Generating explosive gases from sand

Introduction: Did you know that sand can be converted into a mixture of gases that spontaneously ignites in air? The procedures involved are relatively simple to perform, spectacular to observe, and relate to a rich assortment of chemical principles.
To start the process, sand and granular magnesium (I use 40-80 mesh) are combined and heated in a test tube. This results in the reduction of silicon dioxide in sand by magnesium to form magnesium oxide and elemental silicon:1,2
SiO2(s) + 2 Mg à 2 MgO(s) + Si(s) Equation 1
You can watch a quick synopsis of this extraction of elemental silicon from sand in the video below (Video 1):
Video 1: Getting silicon out of sand, pchemstud on TikTok. Dec 9, 2021
In conjunction with the above process, magnesium silicide (Mg2Si) is formed due to the following reactions:1-3
SiO2(s) + 4 Mg(s) à Mg2Si(s) + 2 MgO(s) Equation 2
Si(s) + 2 Mg(s) à Mg2Si(s) Equation 3
Once the test tube has cooled, dilute HCl is added to the products. The acid reacts with Mg2Si to produce a variety of silicon-based gases that are analogous to the carbon-based alkanes:4-5
Mg2Si(s) + 4 HCl(aq) à SiH4(g) + 2 MgCl2(aq) Equation 4
2 Mg2Si(s) + 8 HCl(aq) à Si2H6(g) + 4 MgCl2(aq) + H2(g) Equation 5
3 Mg2Si(s) + 12 HCl(aq) à Si3H8(g) + 6 MgCl2(aq) + 2 H2(g) Equation 6
4 Mg2Si(s) + 16 HCl(aq) à Si4H10(g) + 8 MgCl2(aq) + 3 H2(g) Equation 7
This group of gases is collectively termed the silanes. In particular, SiH4 is called silane, Si2H6 disilane, Si3H8 trisilane, and Si4H10 is called tetrasilane. The silanes formed spontaneously ignite on exposure to oxygen in the air, and this produces a lot of sparks:
SiH4(g) + 2 O2(g) à SiO2(s) + 2 H2O(l) Equation 8
2 Si2H6(g) + 7 O2(g) à 4 SiO2(s) + 6 H2O(l) Equation 9
Si3H8(g) + 5 O2(g) à 3 SiO2(s) + 4 H2O(l) Equation 10
2 Si4H10(g) + 13 O2(g) à 8 SiO2(s) + 10 H2O(l) Equation 11
Note that these spark-forming reactions are analogous to the combustion of alkanes.5 However, alkane combustion generally requires a source of ignition under normal temperature and pressures:
CH4(g) + 2 O2(g) à CO2(s) + 2 H2O(l) Equation 12
2 C2H6(g) + 7 O2(g) à 4 CO2(s) + 6 H2O(l) Equation 13
C3H8(g) + 5 O2(g) à 3 CO2(s) + 4 H2O(l) Equation 14
2 C4H10(g) + 13 O2(g) à 8 CO2(s) + 10 H2O(l) Equation 15
The video below (Video 2) outlines the various experiments involved in generating flammable gases from sand. These processes feature the chemical reactions that occur when a mixture of sand and magnesium is heated (Equations 1-3), when acid is added to magnesium silicide (Equations 4-7) and the combustion of silanes (Equations 8-11).
Video 2: Converting Sand Into Flammable Gas, Tommy Technetium YouTube Channel. Dec 30, 2021
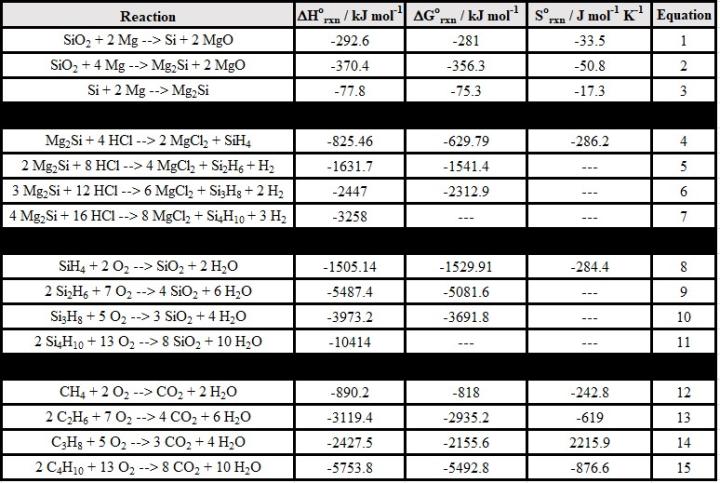
Discussion: These experiments connect to a variety of chemical topics. Standard enthalpies of formation (Table 1) may be used to calculate the standard enthalpies of all reactions involved. Standard Gibbs energies and standard entropies of reactions may also be calculated. The standard enthalpies, Gibbs energies, and entropies of all reactions listed herein are tabulated in the Appendix.
Table 1: Selected thermodynamic values6-7
It is interesting to compare the enthalpies of combustion (Figure 1) of each silane (Equations 8-11) to the enthalpy of combustion of its corresponding alkane (Equations 12-15):
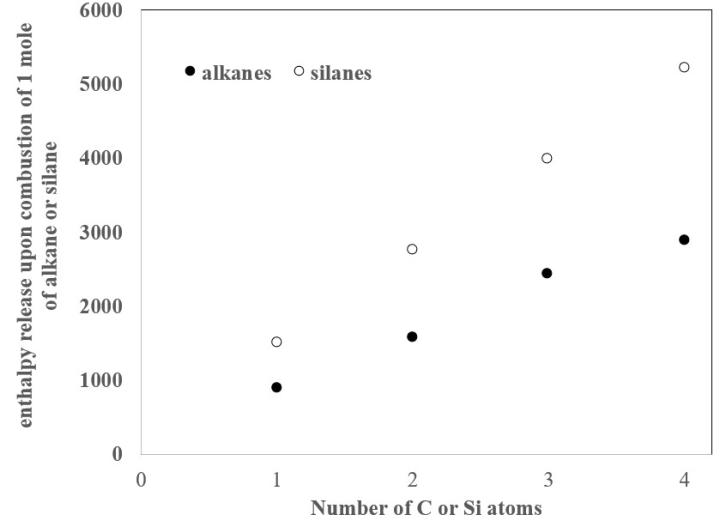
Figure 1: Enthalpy released upon combustion of the silanes and alkanes.
The fact that the silanes release more energy on combustion than the alkanes indicates that alkanes are more stable than silanes in the presence of oxygen. This fact harmonizes well with the observation that the silanes spontaneously combust in air, while the alkanes require a source of ignition to combust. Furthermore, the stability of the alkanes as compared to the silanes provides insight into the observation that long-chained molecules comprised of carbon, but not silicon, are ubiquitous. This latter observation has clear implications for why carbon-based molecules rather than silicon-based molecules provide the molecular foundation for life.
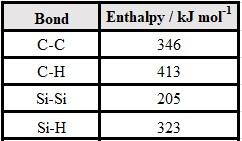
These ideas can be expanded upon further by comparing the average bond enthalpies of various chemical bonds that contain carbon and silicon (Table 2). Notice that on average it requires 141 kJ mol-1 more energy to break the carbon-carbon bond as compared to the silicon-silicon single bond. Similarly, it takes an average of 90 kJ mol-1 more energy to break the carbon-hydrogen bond as compared to the silicon-hydrogen bond. The alkanes are thus more resistant to chemical bond breaking – and therefore more resistant to chemical change – than the silanes. Further discussion on the stability of carbon-chained molecules vs. silicon-chained molecules may be found in reference 8.
Table 2: Average bond enthalpies.8,9
Conclusion: The process of converting sand into flammable silanes is intensely interesting to observe, and it entails a rich assortment of chemical topics. The array of reactions (Equations 1-11) related to the process provide a platform to discuss concepts in chemical thermodynamics and chemical bonding. Comparing the thermodynamics of silane combustion to alkane combustion allows for a discussion of the ability of carbon to form stable, long-chained molecules. Given the wide array of chemical reactions relevant to this experiment, there are certainly other chemical topics that could be discussed. I’d love to hear what chemical subjects come to your mind as you try out these experiments. I look forward to hearing your thoughts in the comments.
Happy experimenting!
Appendix: Standard enthalpies, Gibbs energies, and entropies of reactions outlined in Equations 1-15.
References:
- Borshchev, D’yachenko, Kiselev, and Kraidenko, Russian Journal of Applied Chemistry, 2013, 86 (4), 493−497.
- Favors, Wang, Hosseini Bay, Mutlu, Ahmed, Liu, Ozkan, and Ozkan, Scientific Reports, 2014, 4, 5623.
- Takamori, Osawa, Kimura, Liu. and Mukai, Materials Transactions, 2008, 49, (5) (2008), 1089-1092.
- Johnnson and Isenberg, J. Am. Chem. Soc., 1935, 57, 1349-1353.
- Johnnson, J. Chem. Educ., 1934, 11 (4), 256.
- Dean, Lange’s Handbook of Chemistry, 12th ed.; McGraw-Hill: New York, New York, 1979; pp. 9-4 - 9-94.
- Sax and Kalcher, J. Phys. Chem., 1991, 95 (4) 1768-1783.
- Smith, J. Chem. Educ., 1988, 65 (5), 414-415.
- Kotz, Treichel, and Weaver Chemistry and Chemical Reactivity, 6th ed.; Thomson Learning: Belmont, CA, 2006, p. 422.
Comments
5Comparing stability
Hi Tom,
It is a very interesting set of reactions and the procedures are so simple but the effects are spectacular. I think typical high school students will very much enjoy this experiment.
I have one question in regard to your discussion. As you mention both C-C and C-H bonds are more stable than Si-Si and Si-H bonds, should we say alkane is more stable than silane? The fact silane spontaneously combusts in the air but alkane needs some initial ignition. I am confused about your statement "The fact that the silanes release more energy on combustion than the alkanes indicates that silanes are more stable than alkanes in the presence of oxygen."
Yu-Sung
In reply to Comparing stability by Yu-Sung Wu
Thanks for pointing out my error!
Yu-Sung:
You are indeed correct: The alkanes are more stable than the silanes. Thank you so much for catching my mistake. I will fix the erroneous sentence you cited to reflect this.
With appreciation,
Tom
Share Your Thoughts