stressed again that the dissolution of dyes into various layers is complex and depends on a wide variety of factors, not just molecular size and hydrogen bonding capabilities.7-9 Nevertheless, the color separations observed when various dyes are included in salting out experiments can often be described using these two characteristics.6
One final note: beginning January 2027, the United States will no longer allow Red Dye #3 in food products.10 Thus, if you want to try this experiment in your classes, you might want to stock up on purple food dye that contains Red Dye #3. I have observed that a mixture of Blue Dye #1 and the more common Red Dye #40 does result in a pink-red color separation, but the color separation is not as sharp.
As always, if you try this experiment or any variations, I’d love to hear your results.
Happy Experimenting!
References
- Kuntzleman, T., Chemical Mystery #8: Go Blue! November 2016.
- Kuntzleman, T., Solution to Chemical Mystery #8: Go Blue! November 2016.
https://www.chemedx.org/blog/solution-chemical-mystery-8-go-blue
- Kuntzleman, T., The Density Bottle Strikes Again, December 2017.
- Webb, J. and Rothenberger, O., The Salting-Out Effect: A Colorful Demonstration That Leads to Student - Teacher Activities, April 2023.
- Fleming, D., Battle of the Forces(link is external), March 2023.
- Kuntzleman, T. Exploring Color Separation in Salting-Out Experiments May 2024.
https://www.chemedx.org/blog/exploring-color-separation-salting-out-exp…
- Endo, S.; Pfennigsdorff, A.; Goss, K.-U. Salting-Out Effect in Aqueous NaCl Solutions: Trends with Size and Polarity of Solute Molecules. Environ. Sci. Technol. 2012, 46, 1496-1503.
- Hyde, A. M.; Zultanski, S. L.; Waldman, J. H.; Zhong, Y.-L.; Shelvin, M.; Peng, F. General Principles and Strategies for Salting-Out Informed by the Hofmeister Series. Org. Proc. Res. Dev. 2017, 21, 1355-1370.
- De Stefano, C.; Lando, G.; Malegori, C. Oliveri, P. Sammartano, S. Prediction of water solubility and Setschenow coefficients by tree-based regression strategies. J. Molec. Liq. Dev. 2019, 282, 401-406.
- https://cen.acs.org/food/food-ingredients/FDA-bans-red3-food-drugs/103/…