Effect of Temperature on the Whoosh Bottle

I recently had the opportunity to attend a conference of the Associated Chemistry Teachers of Texas (ACT2). I had great time interacting with and learning from a whole bunch of wonderful chemical educators from the great state of Texas. One of the most interesting things I learned was in reference to the classic “whoosh bottle” experiment, which is powered by the combustion of isopropyl alcohol:
2 C3H7OH(l) + 9 O2(g) → 6 CO2(g) + 8 H2O(g) Equation 1
Interestingly, the reaction in this experiment yields different results when conducted at different temperatures (Video 1).
Video 1: Effect of temperature on a combustion reaction, pchemstud on TikTok. June 20, 2022.
That’s quite a significant difference! When I shared this video online, I had a few people comment that the difference observed might not be due to a faster reaction rate at the higher temperature. Instead, it was argued that the difference was because more alcohol vapor is contained in the warm bottle as compared to the cold one, on account of the higher vapor pressure of alcohol at higher temperatures. We can see this quantitatively by first noting the vapor pressures of isopropyl alcohol at 5.0oC (12 mmHg = 0.016 atm) and 32 oC and 20oC (32 mm Hg = 0.042 atm).1 Because the volume of the bottle is known (5 gallons = 19 L), we can then use the idea gas law (as n = PV/RT) to calculate the moles of isopropyl alcohol present in each bottle:
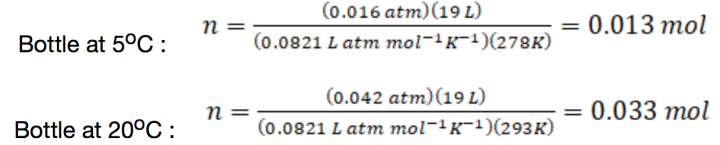
That’s about 2.5 times more alcohol vapor in the warm bottle over the cool one. Because reaction rates increase with higher concentration of reactants, perhaps the effect observed really is due to the presence of more fuel in the warm bottle.
Or is it…?
I decided to look into this a bit further, attempting to use the Arrhenius Equation to compare the kinetic rates of the reaction at the different temperatures:
Where k is the rate constant for the reaction, A is the pre-exponential factor, Ea is the activation energy for the reaction, T is temperature, and R = 8.314 J mol-1 K-1. The ratio of the values of the rate constants at 20oC (293 K) and 5oC (278 K) would therefore be:
Substitution of 125 kJ mol-1 as an estimate for the activation energy of the combustion of isopropanol2-4 into Equation 3 yields:
Therefore, this analysis suggests combustion in the warm bottle should proceed about 16 times faster than in the cold bottle.
So which is it?
Well, 16 (kinetic temperature effect) is over 5 times bigger than 2.5 (amount of fuel effect), so perhaps these analyses should lead me to conclude that combustion in the hot “whoosh bottle” goes faster because of the effect of temperature on reaction rates – not from the presence of more fuel in the hot bottle as compared to the cool one. But honestly, I’m not sure. I’m not entirely comfortable with my estimation of the activation energy of isopropanol combustion used in the Arrhenius analysis. I’m also not sure that the Arrhenius analysis I did is entirely appropriate. Furthermore, what I am confident about is that multiple types of combustion took place in the warm bottle but not in the cool bottle. I say this because I saw soot had formed on the outside of the warm bottle, but not the cool one after the reaction subsided (look for this carefully in Video 1). The formation of soot indicates incomplete combustion, in which carbon monoxide (Equation 4) and soot (Equation 5) are formed:
C3H7OH(l) + 3 O2(g) → 3 CO(g) + 4 H2O(g) Equation 4
2 C3H7OH(l) + 3 O2(g) → 6 C (s, soot) + 8 H2O(g) Equation 5
If multiple reactions occurred in the warm bottle but only complete combustion in the cool bottle, this complicates matters further. In the end, if I had to guess, I’d venture that both effects play a role.
But I’d like to know what you think. Why does the “whoosh bottle” go faster at higher temperatures? Is it simply because of the effect of temperature on reaction kinetics? Or is it because more alcohol vapor is present in the warmer bottle due to the higher vapor pressure of alcohol at increased temperature? Might both impact the reaction rate? What thoughts or criticisms do you have on my analysis presented here? Do you have any suggestions for experiments I might try to test which of these two possibilities better describes the observations? I look forward to hearing from you, and to carrying out more experiments on the effect of temperature on the “whoosh bottle.”
Happy Experimenting!
Acknowledgement:
Thanks to Dr. Bob Shelton from Texas A&M University in San Antonio for showing me this demonstration.
References:
- Parks, G. S.; Barton, B. J. Am. Chem. Soc. 1928, 50, 24-26.
- Vandenabeele, H.; Corbeels, R.; van Tiggelen, A. Combustion and Flame, 1960, 4, 253-260.
- Frassoldati, A.; Cuoci, A.; Faravelli, T.; Niemann, U.; Ranzi, E.; , Seiser, R.; Seshadri, K. Combustion and Flame, 1960, 4, 253-260.
- I could not find a literature value for experimentally measured values for the activation energy of the combustion of isopropanol. However, reference 3 does mention a value of less than 35 kcal mol-1 (146 kJ mol-1) based on a combination of measurements and theoretical calculation. Further, reference 2 cites 36.5 kcal mol-1 (153 kJ mol-1) as the activation energy for methane combustion. These considerations suggest 125 kJ mol-1 is a fair estimate.
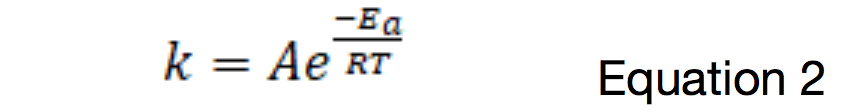
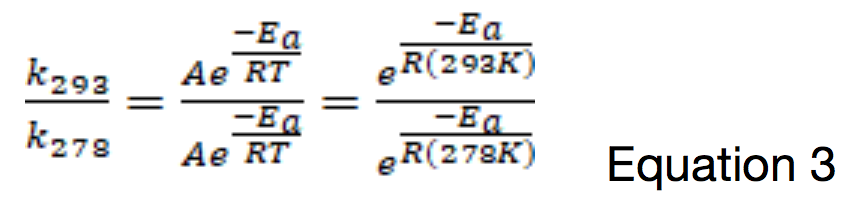
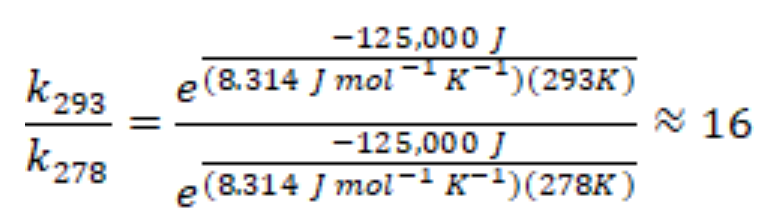
Comments
6On whether the concentration or temperature influences more.
I think that to verify this, it is enough to measure the time it takes to carry out each of the reactions, since it is very easy to calculate that time on video, even if it is approximate.
I have taken the trouble to measure this time, and in the first reaction at 5ºC it takes approximately 5 s to complete and in the second at 20ºC it takes approximately 2 s, then the first is 2.5 times slower than the second or vice versa the second is 2.5 times faster, curiously exactly the largest amount of alcohol there is, this is no coincidence.
If we remember the kinetics of the reactions, in the first place we have the concentration of reagents and secondly the Temperature, and studying it like this in that order is not by chance seeing what happened.
It can be assumed that it is at the same concentrations where the effect of temperature is relevant, but at different concentrations the temperature hardly has any effect on the kinetics of the reaction.
Saludos.
(Sobre si influye mas la concentración o la temperatura.
Creo que para comprobar esto es suficiente con medir el tiempo que tarda en realizarse cada una de las reacciones, ya que esta en video es muy fácil calcular ese tiempo, aunque sea aproximado.
M ehe tomado la moletia de medir dicho tiempo, y en la primera reaccion a 5ºC tarda aproximadamente 5 s en completarse y en la segunda a 20ºC tarda aproximadamente 2 s, luego la primera es 2,5 veces mas lenta que la segunda o al revés la segunda es 2,5 veces mas rápida, curiosamente exactamente la cantidad mayor de alcohol que hay, esto no es casualidad.
Si recordamos la cinética de las reacciones, en primer lugar tenemos la concetración de reactivos y en segundo lugar la Temperatura, y estudiarlo así en ese orden no es casualidad viendo lo ocurrido.
Se puede suponer que es a iguales concentraciones donde el efecto de la temperatura es relevante, pero a distintas concentraciones la temperatura apenas tiene efectos sobre la cinética de la reacción.)
In reply to On whether the concentration or temperature influences more. by Josefpm Perez
Nice analysis!
Hi Josefpm,
Thank you so much for taking the time to analyze how long it takes for each of these reactions to complete. What a great idea! It is indeed very interesting that the reaction happens 2.5 times more quickly at a warmer temperature than at a cooler temperature, and that this matches the estimated difference in concentration of the vapor in each container! In my opinion, this does not demonstrate that the reaction is only influenced by the differences in concentration, and that temperature has no effect on the kinetics. I would argue that the match in concentration and reaction time could indeed be coincidence. I say this because we certainly know that temperature influences the rates of reactions. Of course concentration does as well, as you rightly point out.
What do you think, Josepfpm? Am I off base? How might you provide further evidence to convince me that the difference in reaction kinetics is due only to concentration and not temperature?
I wonder what others think about this. Anyone else want to chime in?
Again, thanks so much for the discussion!
I agree but not completely ;-)
Oh, thanks for taking the time to answer, I agree with you, that does not mean that the temperature does not affect it, but I do believe, as I indicated, that at different concentrations the determining factor is the concentration and not the temperature, although the temperature has its effect, this effect is much less.
That is why I suppose that at the same concentration the effect of temperature or at very high temperatures would be much more decisive.
could it be coincidence? Yes, it could be, but wouldn't it be too much of a coincidence? and more when we know that the concentration in a factor that influences the speed of reaction.
It would be a matter of carrying out the experiment with other concentrations and measuring the speed and seeing if that relationship is maintained or, as he says, it was coincidence.
The question would not be to see if the temperature affects or not the speed of reaction, or if the concentration affects or not, it is evident that both affect; The question would be to what extent do they affect? which is more decisive?
It occurs to me that you can try carrying out the HCl+NaHCO3 reaction, using the same concentration at two temperatures and measuring the reaction time, it would be approximate, for example at room temperature and inside a refrigerator or cold room... umm I'll try to do it this summer if I have time...
--------
(Oh, gracias por tomarte tiempo en contestar, coincido con usted, eso no indica que la temperatura no afecte, pero si creo, como indico, que a diferentes concentraciones el factor determinante es la concentración y no la temperatura, aunque la temperatura tenga su efecto este efecto es mucho menor.
Por eso supongo que a igual concentración si sería mucho mas determinante el efecto de la temperatura o a muy altas temperaturas.
¿podría ser casualidad? Si, podría ser pero, ¿no sería demasiada casualidad? y más cuando sabemos que la concentración en un factor que influye en la velocidad de reacción.
Sería cuestión de realizar el experimento con otras concentraciones y medir la velocidad y ver si se mantiene ese relación o como dice fue casualidad.
La cuestión no sería ver si la temperatura afecta o no a la velocidad de reacción, o si la concentración afecta o no, es evidente que ambas afectan; la cuestión sería ¿en qué medida afectan? ¿cual es mas determinante?
Se me ocurre que se puede probar a realizar la reacción del HCl+NaHCO3, usando al misma concentración a dos temperaturas y medir el tiempo, de reacción, sería aproximado, por ejemplo a temperatura ambiente y dentro de un refrigerador o cámara frigorífica... umm intentaré hacerlo este verano si tengo tiempo...)
Share Your Thoughts