A Multi-Colored Equilibrium Experiment

I’d like to describe a very colorful system you can use to explore many facets of chemical equilibrium. The experiment is extremely easy to prepare, and avoids the use of concentrated acid that is used in many equilibrium experiments.1-3 To prepare the experiment, simply mix about 0.3 grams of anhydrous copper (II) chloride into 100 mL of acetone, and swirl until a dark yellow-green solution has formed. It’s okay if all of the copper (II) chloride doesn’t dissolve. The resulting solution is able to produce a variety of copper complexes that display various shades of blue, green, yellow or orange depending upon the conditions (Scheme I).4-13
Scheme I: Putative compounds, reactions, and colors involved in the formation of various copper-chloro complexes in acetone.

Putative compounds, reactions, and colors involved in the formation of various copper-chloro complexes in acetone.
In video 1 below you can view a variety of possible experiments:
Video 1: Colorful Copper Equilibrium from Tommy Technetium's YouTube Channel, March 5, 2020 (accessed 3/6/2020).
Discussion
Copper (II) ion reacts with chloride ion to form several chloro-containing complexes including [Cu(H2O)5Cl]+, [CuCl2(H2O)2], [CuCl3(H2O)]-, and [CuCl4]2-.4-13 These complexes, which tend to form best in non-aqueous solvents,6,8-10,13 form an array of vivid colors that span from blue to orange.12 A complete description of the relationship between the various colors and structures is not fully understood. Further, the colors and structures involved seem to vary greatly with conditions and solvent. Nevertheless, the reactions, compounds, and colors proposed in Scheme I are fairly consistent with reports in the literature4-13 and also the experiments seen in video 1.
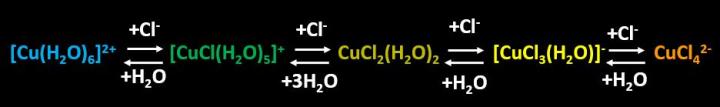
Figure 1: Abbreviated form of equations displayed in Scheme I.
Figure 1: Abbreviated form of equations displayed in Scheme I.
Addition of chloride ion (by addition of NaCl from table salt) tends to shift the equations above to the right (Figure 1), causing color changes from blue to green, green to yellow-green, yellow-green to yellow, and yellow to orange. Such results are entirely consistent with Le Châtelier's Principle, which insures that the addition of a compound to one side of a system at equilibrium will cause that system to shift to the other side.
Addition of water shifted each equation to the left (Figure 1). Generally speaking, changes in water concentration do not affect equilibria in aqueous solution in which water is the solvent. Under such conditions the amount of water is so large that changes in water concentration display little effect. However, acetone is the solvent in the experiments shown in the video above. Because of this, additions of water strongly increase the concentration of water in the system, shifting all equations to the left.
In a future post I will share how addition of AgNO3 (for chloride removal) and temperature affect this system of non-aqueous copper (II) chloro complexes. Please do leave a comment if you have any other suggestions for further experimentation, or any other comments on this work. Happy experimenting!
References
1. Shakhashiri, B. Z. (1989). Chemical Demonstrations: a Handbook for Teachers of Chemistry Volume 1. Madison, WI: The University of Wisconsin Press.
2. Grant, A. W. Cobalt complexes and Le Chatelier, J. Chem. Educ. 1984, 61, 466.
3. DeGrand, M. J.; Abrams, M. L.; Jenkins, J. L.; Welch, L. E. Gibbs Energy Changes during Cobalt Complexation: A Thermodynamics Experiment for the General Chemistry Laboratory. J. Chem. Educ. 2011, 88, 634−636.
4. Helmholz, L.; Kruh, R. F. The Crystal Structure of Cesium Chlorocuprate, Cs2CuCl4, and the Spectrum of the Chlorocuprate Ion J. Am. Chem. Soc.1952, 74, 1176-1181
5. Khan, M. A.; Schwing-Weill, M. J. Stability and Electronic Spectra of the Copper (II) Chloro Complexes in Aqueous Solutions Inorg. Chem. 1976, 15, 2202-2205.
6. Elleb, M.; Meullemeestre, J.; Schwing-Weill, M. J.; Vierling, F. Stability, Electronic Spectra, and Structure of the Copper (II) Chloride Complexes in N, N-Dimethylformamide Inorg. Chem. 1980, 19, 2699-2704.
7. Ramette, R. W.; Fan, G. Copper (II) Chloride Complex Equilibrium Constants Inorg. Chem. 1983, 22, 3323-3326.
8. Khan, M. A.; Meullemeestre, J.; Schwing-Weill, M. J.; Vierling, F. Detailed Spectrophotometric Study of Copper (II) Halides in Anhydrous Methanol Inorg. Chem. 1989, 28, 3306-3309.
9. Benghanem, S. D.; Khan, M. A.; Meullemeestre, J.; Vierling, F. Halogenocomplexes of Copper (II) in Anhydrous Propan-2-ol Polyhedron, 1991, 10, 2529-2533.
10. Katzin, L. I. Ionization Differences Between Coordination Statsof a Cation. Octahedral-Tetrahedral Equilibrium of Transition-Element Chlorides in Dimethylformamide, J. Chem. Phys. 1962, 36, 3034-3041.
11. Yi, H.-B.; Xia, F.-F.; Zhou, Q.; Zeng, D. [CuCl3]- and [CuCl4]2- Hydrates in Concentrated Aqueous Solution: A Density Functional Theory and ab Initio Study J. Phys. Chem. 2011, 115, 4416-4426.
12. De Vreese, P.; Brooks, N. R.; Van Hecke, K.; Van Meervelt, L.; Matthijs, E. Binnemans, K.; Van Deun, R. Speciation of Copper (II) Complexes in an Ionic Liquid Based on Choline Chloride and in Choline Chloride/Water Mixtures Inorg. Chem. 2012, 51, 4972-4981.
13. Ohtaki, H. Structural studies on solvation and complexation of metal ions in nonaqeous solution Pure & Appl. Chem. 1987, 59, 1143-1150.