Dry ice in five different liquids

I recently posted a video on Twitter of an experiment my students were conducting in class. I thought I’d blog about the experiment, since it seemed to generate a lot of interest. In the experiment, solid carbon dioxide (dry ice) is placed in three different liquids at room temperature: glycerol, ethanol, and water. You can watch the video here:
I think the differences observed are fascinating. In each liquid, the dry ice sublimes:
CO2(s) -->CO2(g)
The sublimation of dry ice causes bubbles to form within each liquid. However, several differences are observed, depending upon the liquid into which the dry ice is placed. Large, slowly rising bubbles are formed in glycerol, but no fog is produced. In ethanol, a rush of tiny bubbles is produced that move chaotically and rise rapidly. Looking carefully, one notices a thin and wispy fog. When dry ice is placed in water, large, rapidly rising bubbles and a thick cloud are observed.
I recently went into the lab to extend this experiment. I wanted to see what happened when dry ice was added to acetone and hot glycerol (over 150oC). You can see the results in the video below:
Addition of dry ice to acetone causes a frantic burst of tiny bubbles that almost looks like a tiny explosion! Also, a very thin cloud is produced. When dry ice is placed in hot glycerol, large bubbles that rise rapidly and a thick, sticky cloud are formed. Table 1 summarizes the results from all 5 liquids:
|
Liquid |
Size of bubbles produced |
Character of bubble motion |
Cloud produced? |
Character of cloud produced |
|
Water |
large |
rapid rise |
yes |
thick, persistent |
|
Glycerol |
large |
slow rise |
no |
not applicable
|
|
hot glycerol |
large |
rapid rise |
yes |
very thick and sticky, persistent |
|
Ethanol |
small |
very rapid, chaotic rise |
yes |
thin, transient |
|
Acetone |
very small |
extremely rapid, extremely chaotic rise |
yes |
very thin and very transient |
Table 1 -Bubbling behavior and cloud characteristics observed when dry ice is placed in various liquids.
In addition to the observations listed in the table, I also noted that the time it takes for dry ice to sublime varies in each liquid. In the first three liquids (water, glycerol, and hot glycerol), the dry ice took a very long time to completely sublime. In ethanol, the dry ice took a minute or two to fully sublime. In acetone, the dry ice sublimed away in less than a minute!
I’m wondering if it would make a good lesson to have students carry out these experiments and then try to explain differences observed on the basis of the physicochemical properties of each liquid (surface tension, vapor pressure and viscosity). I have tried something similar with my students in laboratory, but not with all five liquids. Also, I have never had students focus on all the parameters (bubble size, character of bubble motion, character of cloud produced, and sublimation time) and liquid properties listed herein. If I try out this experiment, it will certainly be helpful to list the properties of each type of liquid (Table 2). I also think a discussion of the strength of intermolecular forces between molecules in each liquid would be helpful.
|
Liquid |
Surface tension / mN m-1 |
Vapor pressure / torr |
Viscosity / cP |
|
water |
73 |
18 |
1.00 |
|
glycerol |
63 |
0.0003 |
1410 |
|
hot glycerol |
52 |
4.5 |
3.8 |
|
ethanol |
23 |
44 |
1.26 |
|
acetone |
24 |
182 |
0.32 |
Table 2 - Properties of liquids at 20oC (except for hot glycerol) used in this experiment. Properties for hot glycerol at 150oC.
Perhaps you and your students would like to try out this experiment and come up with your own explanations for what you observe. If this is the case, don’t read on, because below I’ll be sharing how I currently think about the different results based on the properties of each liquid.
Bubble motion: Speed of bubble rise seems to correlate somewhat with viscosity. This makes sense if one considers that viscosity is defined as resistance to flow. This correlation is brought home most emphatically if one compares the slow bubble rise in glycerol (viscosity = 1410 cP) with the explosive bubble flow in acetone (viscosity = 0.32 cP).
Bubble size: Bubble size appears to correlate well with surface tension. Liquids with high surface tension (>50 mN m-1) tend to form large bubbles while those with low surface tension (~25 mN m-1) tend to form small bubbles. This difference can be approached semi-quantitatively using the Laplace pressure:
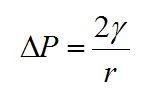
Where
is the difference in pressure inside and outside a spherical gas bubble in a liquid,
is the surface tension of the liquid and r is the radius of the bubble. If we assume a similar pressure difference in each experiment and rearrange the above equation we find that the bubble radius depends upon the surface tension:
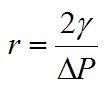
Thus, we would expect larger bubbles in liquids with higher surface tension, in agreement with observations.1
Time for dry ice to sublime: Solid pellets of dry ice take a long time to sublime away when placed in water, glycerol, or hot glycerol. On the other hand, dry ice sublimes away fairly quickly when placed in ethanol. And when placed in acetone, the dry ice sublimes away in less than a minute! How can these differences be explained? When dry ice is placed in water or glycerol, the dry ice undergoes film state sublimation (Figure 1). In this case a single large bubble forms a film around the solid dry ice. This film forms a protective barrier around the solid dry ice that insulates it from the bulk liquid. Because of this insulating barrier the transfer of energy from the bulk liquid to the dry ice occurs slowly, making the dry ice sublime away slowly.

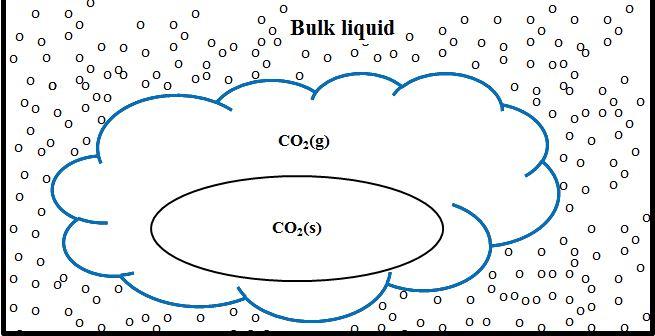
Figure 1 -In film state sublimation, a large bubble forms a protective insulating film around the solid dry ice. There is no direct contact between the liquid molecules and the solid dry ice. The oval in the center represents solid dry ice, the cloud surrounding the oval represents the protective film of CO2 gas, and the small circles represent liquid molecules.
When dry ice is placed in ethanol or acetone, the dry ice undergoes nucleate state sublimation (Figure 2). During nucleate state sublimation, an enormous number of tiny bubbles form on the surface of the solid dry ice. In this case, no protective insulating barrier is formed. Rather, the dry ice comes into direct contact with the bulk liquid and energy transfer is rapid. Thus, the dry ice sublimes away quickly – in fact VERY quickly in acetone! It is interesting to note that film state sublimation requires large bubbles, so it makes sense that this type of sublimation occurs in liquids with high surface tension. Many small bubbles form in nucleate state sublimation, so it makes sense that this type of sublimation would occur in liquids with low surface tension.

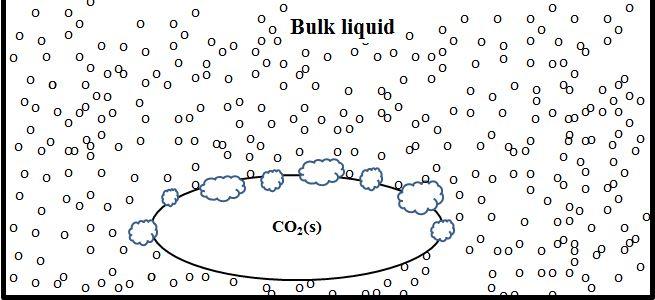
Figure 2 - During nucleate state sublimation, many tiny bubbles form on the surface of the dry ice. No protective barrier is formed, so liquid molecules may come into direct contact with the solid dry ice. The oval in the center represents solid dry ice, the several clouds surrounding the oval represent CO2 bubbles, and the small circles represent liquid molecules.
It now makes sense why the dry ice sublimes quickly in acetone and ethanol but slowly in the other liquids. The tiny bubbles observed when dry ice is placed in ethanol and acetone are indicative of nucleate state bubbling. Thus, acetone or ethanol molecules in the bulk liquid can rapidly transfer energy to the solid dry ice through direct contact of molecules, causing speedy dry ice sublimation. The large bubbles formed in glycerol, hot glycerol, and water indicate film state sublimation. In these liquids, the bubbles produced form a film that prevents liquid molecules from directly contacting the solid dry ice. Thus, the transfer of energy from liquid to dry ice is sluggish and the dry ice sublimes slowly.
Cloud Production and Persistence
A simple way to relate cloud production to vapor pressure is to state that cloud production requires a liquid to have a high enough vapor pressure. This is evidenced by the observation that only glycerol does not form a cloud of any sort, while a cloud is formed in all other liquids. However, it appears that if the vapor pressure is too high, a thin and transient cloud results (ethanol and acetone form very thin and transient clouds, while water and hot glycerol form thick and persistent ones). Thus, it appears that in order to achieve a thick, long-lasting cloud, dry ice should be placed in a liquid with a vapor pressure that is neither too high nor too low.
In order to understand why this is so, it is important to note that any cloud produced in this experiment comes from the liquid into which the dry ice is placed. Thus, when dry ice is placed in acetone, a fog consisting of tiny liquid droplets of condensed acetone is formed. Likewise, dry ice in water forms tiny liquid droplets of condensed water vapor, and dry ice in hot glycerol forms a glycerol fog. You can read more about how this might happen here http://pubs.acs.org/doi/abs/10.1021/ed400754n or here https://www.chemedx.org/blog/dry-ice-water-cloud .
Now we can make sense of the persistence of each fog formed. Tiny droplets of acetone will evaporate quickly because liquids with high surface tension evaporate easily. On the other hand, tiny droplets of glycerol will persist for quite some time, because liquids with low surface tension do not evaporate easily
To summarize: a high enough vapor pressure is required to get a cloud to form in the first place, but once the cloud is formed, its liquid droplets will persist longer if that liquid has a low vapor pressure. Isn’t it fantastic that plain old water at room temperature strikes a perfect balance of surface tension so that clouds are produced upon adding dry ice to it?
If you try this experiment out in your classroom (or on your own) please be sure to let me know in the comments. How did things work out? Did you try any extensions? I also welcome comments and criticisms on my explanations. Where do you think my explanations are on track? Where am I off the mark? Do you have any experiments that might help convince me to change my current thinking?
Happy experimenting!
Acknowledgements: Thanks to to Winthrop Chemistry (@WinthropChem) for alerting me to consider liquid viscosities and intermolecular forces in this experiment.
Additional Notes:
1. If you’d like to take this a bit further, note that the surface tension of water is about 3 times bigger than that of ethanol or acetone. Assuming spherical bubbles, recognizing that r is direction proportional to g, and noting that volume depends upon r3, we would expect bubbles in water to be approximately 9 times bigger than those observed in ethanol or acetone. This seems to fit pretty well with observations. I think a good exercise might be to have students film the bubbles formed in this experiment using slow motion video (most smart phones can do this). Students could then take measurements of the bubbles to quantitatively test the prediction that bubble radius is directly proportional to surface tension.