The Chemistry of Weight Loss

If you want to lose weight, you have to burn calories. Anyone who has gone on a diet knows this. But when someone loses weight, have you ever wondered where the lost mass goes?
As chemists, we know that the law of conservation of mass assures that “weight loss” cannot cause matter to vanish into oblivion. Therefore, what is going on chemically when someone loses weight?
Two researchers in Australia answered this question in the following way. They posited that the process of weight loss can be understood through the following chemical equation, which represents the oxidation of an average fat molecule (C55H104O6):
C55H104O6 + 78 O2 à 55 CO2 + 52 H2O Equation 1
The main idea is that fat in the body gets converted to carbon dioxide and water, both of which leave the body. It’s that simple. We lose weight by breathing out CO2 and also by excreting water (primarily though exhalation, urination, and sweating). As you will see below, 84% of the mass exits the body as CO2. Isn’t that remarkable? Weight loss is primarily achieved by, well, breathing.
We can look at Equation 1 in detail to gain more insight into how the body loses mass. Note that the weight lost by the body comes from the fat molecule only. While O2 is a necessary reactant, this component is supplied by the atmosphere. Note that when a molecule of fat is consumed, all 55 carbon atoms in a fat molecule ends up in CO2, which we exhale out of the body. All of the 104 hydrogen atoms in a molecule of fat ends up in H2O, which is lost through sweating, urination, and exhalation. Notice that each molecule of fat also contains 6 oxygen atoms. Where do these 6 atoms go? Well, isotope labelling studies have shown that 4 of these oxygen atoms end up in CO2, while the remaining two end up in H2O.
Armed with this information and molar masses from the periodic table, we can do an accounting of where the mass in the fat ends up. One mole of fat weighs 860 grams (55 x 12) + (104 x 1) + (6 x 16) = 860. Recall from the previous paragraph that 55 atoms of carbon and 4 atoms of oxygen end up in CO2 from each molecule of fat. On a per mole basis, this means 724 grams of each mole of fat ends up in CO2 (55 x 12) + (4 x 16) = 724. We also see that 104 atoms of hydrogen and 2 atoms of oxygen end up in H2O from each molecule of fat. This works out to 136 grams of H2O for every mole of fat (104 x 1) + (2 x 16) = 136.
I decided to do a chemistry experiment on myself to see how this all fit together. I first devised a simple method to measure my rate of exhalation. To do this, I filled a large glass flask (volume = 2.1 L) with water to the brim, and inverted it in a pan of water. I placed a rubber tube up the neck of the inverted bottle. While standing, I timed how long it took for me to displace the volume of water in the inverted bottle by breathing into the rubber tube. I didn’t try to rush this – I tried to exhale as normally as possible. In three trials I found that it took me an average of 17 seconds to exhale a volume equal to 2.1 L. During these experiments, the temperature was 24oC and the air pressure was 738 mm Hg in the lab. You can see me carrying out one of these runs in the video below.
The video shows a simple method for calculating the rate of exhalation for the subject.
Next, I used the ideal gas law to determine the moles of gas I exhaled out in 17 seconds:
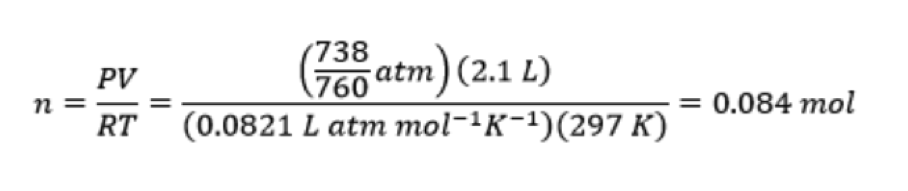
This works out to 0.0049 moles every second, or 17.7 moles of gas every hour. However, exhaled breath is not pure CO2: only about 4% of exhaled breath is CO2. Thus, I was breathing out CO2 at the rate of 0.71 moles (31 g) of CO2 every hour (about 9 mg of CO2 per second).
Using Equation 1, we see that 0.71 moles of CO2 per hour converts to 11.1 grams (0.024 pounds) of fat consumed every hour while standing. Dieticians tell us that 3500 Calories corresponds to 1 pound of fat. Therefore, I can report the results of my experiment in a more orthodox manner: I burned roughly 85 Calories per hour while standing during my experiment.
This exercise included an application of several chemical ideas (ideal gas law, stoichiometry, nutrition, and simple kinetics). Given this, and the fact that the Australian authors implored chemistry teachers to cover the chemical concepts involved in losing weight, I’m toying with the idea of converting this small investigation into a laboratory experiment for my students. Thus, I would greatly appreciate suggestions to improve my experimental protocol (which you can see in the video above). If any of you try this experiment out (either or your own or with your students) let me know how it works out for you.
Happy experimenting!
Reference
Ruben Meerman and Andrew J. Brown When somebody loses weight, where does the fat go?
Comments
5Mass v. energy conservation in analysis of weight loss
Great idea applying mass conservation and stoichiometry to an analysis of weight loss. Most analysis of this kind that I have seen use energy conservation instead of mass conservation, which leads one to lose any hope of ever losing weight with exercise.
John Hill in the 1980s wrote a couple of articles in the Journal of Chemical Education that applies chemistry principles to weight-loss and diets using energy conservation.
BTW, love the title. Using 'weight loss' in the title should bring a whole new category of people to ChemEd X, if the chemistry does not scare them away. ?
In reply to Mass v. energy conservation in analysis of weight loss by Jon Holmes
Thank you for pointing me to these articles, Jon. I'll be sure to check them out. I agree that the orthodox way of viewing weight loss via calorie consumption adds an unnecessary layer of complication. The article by Meerman and Brown allow us to clearly see that weight loss comes as we expel molecules, namely CO2 and H2O. For years I had suspected that weight loss occured by breathing out CO2, and had even mentioned this in my classes several times. I greatly appreciate the work of Meerman and Brown, as it gives a solid, quantitative, and chemical way to approach weight loss.
Share Your Thoughts