The WEIRDEST Chemical Reaction I've Ever Seen!

Galinstan is the tradename for a liquid metal alloy that is comprised of three elements: gallium (68.5%), indium (21.5%), and tin (10%).1 It behaves a lot like mercury but has significantly lower toxicity. Because of this, Galinstan is used as a replacement for mercury in a variety of applications. For example, Bingxing Wang and coworkers reported how Galinstan can be used in place of mercury in the famous mercury “beating heart” demonstration.2,3
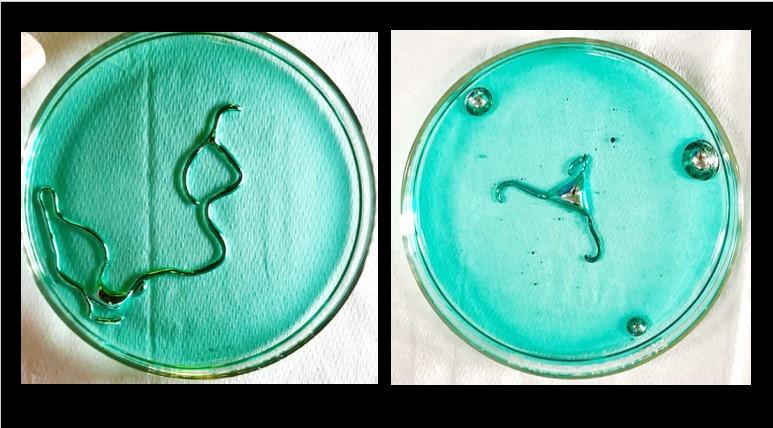
Well, Wang and coworkers are at it again with Galinstan. This time, they report on how liquid blobs of Galinstan can be coaxed to undergo some chemistry that is simultaneously bizarre and beautiful.4 Wait until you check this out (Video 1).
Video 1: Did I Make Venom Using Chemistry?! Tommy Technetium YouTube Channel, September 2, 2024.
Isn’t this some fascinating chemistry? It looks like it’s alive! I was blown away when I saw the octopus-like deformations that occur in the Galinstan droplet when I first got this experiment to work. The convulsing blobs reminded me of a couple of villains: Venom from Spiderman and T1000 in Terminator 2.
By the way, if you try this experiment on your own, I recommend first trying the experiment wherein crystals of copper (II) chloride are added to the Galinstan droplet immersed in a solution of acidified copper (II). This was easier for me to get to work than the experiment where the Galinstan spontaneously deformed when injected into acidified copper solution.
I’m thinking that the easiest way to connect this weird experiment to the chemistry curriculum is to discuss the electrochemistry occurring on the surface of the Galinstan. The authors state that copper nanoparticles are formed on the surface via the reduction of copper (II). Students could be challenged to predict which metals could potentially reduce the copper (II) ions, given the standard reduction potentials of copper (II) and the three metals in Galinstan:
Cu2+(aq) + 2e- → Cu(s) E0 = +0.34 V Equation 1
Ga3+(aq) + 3e- → Ga(l) E0 = -0.53 V Equation 2
In3+(aq) + 3e- → In(l) E0 = -0.34 V Equation 3
Sn2+(aq) + 2e- → Sn(l) E0 = -0.14 V Equation 4
Assuming standard conditions, coupling the oxidation of Ga(l) to the reduction of Cu2+(aq) results in the most thermodynamically favorable reaction:
3 Cu2+(aq) + 2 Ga(l) → 3 Cu(s) + 2 Ga3+(aq) E0 = +0.87 V Equation 5
3 Cu2+(aq) + 2 In(l) → 3 Cu(s) + 2 In3+(aq) E0 = +0.68 V Equation 6
Cu2+(aq) + Sn(l) → Cu(s) + Sn2+(aq) E0 = +0.48 V Equation 7
Nevertheless, the positive cell potential for all three reactions indicates that all three reactions (Equations 5-7) likely occur on the surface of the Galinstan.
I’m curious to hear what the spastic liquid protrusions seen in this experiment bring to your mind. Also, do you have any ideas for how this experiment can be connected to any topics in the chemistry curriculum? Let me know your thoughts in the comments. And as always, if you try this experiment out on your own, let me know about this!!
Happy Experimenting!
References:
- It should be noted that the exact ratio of these three metals in gallium-based liquid alloys of these three metals varies.
- Wang, B.; Jiang, X.; Zhang, Y.; Yu, L.; and Zhang, Y. Journal of Chemical Education 2022, 99 (2), 1095-1099.
- https://www.chemedx.org/blog/my-heart-beats-chemistry
- Wang, B.; Zhang, Y.; Wang, S.; Jiang, X.; Liu, L.; Zhao, D., Journal of Chemical Education 2024, 101. https://pubs.acs.org/doi/epdf/10.1021/acs.jchemed.4c00301