Equilibrium Snowball Fight

The Next Generation Science Standards (NGSS) emphasize a new vision for science education; one that is focused on real scientific inquiry and skills, rather than rote memorization of facts. An essential component of the NGSS is developing and using models to represent concepts. Scientific models are tools to understand abstract knowledge and phenomena, and include diagrams, drawings, physical replicas, mathematical representations, analogies, and computer simulations.1
Dynamic equilibrium is a particularly challenging and fundamental concept in chemistry. Although students are generally able to solve numerical problems related to equilibrium, they often rely on memorized formulas and do not have a conceptual understanding of the topic.2 Consequently, misconceptions are common. This activity will address the following:
- No reaction occurs at equilibrium.
- The concentrations of the reactants are equal to the concentrations of the products at equilibrium.
- The rate of the forward reaction is greater than the reverse reaction at equilibrium.
- Equilibrium can only be approached in the forward direction.
Conceptual models have been beneficial in correcting misconceptions related to equilibrium.3 For dynamic equilibrium, I like to use a physical analogy that pits students against each other in a classroom-wide “snowball” fight.4 Not only is this activity great for building students’ conceptualization of dynamic equilibrium, but it is also really fun!
Overview
In this activity, student’s make “snowballs” out of sheets of scrap paper and engage in a “snowball” fight. The classroom is divided in half so that there is a product side and a reactant side. After a 5-second round, the number of “snowballs” on each side are counted and plotted on a graph. Although the data will look best with larger numbers of students and snowballs, the activity still works well with smaller classes. The data presented below was obtained with only 13 students. In any case, equilibrium is usually achieved after about six rounds.
Identify Misconceptions
Before we begin constructing the model, I use an “entrance card” (figure 1) to uncover the misconceptions that we are attempting to remedy.
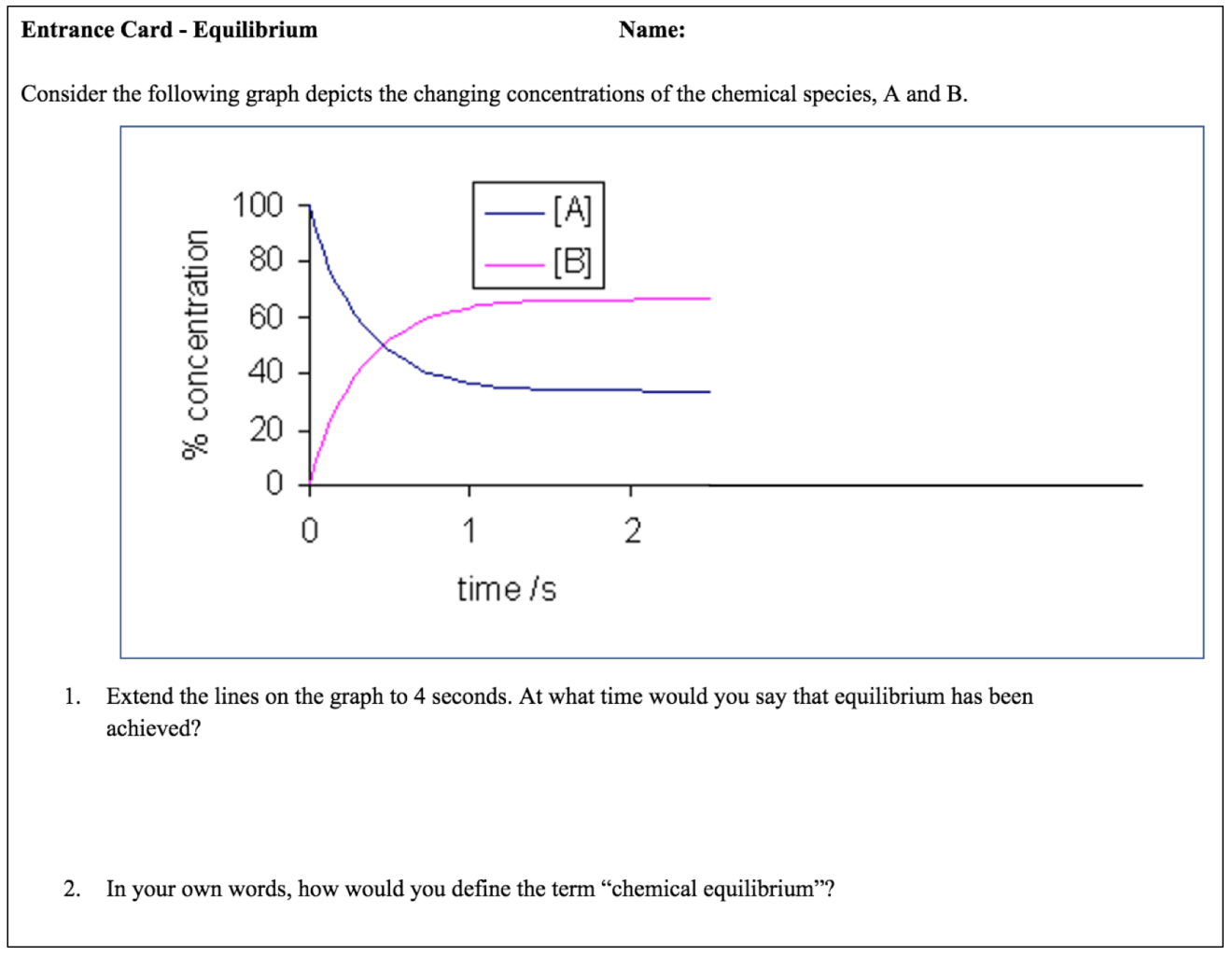
Figure 1: Entrance card
We grade the entrance cards as a class and do a quick poll to see what questions gave the most trouble. When asked to identify the point that equilibrium was achieved, I find that many students select the point at which the two lines crossed. Subsequently, those students defined equilibrium as “the point at which the concentrations of products and reactants are equal.”
Rules
Generally, students are very eager to throw “snowballs” at each other; thus, it’s important to set the ground rules and ensure active and complete student participation.
- The goal is to have the fewest snowballs on your side.
- You must try to hit an opposing player
- You can only have one snowball in your hand at a time
- You must drop all snowballs when the time is called (5 seconds per round)
I had each student make two “snowballs." Additionally, I recommend a loosely packed “snowball” (figure 2) because they won’t fly as far or hit other students with too much force.

Figure 2: Loosely packed "snowballs" work best
Match #1: Equal Driving Force
For the first match, students were evenly divided on the product and reactant side. All the snowballs were placed on the reactant side, and data were recorded for T = 0 s. I directed the student through six rounds, recording the number of “snowballs” on both sides after each round (figure 3).
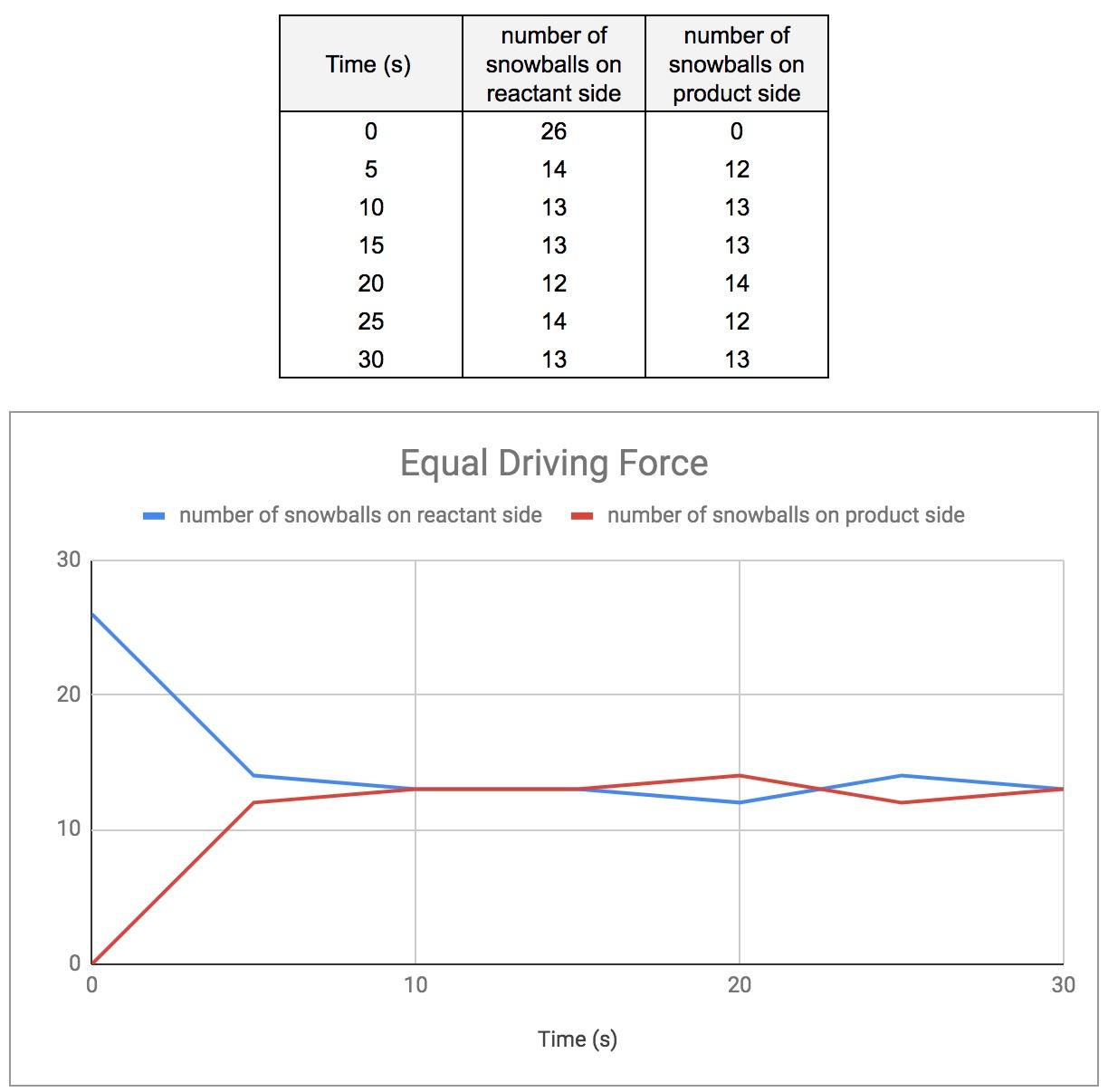
Figure 3: Equal Driving Force (13 students, 26 snowballs)
Match #2: Greater Reverse Driving Force
For the second match, I moved all except two students to the product side. Once again, all of the “snowballs” started on the reactant side. Since there were only two students on the reactant side, they were quickly overwhelmed with snowballs. Although, they still managed to get a few snowballs over to the product side in each round (figure 4).
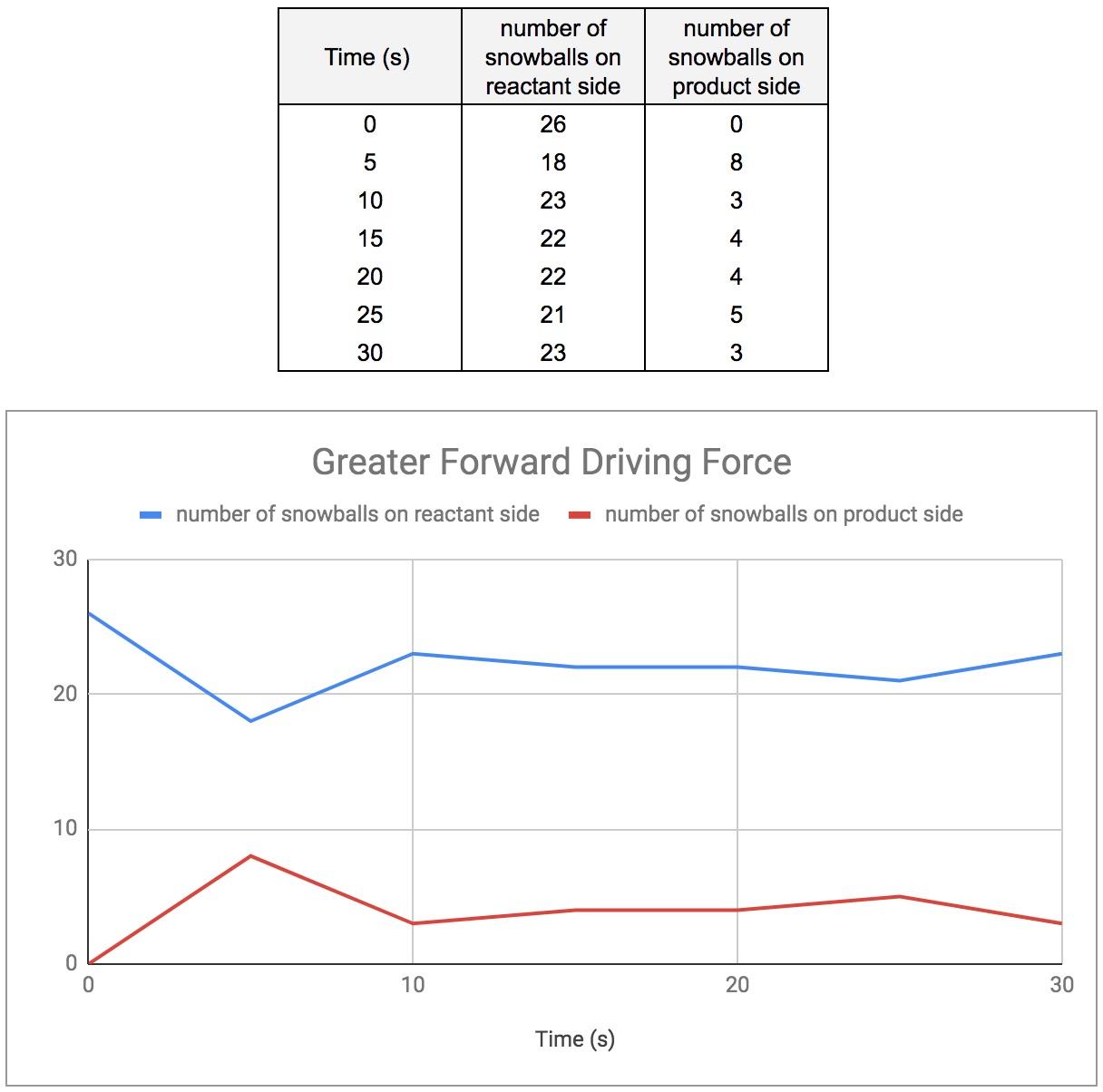
Figure 4: Greater Forward Driving Force (13 students, 26 snowballs)
Match #3: Greater Forward Driving Force
Finally, I had the students switch sides. This time, there were only two students on the product side, and they accomplished similar results as when they were on the reactant side (figure 5).
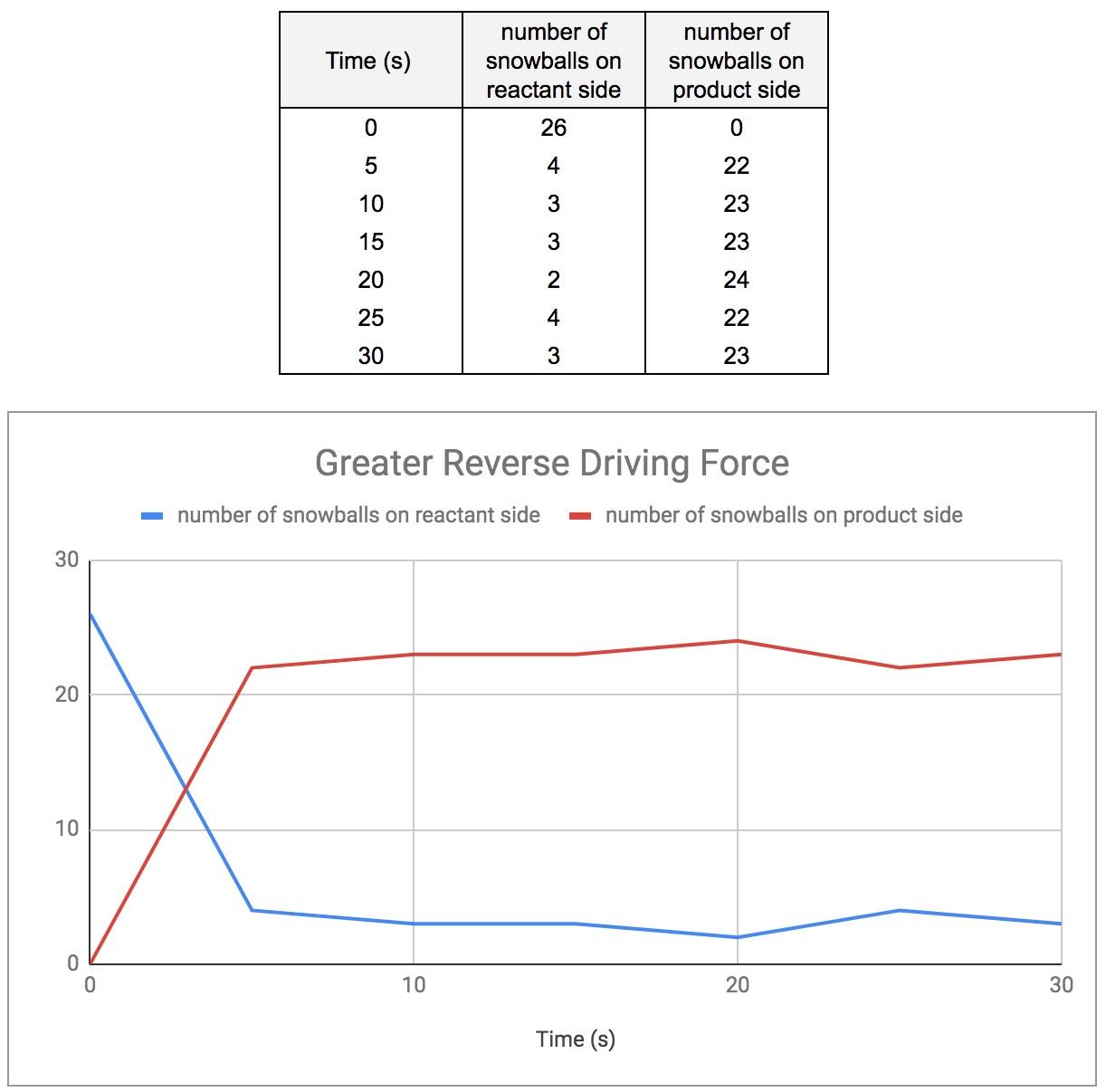
Figure 5: Greater reverse driving force (13 students, 26 snowballs)
Connecting to Chemical Equilibrium
The three matches represented three different equilibrium concentrations: equal product and reactant concentration (match #1), reactant favored (match #2), and product favored (match #3). The average equilibrium concentrations from each trial are used to introduce the concept of the equilibrium constant (Keq). The equilibrium constant is an expression that compares the concentration of products and reactants at equilibrium (equation 1).
Equation 1
After presenting this concept and expression to the students, I have them calculate the average concentrations and then the value of the equilibrium constant for each match (figure 6).
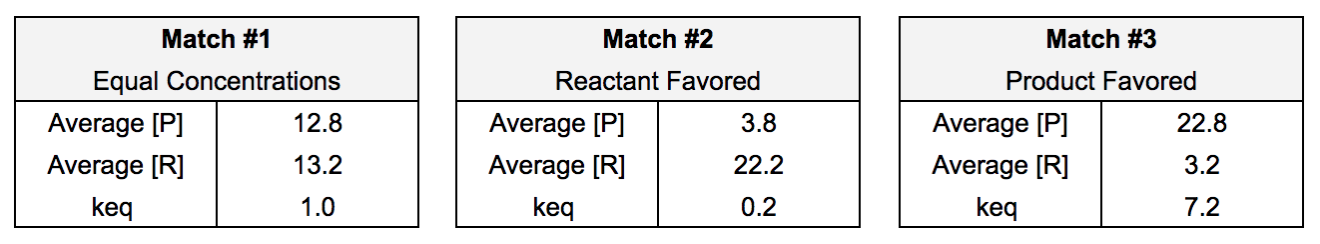
Figure 6: Keq for each match
With these results, we can clearly distinguish between different values of Keq.
If K = 1, then the concentrations of products and reactants are equal
If K > 1, then equilibrium favors the products
If K < 1, then equilibrium favors the reactants
Conclusion
Scientific models can be beneficial in correcting misconceptions related to foundational concepts in chemistry. Dynamic equilibrium is an especially important concept where misconceptions arise in students' understanding of the topic. This model is an excellent way to correct those misconceptions and develop students conceptual thinking about equilibrium.
Download the Power Point I use during this actiity.
References
- NGSS Lead States. 2013. Next Generation Science Standards: For States, By States. Washington, DC: The National Academies Press.
- Hackling, M. W., & Garnett, P. J. (1985). Misconceptions of chemical equilibrium. The European Journal of Science Education, 7(2), 205-214.
- Pekmez, E. S. (2010, December). Using analogies to prevent misconceptions about chemical equilibrium. In Asia-Pacific Forum on Science Learning & Teaching (Vol. 11, No. 2).
- Carson, S. R. (1999). An interactive pupil demonstration of the approach to dynamic equilibrium. Physics education, 34(1), 32.
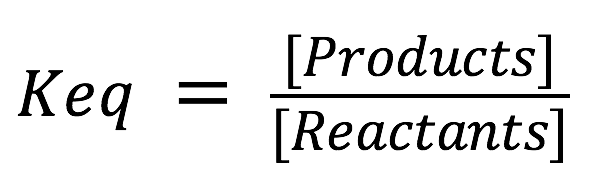
Comments
7Thanks for posting this!
Hey thanks for posting this! I was thinking of describing a snowball fight for equilibrium in class tomorrow and then found this. I really like your work on this. One question: The dip in the trend data in Figure 4 is reminiscent of an oscillation. I wonder if Tom Kuntzleman has some thoughts on that.
oscillations, quantum fluctuations, and the second law
This is indeed a fantastic activity! Those oscillations are a function of the low number of students in the class. I suppose they're kind of like "quantum fluctuations", which become visible at low numbers of particles. At low numbers of particles indeed you can see fluctuations away from equilibrium values - in violation of the second law!
I wish I could do this activity in my class of 230 students...the data would appear much smoother. And if we could do the experiment with a mole of students....well, I think you get the idea.
At least this is my thinking on the issue. Anyone have any other ideas?
Netlogo Chem Equilibrium Model
Try this - <a href="https://www.netlogoweb.org/launch#https://www.netlogoweb.org/assets/mod…">Chemical Equilibrium</a>
Great Activity
I will be doing this activity with my grade 12 IB class. Thanks for sharing.
Share Your Thoughts