The Chemistry of Outer Space

By Tom Kuntzleman, Mike Nydegger, and Mike Buratovich
During June of 2018, we held our 14th annual science camp at Spring Arbor University (SAU): Over 120 K – 8th grade students enjoyed an entire week of science experiments, demonstrations, and activities!
This year, the theme of our camp was “The Science of the Space” (Figure 1). During the week we performed several experiments that illustrate how chemistry is intimately tied to the science of the solar system, space travel, and outer space. In this post, we’ll describe several of these activities. Perhaps you might fit some of these ideas into your curriculum, or suggest additional activities not covered here that connect chemistry to the exploration of outer space.
Figure 1 - Logo for 2018 Cougar Science Camp (the cougar is SAU’s mascot)
OBJECTS in the SOLAR SYSTEM
Several of our activities at camp involved showcasing the chemistry of various objects in the solar system, some of which we describe below.
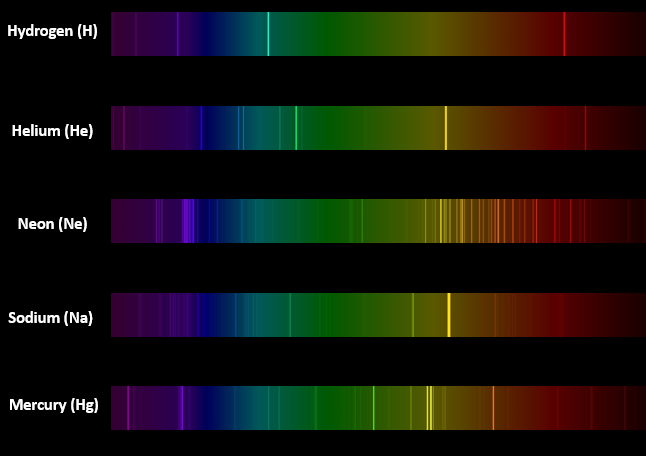
Figure 2 - Emission spectra of 5 elements
THE SUN
We stressed the idea that many aspects of the chemical composition of the Sun can be determined by studying the light emitted by the Sun. For example, the element helium was discovered to be on the Sun prior to it being discovered on Earth.1 To illustrate how this works, we had campers view a spectrum tube containing hydrogen through a pair of diffraction glasses.2 Next, campers were asked to compare the pattern of light observed through the diffraction glasses to the emission spectra of hydrogen, helium, neon, sodium, and mercury (Figure 2). The campers were then asked to predict what element was in the spectrum tube. Students easily recognized that hydrogen was in the tube, even though they did nothing other than observe the light emitted from the tube using diffraction glasses. It was emphasized to campers that no direct chemical analysis was needed to make this determination, and that the composition of the Sun can similarly be determined by analyzing its light. We continued the process by having students observe, in turn, spectrum tubes containing helium, neon, sodium, and mercury. After observing each tube using the diffraction glasses, students were asked to predict which element was contained in the tube. We extended the activity by conducting simple flame tests for sodium (emits yellow light), copper (emits green light), potassium (emits lavender light), and lithium (emits red light). We finished off this suite of demonstrations by having students predict that sodium was present in a pickle by running electricity through a pickle and observing the bright yellow emission characteristic of sodium (Figure 3).

Figure 3 - Running electric current through a pickle causes it to emit yellow light.
VENUS
This planet provided us with many connections to chemistry! First, the clouds on Venus are comprised of concentrated sulfuric acid droplets,3 and this fact has major implications for any space vehicles that humans might send to this planet. To allow students to observe the effect of acids on metal, we dropped pieces of zinc into a large test tube half-filled with 6 M HCl:
Zn(s) + 2 HCl(aq) --> ZnCl2(aq) + H2(g)
The reaction is vigorous enough that the hydrogen produced can be ignited using a nozzle-nose lighter, a fact that campers thoroughly enjoyed. To give students hands-on experience with the effect of acids on metals, we allowed them to drop pieces of magnesium metal into vinegar. The reaction between the acetic acid in the vinegar and magnesium dissolved the metal:
Mg(s) + 2 CH3COOH(aq) --> Mg(CH3COO)2(aq) + H2(g)
By adding a little bit of soap to the vinegar, small amounts of the hydrogen gas produced could be trapped and ignited with a lighter! See video below:
While carrying out these activities, we informed students that in some cases, space probes to Venus are being lined with Teflon to protect them from the acid droplets in the Venusian atmosphere.4
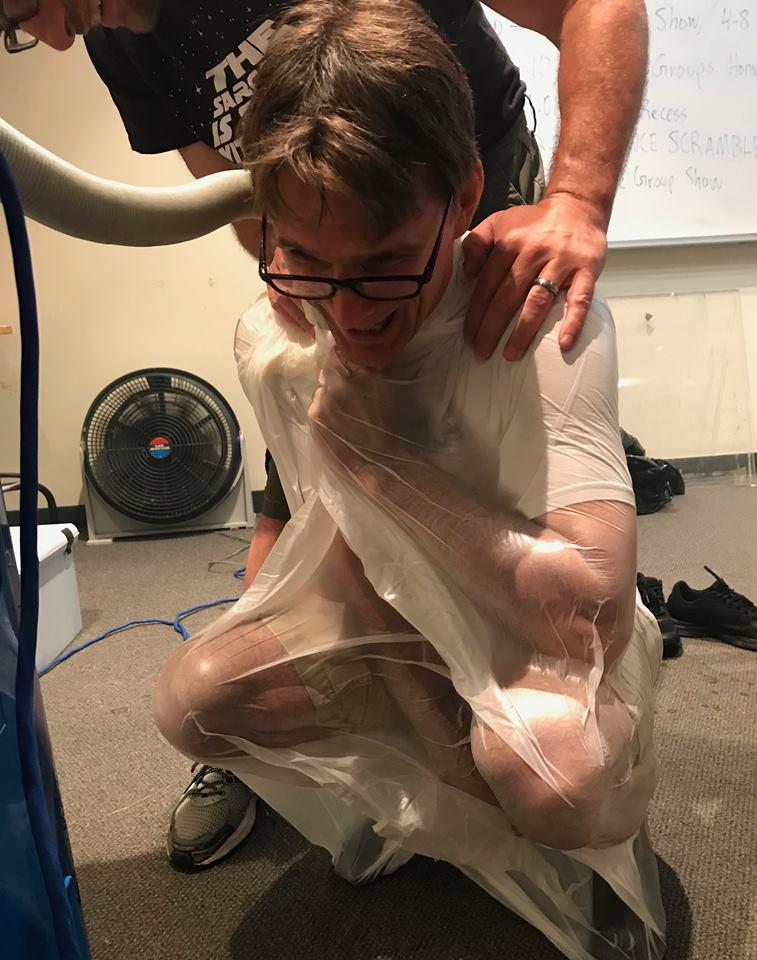
Second, the atmosphere of Venus is about 100 times as thick as the atmosphere on Earth! We therefore used the thick atmosphere of Venus as a springboard to discuss the concept of gas pressure. We found the can-crushing and egg-in-a-bottle demonstrations well-suited for this purpose. We also tried a fascinating experiment wherein a person is wrapped up to their neck and sealed tight in a large garbage bag, and the air is pumped out of the garbage bag using a vacuum cleaner.5 This experiment allows the person inside the garbage bag to feel the effects of air pressure. One of us (TK) tried this experiment (Figure 4) and was amazed at the force pressing down on his chest as the vacuum pumped the air from out of the bag! Use caution if you try this experiment: be certain that the operator of the vacuum cleaner knows to turn off the power if the person in the bag signals they have had enough.
Figure 4 - One of the authors feels the force of atmospheric pressure.
Finally, the average surface temperature of Venus is 735 K, the highest of all the planets. Its surface is hot enough to melt many (but not all) metals. For example, zinc (m.p. = 693 K), but not copper (m.p. = 1358 K) would melt on the surface of Venus. We heated samples of copper and zinc in the flame of a blow torch to show students what might happen to these metals on the surface of Venus.6 We used new U.S. pennies (minted after 1982) as our source of zinc and old U. S. pennies (minted prior to 1982) as our source of copper.7 We went on to point out to campers why Venus is hotter than Mercury, even though Venus is further from the Sun: Venus has an extremely thick atmosphere of 95% CO2, which is a potent greenhouse gas. On the other hand, Mercury has no atmosphere. We also attempted to impress upon campers the relationship between atmospheric CO2 and the average temperature on Earth, and the implications atmospheric CO2 has on global climate change.
EARTH
Earth is unique among the planets for several reasons, one of which is in that it contains copious amounts of water. Most of this water is in the world’s oceans, but some of it is suspended in the atmosphere as minute liquid droplets of water. Collections of these tiny water droplets are of course known as clouds. People always seem to enjoy observing the cloud that is produced when dry ice is placed in water, so we had campers do this to make their own clouds. Another favorite is the demonstration of the formation of a cloud by rapidly dumping hot water on top of several liters of liquid nitrogen in a large barrel. A huge cloud results, and campers had a blast running through the ensuing cloud that formed (Figure 5).

Figure 5 - A large cloud forms when hot water is dumped on liquid nitrogen.
MARS
Like Earth, the poles of Mars are capped with ice. Unlike Earth, the ice caps are Mars are made mostly of solid CO2. That’s right, the ice caps on Mars are made of dry ice! Because dry ice does not melt but rather undergoes sublimation, the polar ice caps sublime in the summer and enter the Martian atmosphere. In the winter, as much as one-third of the atmosphere on Mars deposits as solid CO2 on the poles again! We demonstrated this Martian CO2 cycle to campers we first filled gallon-sized Ziploc bags with CO2. To illustrate the deposition of solid CO2 during the winter, we immersed these Ziploc bags in liquid nitrogen. Upon doing so, crystals of dry ice formed on the sides of the Ziploc bag and the bag deflated. To show the effect of warmer temperatures during the summer, the baggie was removed from the liquid nitrogen and placed on a table at room temperature. When this was done, the crystals of dry ice sublimed, refilling the bag.
SATURN
Students were fascinated to learn that because the density of Saturn (0.69 g cm-3) is lower than the density of water (1.0 g cm-3), this planet would float on water! Of course you’d need a pretty big bathtub to pull off this trick. Nevertheless, we used floating and sinking activities that connect to the concept of density to help campers understand why Saturn would float on water. For example, 11 pound float, while 13 pound bowling balls sink in water (Figure 6). Several other floating and sinking activities that connect to the concept of density exist.8

Figure 6 - A camper discovers that not all bowling balls sink in water
URANUS and NEPTUNE
Experiments with liquid nitrogen are a natural fit with these planets, due to the extremely low temperatures (roughly 50 K) they display. For example, campers enjoyed hearing us claim “let’s see what would happen to a balloon on the surface of Neptune”, prior to dipping a balloon in liquid nitrogen.9 It’s certainly fun to put other objects, such as racquetballs or a Scrub Daddy10 sponge in liquid nitrogen. Campers were fascinated to see that the temperature on these planets are cold enough to liquefy air.11

Figure 7 - Balloons in liquid nitrogen is always a favorite demonstration
EARTH'S MOON
We placed items under vacuum to give students a fun way to visualize what might happen to various objects placed on the surface of the moon, which has no atmosphere. Campers enjoyed seeing room temperature water boil, and balloons or marshmallows expand when placed in a vacuum chamber.
TRITON
Triton, which is one of the moons of Neptune, has a surface made of solid nitrogen. Some of this solid sublimes into the atmosphere of Triton to form an atmosphere of nitrogen. To give campers an idea of what the surface of Triton might look like, we formed solid nitrogen by put liquid nitrogen under vacuum. The phase diagram of nitrogen12 can be used to show that given the conditions of pressure and temperature on the surface of Triton, it is not possible for liquid nitrogen to exist on the surface of Triton. See the video below for this experiment.
TITAN
One of the moons of Saturn, Titan, has hydrocarbon lakes on its surface. We therefore burned small amounts of hexane to familiarize campers with the flammable properties of hydrocarbons:
2 C6H14(l) + 19 O2(g) --> 12 CO2(g) + 14 H2O(g)
However, because the atmosphere of Titan is made of inert nitrogen gas, the hydrocarbon lakes on Titan do not burst into flame. To illustrate why this is the case, we poured a bit of nitrogen vapor out of a Dewar containing liquid nitrogen onto a small amount of burning hexane. Of course the hexane stopped burning, illustrating that the nitrogen atmosphere on Titan does not support combustion of its hydrocarbon lakes.
OTHER EXPERIMENTS
We now move on to describe a few activities we did that were not connected to objects in the solar system.
Rocket Fuels: A variety of chemical reactions can be used to illustrate how rockets are powered. Hydrogen has been used as a rocket fuel,13 so we exploded a few balloons filled with hydrogen to demonstrate the powerful reaction between hydrogen and oxygen:
2 H2(g) + O2(g) --> 2 H2O(g)
Campers enjoyed hearing they were making rocket fuel while they conducted the experiment wherein magnesium is placed in vinegar to generate hydrogen gas (see section on Venus, above).
Darth Vader and Photons: The Star Wars movies, which certainly include aspects of outer space, provided a backdrop for some activities. Darth Vader himself made a visit to camp and he asked us to help him determine which color of light would emit photons of highest energy. Vader was interested in this question, given that his light saber emits red light, while those of the Jedi Knights emit violet (Mace Windu), blue (Luke Skywalker), and green (Yoda). To help him figure this out, we shined violet, blue, green and red LED lights onto various glow toys (Figure 8, top left). The violet and blue photons clearly had enough energy to cause the toy to glow, as did the green photons to a lesser extent. The Dark Lord was not happy to learn that red photons contain the lowest energy of all the colors of visible light, and he ended up chasing us with his light saber! Fortunately, we were able to fight him off using light sabers that emitted photons of higher energy (Figure 8 top right and bottom).

Figure 8 - (Top left) Photons from a violet LED have sufficient energy to cause a glow square to glow. (Top right and bottom) Darth Vader learns the hard way that red photons don’t have as much energy as photons of other colors of visible light.
Space time Fabric: While this experiment is not directly connected to chemistry, both we and our campers greatly enjoyed this next activity. The bending of space time by massive objects can be demonstrated by placing heavy objects on a spandex sheet stretched tightly across a sturdy circular ring.14 To make our space time model, we made a ring by connecting three pool noodles with PVC pipe and duct tape. After assembling three rings in this manner, we used duct tape to connect the three rings on top of one another (Figure 9, top). Next, we tightly stretched spandex sheet across the ring (Figure 9, bottom). Students really enjoyed watching large steel spheres “attract” each other or smaller spheres and marbles into orbit by the bending the spandex space time fabric.

Figure 9 - (Top) Ring assembled using pool noodles, PVC pipe, and duct tape. (Bottom) Spandex sheet stretched across the ring and held tightly in place using bricks and books. The small sphere is in “orbit” around the “bend in space time” caused by the larger sphere.
CONCLUSION
We had a lot of fun designing and carrying out these experiments that connect chemistry to the science of outer space. Actually, we specifically chose this year’s camp theme to coincide with the 2018 theme for National Chemistry Week (NCW): Chemistry is Out of This World. Because we always participate in NCW, we thought it would be a good idea to connect this year’s science camp theme to the NCW theme. By doing so, we figured we’d learn quite a bit about how to connect simple science activities to the science of outer space. We plan on presenting many of the experiments presented herein during NCW this year (October 20–26). Perhaps you may find some of these ideas useful in your chemistry curriculum or as you begin to plan for NCW. Please do let us know if you have any ideas on how chemistry is related to the science of space. We would love to try some new experiments during NCW in October!
ACKNOWLEDGEMENTS
We wish to thank the many SAU staff, faculty, students, parents, grandparents, and high school students who worked at our science camp. We also thank the Hurst Foundation and the Bauervic Foundation for financial support.
REFERENCES (all accessed 7/1/2018)
1. https://pubs.acs.org/doi/pdf/10.1021/ed081p944
2. https://www.rainbowsymphony.com/diffraction-gratings/
3. https://pubs.acs.org/doi/pdf/10.1021/ed080p362
4. https://www.newscientist.com/article/dn16797-nasa-may-send-fleet-of-spa…
5.https://www.youtube.com/watch?v=q_X2PkcMHUU
6. Copper pennies do not melt in the flame, even though the temperature of the flame (> 2000 K) is higher than the melting point of copper. Perhaps this is due to copper being a good enough conductor of heat that it dissipates the energy from the flame to the air too quickly to gain enough energy from the flame to melt.
7. https://www.chemedx.org/blog/melting-pennies
8. See, for example, https://www.chemedx.org/blog/chemistry-bottle
9. https://www.chemedx.org/blog/chemical-mystery-4-case-misbehaving-balloon
10. https://www.chemedx.org/blog/temperature-experiments-scrub-daddy-sponge
11. https://www.chemedx.org/blog/collection-and-experiments-liquid-air
12. http://www.iaea.org/inis/collection/NCLCollectionStore/_Public/07/255/7255152.pdf
13. https://www.nasa.gov/topics/technology/hydrogen/hydrogen_fuel_of_choice.html
14. https://www.youtube.com/watch?v=MTY1Kje0yLg