Solution to Chemical Mystery #19: Multi-colored Mixture

In Chemical Mystery #19,1 a yellow liquid is poured into a test tube, and the liquid turns red. When a white sold is added to the red liquid in the test tube, the liquid turns blue. When the test tube is stoppered and shaken, the blue liquid separates into two layers: blue on top and colorless on bottom.
This mystery makes use of acid-base indicators and the “salting-out” effect. In Video 1, you can see how this experiment is set up and carried out.2
Video 1: Colorful Chemistry Experiment, Tommy Technicium YouTube Channel, February 5, 2022.
Let’s look at this experiment in a bit more detail. Doing so might provide some insight into ways you might use this experiment in your classroom. It might also shed light on methods for engineering this experiment to give effects with different colors than those seen here.
The indicator used in this experiment is thymol blue (Figure 1), which is red at low pH, yellow at intermediate pH, and blue above pH 9.3 The color of thymol blue at different pH values explains the color changes observed. Thus, the indicator appears yellow at the beginning when it is in a mixture of alcohol and water, turns red when added to the test tube containing drops of acid, and finally changes to blue upon addition of copious amounts of K2CO3, a basic salt.
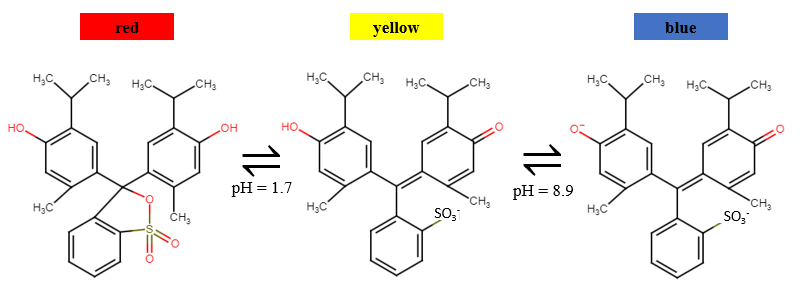
Figure 1: Thymol blue indicator red at low pH, blue at an intermediate pH, and blue at a pH over 9.
While addition of K2CO3 causes a shift in color to blue, it also separates the mixture into an alcohol-rich layer and a water-rich layer. The separation of the water and alcohol occurs because of an effect known as "salting out".4 Isopropyl alcohol dissolves in water due to hydrogen bonding between the O-H groups and O atoms on both water and alcohol molecules. When K2CO3 is added to a mixture of isopropyl alcohol and water, the salt dissociates into K+ and CO32- ions. These dissolved ions interact strongly with water. In fact, the K+-
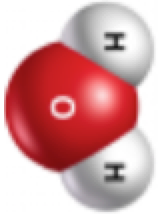
and CO32--
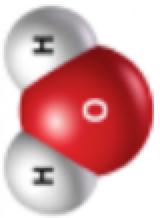
interactions that form are much stronger than the water-alcohol hydrogen bonding interactions. These ion-water interactions outcompete the hydrogen bonds between the water and the alcohol, causing the mixture to separate into two layers. Depending upon the properties of the indicator used, it will either end up dissolving in the alcohol layer or the carbonate-water layer. The thymol blue clearly dissolves in the upper alcohol layer. This probably occurs because thymol blue, with 27 carbon atoms total, has substantial non-polar character (Figure 1, structure on right). While the overall -2 charge of the blue form of thymol blue and its 5 oxygen atoms (which could serve as hydrogen bond acceptors) would favor dissolving in water, these factors don’t appear to be enough to coax thymol blue to dissolve in the layer of potassium carbonate and water.
As you can see at the end of the video (Video 1), you can toy with this experiment to give different colors. How do you think I pulled off the effect of causing the mixture to separate into blue and pink layers? I’d like to hear your thoughts about this in the comments. Also, I’d like to challenge you and your students to create some different colors to display in this experiment. Accepting this challenge might be a good way to get your students thinking about acid-base chemistry and intermolecular forces all at the same time...
Happy Experimenting!
References:
1. Kuntzleman, Tom, Chemical Mystery #19: Multi-colored Mixture, ChemEd X, January 31, 2022.
2. Tommy Technetium YouTube Channel, Colorful Chemistry Experiment, February 5, 2022.
3. Shakhashiri, B. Z. Chemical Demonstrations; University of Wisconsin Press: Madison, WI 1989; vol. 3, pp. 3-26.
4. Shakhashiri, B. Z. Chemical Demonstrations; University of Wisconsin Press: Madison, WI 1989; vol. 3, pp. 266-268.