Effect of Temperature on Chemical Equilibrium using Copper Complexes

A popular method for demonstrating the effect of temperature on chemical equilibrium involves the use of Co2+ complexes. When Co2+ is dissolved in water it appears pink, but it forms a blue-colored complex in the presence of high concentrations of chloride ion:1-3
Co2+(aq) + 4 Cl-(aq) ßà CoCl42-(aq) Equation 1
The reaction that forms CoCl42- is endothermic. Therefore, the above reaction can be shifted to the right at high temperatures or to the left at low temperatures (Video 1).
Video 1: Cobalt equilibrium and thermodynamics, Tommy Technetium YouTube Channel (accessed 4/7/2021)
In this demonstration, concentrated HCl (12 M) is used as the source of Cl- to produce the blue colored CoCl42- species.1-3 Because concentrated HCl presents several hazards, I have instead begun using a system of Cu2+ complexes, which does not require the use of concentrated acid, to explore chemical equilibrium.4 This system of Cu2+ complexes has the added benefit of displaying a larger variety of colors: blue, green, yellow, and orange (Scheme I).
Scheme I: Putative compounds, reactions, and colors involved in the formation of various copper-chloro complexes in acetone. Each reaction is endothermic from left to right.
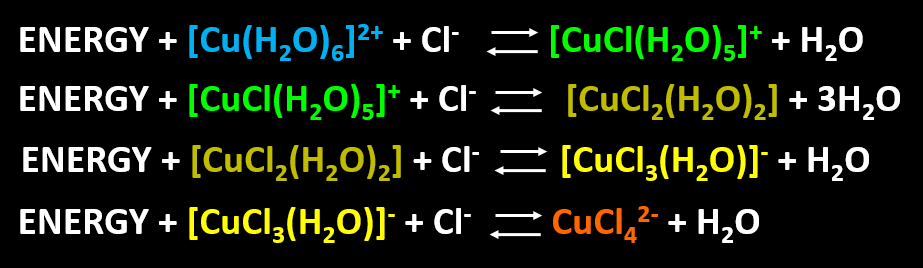
Note that each reaction listed in Scheme I is endothermic from left to right. Because of this, each reaction shifts to the right at increasing temperatures, but to the left at decreasing temperatures. Indeed, I have noted that solutions of CuCl2 in acetone are dirty-yellow at room temperature, green when incubated in a dry-ice acetone mixture, and orange-yellow when incubated in hot water (Video 2).
Video 2: Chemical Equilibrium: Colorful Demonstrations, Tommy Technetium YouTube Channel (accessed 4/7/2021)
Using this system of Cu2+ complexes, I have only been able to generate colors that range from green to yellow by varying temperature. However, you will also note in the video above that addition of salt to this system causes a shift to an orange color, and addition of silver nitrate causes a shift to a blue color. The presence of high Cl- concentration causes each reaction to shift to the right, ultimately forming CuCl42- (Scheme I). On the other hand, addition of silver nitrate causes the removal of Cl- ion because silver ions precipitate out AgCl (s):
Ag+ + Cl- à AgCl(s) Equation 1
Because removal of Cl- ion has the opposite effect of Cl- addition, addition of silver nitrate shifts each equilibrium listed in Scheme I to the left, ultimately resulting in a color change to blue.
Because these experiments avoid the use of concentrated HCl, they are amenable to having students perform these copper-based equilibrium experiments as part of a laboratory-based exercise. I have indeed had my students use some of these experiments as part of a laboratory-based exercise, and on other occasions I have used these experiments as in-class demonstrations.
Do you have any ideas for how to extend or modify the experiments presented here? If so, I’d love to hear any suggestions you might have.
Happy experimenting!
References
1. Shakhashiri, B. Z. (1989). Chemical Demonstrations: a Handbook for Teachers of Chemistry Volume 1. Madison, WI: The University of Wisconsin Press.
2. Grant, A. W. Cobalt complexes and Le Chatelier, J. Chem. Educ. 1984, 61, 466.
3. DeGrand, M. J.; Abrams, M. L.; Jenkins, J. L.; Welch, L. E. Gibbs Energy Changes during Cobalt Complexation: A Thermodynamics Experiment for the General Chemistry Laboratory. J.
Chem. Educ. 2011, 88, 634−636.
4. https://www.chemedx.org/blog/multi-colored-equilibrium-experiment
Comments
5Solvent choice
These are very interesting experiments. I want to know what is the purity of the acetone used in your experiment. Could acetone from home improvement stores be able to produce the range of colors? Have you tried with ethanol, isopropanol or other non-aqueous solvents? Would those solvents work? In a high school setting (limited budget), those solvents are cheaper to purchase from other sources than Fisher or Flinn.
Thank you again for sharing.
Yu-Sung
Hi Yu-Sung, thank you for your question and comment. I used lab grade acetone purchased from Fisher Scientific for these experiments. Having said this, I am almost certain that acetone purchased from Home Depot, etc. would work. I have used other non-aqueous solvents (isopropyl alcohol, for example), and the color response in other solvents is different. Beware that even a tiny bit of water can drastically shift these equilibria. Please let me know how your experiments work for you when you try them!
Share Your Thoughts