misconceptions/discrepant events
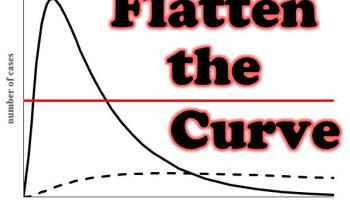
Misconceptions refer to common, erroneous beliefs that students use to understand and explain the world around them. Such assumptions often become entrenched and resist modification by traditional instruction. A discrepant event is an experiment, demonstration, or other occurrence that challenges a misconception by producing a result counter to that predicted by the mistaken belief. When this happens, students are forced to reevaluate and adjust their conceptual framework in order to account for the discrepant event.
Term source
jce