Solution to Chemical Mystery #14: Loves Me...Loves Me Not

In Chemical Mystery #14, neutralization reactions were used to cause some interesting color changes. See the two videos below:
In both experiments an indicator was used that contained a mixture of p-nitrophenol and phenolphthalein (Table 1). p-nitrophenol changes from colorless to yellow around pH = 6, while the phenolphthalein changes from colorless to pink around pH = 9. Mixing these two substances creates an indicator that changes from colorless to yellow at pH = 6, and then from yellow to red at pH = 9 (Table 1).
Table 1: Colors of indicators at various pH levels.
|
Indicator |
Color at pH incidated |
||
|
Below 6 |
6-9 |
Above 9 |
|
|
p-nitrophenol |
Colorless |
Yellow |
Yellow |
|
Phenolphthalein |
Colorless |
Colorless |
Pink |
|
(mixture of phenolphthalein and p-nitrophenol) |
Colorless |
Yellow |
Red |
The first experiment is easy to set up and carry out, while the second is a bit more complicated. Detailed instructions for setting up and carrying out each experiment are listed in the appendix to this blog post.
Explanation of the first experiment: In the first experiment, color changes from red to colorless and back to red were observed. To pull this off, cups were set up with varying amounts of acid, base and indicator formed from the mixture of p-nitrophenol and phenolphthalein (figure 1). If you look carefully, you will notice that cups with “lips” at the bottom were specifically chosen for this experiment. This was done to hide the fact that small amounts of liquid poured into the bottom of many of the cups. Thus, at the start of the experiment, it looked as if only the first cup (A) had any liquid in it. At the start of the experiment only cup B contained any indicator, but because the contents of cup B were acidic (pH = 2), the indicator appeared to be colorless.
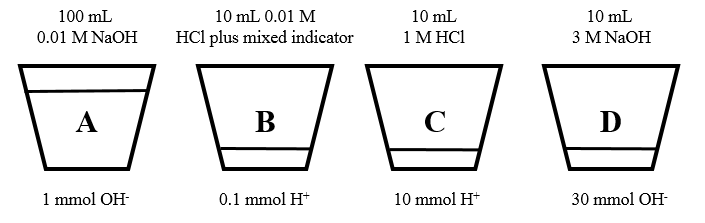
Figure 1: Concentrations and amounts of acid, base, and indicator in the red-to-colorless experiment.
The simple explanation is that when cup A was poured in cup B, a solution with a pH > 9 resulted, causing the contents of the mixture to appear red. When the resulting contents of cup B were poured in cup C, an excess of acid resulted, changing the mixture colorless. Finally, when the resulting mixture in cup C was poured in cup D, an excess of base with pH > 9 was produced, changing the color back to red. A quantitative description of the reactions and pH values that result from each mixing experiment can be found in the appendix.
Explanation of the second experiment: In the second experiment, color changes from red to colorless to yellow to colorless and back to red were observed. Again, cups were prepared with varying amounts of acid and base (figure 2). To achieve the yellow color, sodium bicarbonate (a weak base) rather than sodium hydroxide (a strong base) was used. I’ll discuss what goes on in the first six cups in this experiment (A-F), and from this you should be able to figure out what happens with the last four cups (G-J).
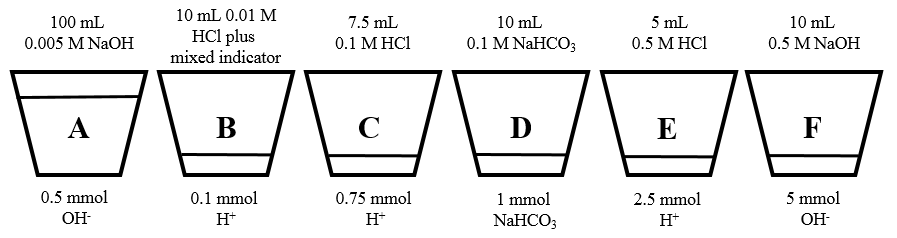
Figure 2: Concentrations and amounts of acid, base, and indicator in the first six cups of the red-colorless-yellow-colorless-red experiment. Not shown: cup G (7.5 mmol H+), cup H (10 mmol NaHCO3), cup I (15 mmol H+), and cup J (30 mmol OH-).
The start of this experiment looked like the first experiment: when cup A was poured into cup B, a solution with a pH > 9 resulted, causing the contents of the mixture to appear red. Also, when the resulting contents of cup B were poured in cup C, an excess of acid resulted, changing the mixture colorless. However when cup C was poured into cup D, a yellow color resulted. This is because the weak base (bicarbonate) present in cup D only increased the pH of the resulting solution to 8.3: high enough to cause a color change to yellow, but not high enough to shift to red. Upon pouring the resulting contents of cup D were poured into cup E, the mixture was acidified, causing a shift back to colorless. Finally, pouring the resulting contents of cup E into cup F caused a shift back to high pH and a change to red color. Again, a quantitative description of the reactions and pH values that result from these mixing experiments can be found in the appendix.
It should be possible to create all sorts of color changing variations using different combinations of indicators, strong acids, strong bases, weak acids, and weak bases. I think it would be a great exercise to challenge students to come up with a particular color changing sequence. For example, is it possible to have colors alternate red-yellow-red-yellow-red without an intervening colorless step? Or, what is the largest number of cups you can get to alternate back and forth between color changes? Please do let me know if you and your students discover some interesting variations!
Appendix: Detailed instructions for setting up each experiment.
Preparation of the indicator mixture: Dissolve both 1.5 g of p-nitrophenol (pKa = 7.2) and 0.75 g phenolphthalein (pKa = 9.7) into 30 mL of 95% ethyl alcohol. Alternatively, the red indicator purchased as part of the Disappearing Rainbow Chemical Demonstration kit can be used. It should be noted that I used the red indicator purchased as part of the kit in all experiments presented in the videos.
Experiment 1: red to colorless to red
Prepare four cups as follows:
Cup A: 100 mL of 0.01 M NaOH
Cup B: 10 mL of 0.01 M HCl + 5 drops of indicator mixture.
Cup C: 10 mL of 1 M HCl
Cup D: 10 mL of 3 M NaOH
When ready to present, pour the contents of cup A into cup B. Then pour the contents of cup B into cup C. Finally, pour the contents of cup C into cup D.
Experiment 2: red to colorless to yellow to colorless to red
Prepare 10 cups as follows:
Cup A: 100 mL of 0.005 M NaOH
Cup B: 10 mL of 0.01 M HCl + 5 drops of indicator mixture.
Cup C: 7.5 mL of 0.1 M HCl
Cup D: 10 mL of 0.1 M NaHCO3
Cup E: 5 mL of 0.5 M HCl
Cup F: 10 mL of 0.5 M NaOH
Cup G: 7.5 mL of 1 M HCl
Cup H: 10 mL of 1 M NaHCO3
Cup I: 5 mL of 3 M HCl
Cup J: 5 mL of 6 M NaOH
When ready to present, pour the contents of cup A into cup B. Then pour the contents of cup B into cup C, cup C into cup D, and so forth finishing with pouring the contents of cup I into cup J.
Quantitative description of the first experiment:
1. Cup A into cup B: First, cup A was poured into cup B, causing the following chemical reaction:
H+ + OH- à H2O Equation 1
The 1 mmol of OH- in cup A completely neutralized the 0.1 mmol of H+ in cup B, leaving an excess of 0.9 mmol of OH- in 110 mL of solution. The pOH of the resulting mixture was equal to –log(0.0009/0.110) = 2.1, which corresponds to a pH = 11.9 – and the solution turned red!
2. Cup B into cup C: Now when the resulting contents of cup B were poured into cup C, the 10 mmol of H+ in cup C neutralized the remaining 0.9 mmol of OH- from cup B. This left an excess of 9.1 mmol of H+, and the solution turned colorless.
3. Cup C into cup D: Finally, when the resulting contents of cup C were poured into cup D, the 0.30 mmol of OH- present neutralized the remaining 9.1 moles of H+. I’ll leave it to the reader to show that the pH of the final solution was 13.2, which is why the solution turned red. Remember, there is a total volume of 130 mL in cup D after the mixing is over!
Quantitative description of the second experiment:
1. Cup A into cup B: Cup A was first poured into cup B, causing the following chemical reaction:
H+ + OH- à H2O Equation 1
The 0.5 mmol of OH- in cup A completely neutralized the 0.1 mmol of H+ in cup B, leaving an excess of 0.4 mmoles of OH- in 110 mL of solution. The pOH of the resulting mixture was equal to –log(0.0004/0.110) = 2.1, corresponding to a pH = 11.6 and a resulting red solution.
2. Cup B into cup C: Now when the resulting contents of cup B were poured into cup C, the 0.75 mmol of H+ in cup C neutralized the remaining 0.4 mmol of OH- from cup B, leaving an excess of 0.35 mmol of H+ in a colorless solution.
3. Cup C into cup D: When the resulting contents of cup C were poured into cup D, the following reaction took place between the 0.35 mmol of H+ remaining in cup C and the 1 mmol of HCO3- present in cup D:
H+ + HCO3- à CO2 + H2O Equation 2
At the conclusion of the reaction, 0.65 mmol of HCO3- remained in a total of 130 mL of solution, yielding a solution containing 0.005 M HCO3-. Using Kb = 2 x 10-8 for HCO3- (and assuming no CO2 formed remains dissolved in solution) it can be shown that the resulting solution has a pH = 9: right on the border of the color change from yellow to red. Experimentally however, this solution appeared yellow, very likely because CO2 remaining in solution lowered the pH below the critical value of pH = 9.
4. Cup D into cup E: When the resulting contents of cup D were poured into cup E, the remaining 0.65 mmol of HCO3- were completed neutralized by the 2.5 mmol of H+ in cup E via Equation 2. This left an excess of 1.85 mmol H+, resulting in a colorless solution.
5. Cup E into cup F: When the resulting contents of cup E were poured into cup F, the remaining 1.85 mmol of H+ were completed neutralized by the 5.0 mmol of OH- in cup F. This left an excess of 3.15 mmol OH- and a red solution.
The results of the remaining mixing events will be left for the interested reader to calculate.