Unit Conversions: The good, the bad and the ugly
Like most chemistry teachers, one of the first things I go over in the beginning of the year is unit conversions. Students come into my class with all sorts of prior knowledge concerning unit conversions; some good, some bad and some downright ugly.
Every year I teach unit conversions I make some changes and I see progress. This year has been the most successful year yet with this topic. I write this post not as someone who has it all figured out but as someone who has seen the fruits of taking chances and making changes with my pedagogy. This post is a compilation of my observations of the prior knowledge students come in with and how I have dealt with them.
First, the good. I use Modeling Instruction pedagogy in my classroom so we do a lot of graphing and discussion of relationships between variables from day one. The good thing is that students come in knowing is how to deal with relationships. A student may give you a blank stare when asked to convert 6.00 meters to centimeters but if you tell that student that for every 1 meter, there are 100 centimeters, chances are that student will be able to answer the original question. Their brains are already wired to think proportionally.
In the beginning of the year, I give each group in my class a piece of cardstock that has been divided into 10 equal sections. Each group has a different length piece of cardstock. Students must measure the length, width and height of their desks with their new “ruler.” I even let them, make up the name for the units on their ruler. That gives us an opportunity to talk about precision and significant figures. Later, I give students the measurements of the length, width and height of their desks in my unit, the “stewart.” I also give students the width of my demo desk in stewarts. It is each group’s task to figure out the width of my demo desk in their units. When I went around to talk to each group this year, many groups explained that they had found the relationship between stewarts and their unit to solve the problem. Groups either set up a proportion between the units or used to the relationship they calculated to scale the measurements they already had. Either way, students were automatically thinking proportionally.
Now the bad. When I say bad, I mean something that is not inherently a bad practice but is bad when taught algorithmically. In this case, I am talking about dimensional analysis. In the beginning of my teaching career, I let students choose whether they wanted to use dimensional analysis or proportional reasoning. I knew student choice was valuable and I wanted students to be able to do what made sense to them. Good idea, not so great in practice. This ended up in a lot of what I call math monsters. A math monster occurs when a student tries to combine proportional reasoning AND dimensional analysis into one equation that makes no sense because there is either no variable to solve for or the other side of the equation is missing so they cannot solve for the variable. I realized that students were creating these math monsters because they were trying to repeat a set of steps they did not understand. Instead of providing students with multiple avenues to solve a problem, I had provided them with multiple algorithms to memorize and confuse.
Finally, the ugly. The ugly includes crutches that I have increasingly seen students use that have hindered their abilities to actually understand unit conversions. I will be the first to admit that I have been both a victim and perpetuator of some of these crutches.
The first "ugly" thing I have seen students do is decimal hopping to convert between metric units. If you don’t know what I’m talking about, it looks like this:
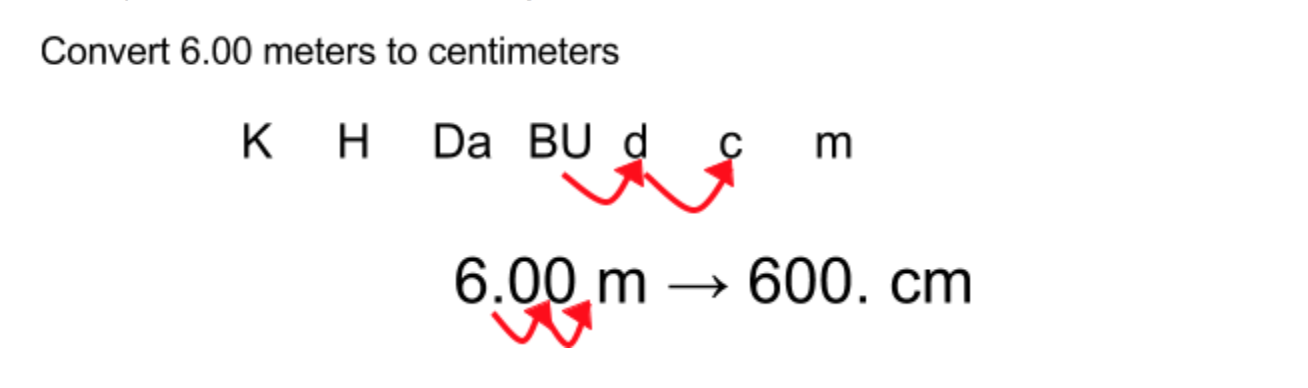
You and I understand that for every 1 meter, there are 100 centimeters, so moving the decimal over 2 spaces is essentially multiplying by 100 cm/m. Students do not see that. They see a shortcut that gets them to right answer without understanding the process.
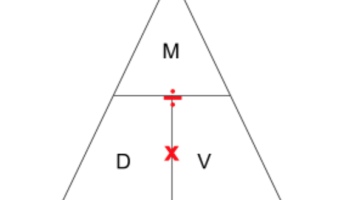
The other ugly thing I have seen is magic triangles, specifically in regards to density (though they can be used for any three-variable equation). If you don’t know what I am talking about, it looks like this:
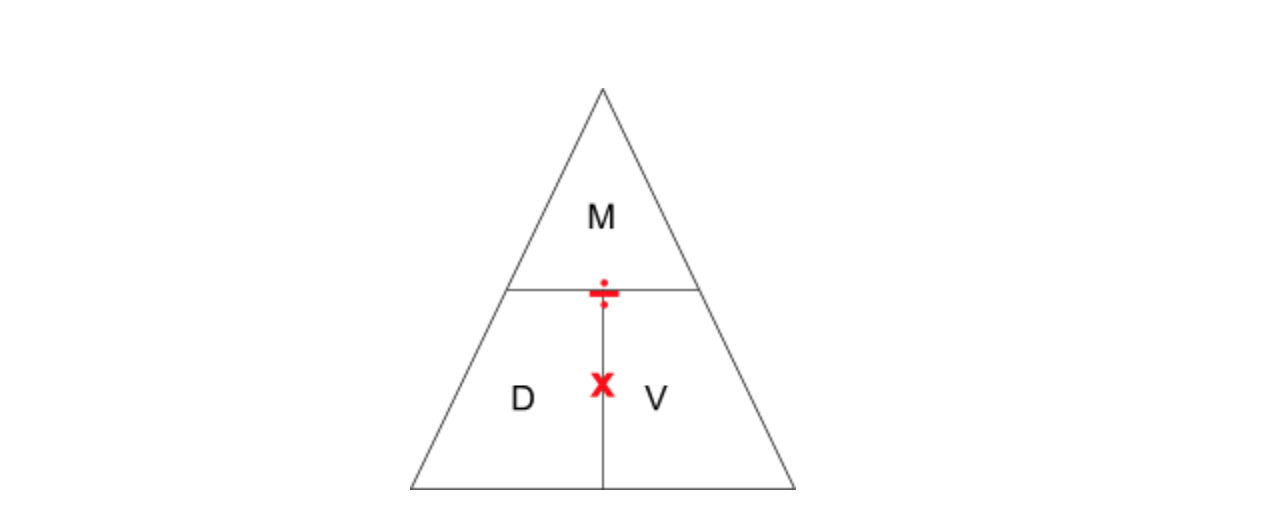
The idea behind the density triangle is you cover up the variable you want to find and then the triangle tells you what operator to use between the other two variables (multiply if the variables are next to each other, divide if the variables are stacked). Again, students see this as a shortcut that leads them to the right answer. The process or any conceptual understanding of the topic does not matter.
What do I do? After collecting these observations for the past few years, I made a radical decision this year that I have been contemplating for awhile: teach only proportional reasoning to solve unit conversions. The problem is, even proportions can be taught algorithmically (see most math classes). To deepen student understanding of what a proportion is, I have outlawed the word “per” in my class. Students often use the word “per” but have no idea what it means or implies. Instead, I have my students talk about conversion factors in terms of the relationship between the two units; for every 1 (insert unit here), there are x (insert unit here).
Since I use Modeling Instruction, my students do a lot of whiteboarding. When a group presents a unit conversion, the first questions I ask are “what were you given?”, “what unit are you trying to convert to”, “what is the relationship between those two units?” Students then set up a proportion to show that the relationship between the two units does not change.
Anecdotally, I have noticed my students talking about unit conversions differently. They talk about units in terms of relationships without me prompting them. Their work is neater and they can actually explain it instead of saying things like “I don’t really know what I did, I just multiplied the numbers and got the right answer.” As a scientist, I know that the plural of anecdote is not data. While it is early in the year and I have not had a lot of chances to collect data, I can tell you that on their first quiz, over 90% of my students demonstrated mastery of unit conversions (I use standards-based grading). I still have work to do with the way I teach unit conversions, but the switch to proportional reasoning is the biggest step I have made in the right direction in years.
Comments
11Dimensional analysis
Do you teach dimensional analysis in honors chemistry or ap chemsitry level? I am nervous about giving it up even though it didn't go well at all last year (1st year chem teaching) - I have used it so much is physics and my ap chem kids loved using it last year. They definitely were using it algorithmically but the arugument from them is that on an exam in college and on the ap exam - getting the right answer makes a difference. I am sure ap would still give them credit for proportions, but I would think it would take longer. Thoughts, if you get a moment, please.
In reply to Dimensional analysis by Penny Shrum
Great question! In honors, I
Great question! In honors, I usually let my students choose what method they prefer. In instances where this backfires and the student creates a dimensional analysis/proportional reasoning math monster, I have them use proportional reasoning until they understand what math they are actually doing. When given the choice, most of my students still choose proportional reasoning because it makes more sense to them.
Teaching Dimensional Analysis
For those interested in lessons that help in teaching dimensional analysis, I have posted two resources for free student and instructor use. The first teaches DA as homework tutorials, and a second consists of mini-lessons that can be taught in class and include “chem-specific” strategies.
The homework lessons have been independently evaluated by both instructors and students. In a recent paper discussed in an ACS online conference, a college instructor reported using an extended version of the homework tutorials in General Chemistry. His students’ scores on the ACS Exam rose -- from an average 42nd percentile in the 3 years before -- to the 60th percentile in the year of the “tutorial homework.” (The median is the 50th percentile).
But the lessons have been tested and work well in high school “college preparatory” Chem I classes as well. One of the problems I have regularly assigned in Chem I is:
"If a drop of water contains 0.050 mL and one million molecules evaporate per second, how many hours would it take for exactly 3 drops to evaporate?"
After they have done both the homework and in-class mini-lessons, students routinely solve this and similar problem easily. That helps with preparation for AP/General Chemistry.
I’ve posted an explanation of using “chem-specific” conversions to teach the “3 drops” problem at www.ChemReview.Net/PrepSteps.PDF (click then check your PDF downloads).
Access to the free homework tutorials is at: www.ChemReview.Net/APFallReview.html .
On the question of “dimensional analysis versus proportional reasoning,” I don’t see a conflict. A conversion factor is simply a proportion to multiply a given quantity by. I certainly see the value in teaching concepts using simple whole number proportions with math students can do automatically “in their heads.” For problems with more complex numbers, however, including most stoichiometry, if we are going to get students ready for college chemistry (and physics) textbooks and lecture, I can’t see a way around starting the teaching of rigorous dimensional analysis in first-year high school chemistry.
If they enter AP or college gen chem without a strong background in dimensional analysis, survival will be difficult at best.
-- Eric (rick) Nelson
In reply to Teaching Dimensional Analysis by Eric Nelson
Hi Eric!
Thanks for sharing resources, I am excited to take a look at them! I really love the idea of chem specific problems. I think sometimes when we try to use things we think students are familiar with, we tend to over complicate it. It is great to start off with simpler proportions of every day relationships but questions like the one you posed do a great job of helping students understand scale in chemistry.
I think we agree that the most important thing is for students to understand the math they are doing. For me, proportional reasoning has been great for that. With my honors students, I let them choose what they want to use. Many of them start with proportional reasoning and then find themselves moving to dimensional analysis once they have the concept of a proportion down. Some of them stick with proportional reasoning. In both cases, I have seen students succeed in solving complex stoichiometry problems. Either way, students are thinking critically, not just plugging and chugging, which is the ultimate goal!
The Necessity to Plug and Chug
Lauren –
In response to my posting, you wrote, “I think we agree that the most important thing is for students to understand the math they are doing.”
Please permit me to explain: That is not what I think. In my reading of science, what I have found is: Experts in learning say that for over 90% of students taking math and science, over 90% of the time, students don’t need to “explain why” they do the steps they do to solve a problem. What IS important, according to science, is that they be able to “plug and chug” and get the right answer.
I know that’s pretty much the opposite of what most of us have been taught, but it is newly discovered science that has been verified to be true. Here’s a short explanation of what science says.
At the time over a decade ago when “reform” programs such as POGIL and Modeling were developed, the assumption was that students could solve problems by “reasoning” to apply new, unfamiliar information as easily as they solved by applying very-well-memorized information. The optimistic hope was that with the internet, or by “thinking like a scientist,” “rote memorization could be de-emphasized.
What cognitive science research has discovered and verified, just in the past decade, is that this assumption is essentially the opposite of what is true. This new science says:
“Working memory” (where the human brain solve problems) is able to apply well-memorized facts and relationships with ease but is exceptionally limited when trying to apply “not-previously-well-memorized” information.
In 2018, this finding is measured, verified, uncontested science.
The implications? Students cannot solve problems by “reasoning” or “modeling” or “critical thinking” until after nearly all of the relationships needed to solve the problem can quickly be recalled from long-term memory. So “rote memorization” of the many facts and procedures of science is an essential first step in learning.
Reasoning skills cannot substitute for knowledge in memory. Scientists can solve so many problems because they have for many years to move facts and procedures into memory, and have practiced their retrieval.
To work around working memory limits, the brain needs to plug and chug algorithms. Working around those limits is why learning "standard algorithms" is necessary in problem solving.
On the specific issue of “understanding,” science says that in your major, you need “explicit” understanding: Being able to explain why. But to work in a scientific field that uses knowledge of other sciences as a tool, you need “implicit” understanding: An intuitive sense of how to get the right answer you need, even when you can’t explain why.
For example, if I ask you and our readers:
For 3x + 2 = 29 , A) Solve for x , then B) explain why you did the steps in the order you did.
Being trained as a chemist, you solve for x automatically, intuitively applying a memorized algorithm. But you have a tough time with B). And that’s OK. Chemists DO need to use math as a tool to get right answers. We don’t need to “know why” in order to solve math.
And for the over 90% of our students who the NSF tells us will not major in chemistry, but will need to use what they learn in chemistry as a tool in engineering and health careers, where there are far more jobs than in “pure chemistry,” what they need from chemistry classes is the chemist’s understanding of math: Implicit understanding.
But don’t take my word on this. Read the science from the experts, written for educators.
On how the brain works, see: http://www.aft.org/pdfs/americaneducator/spring2012/Clark.pdf
On explicit vs. implicit understanding: http://www.cogtech.usc.edu/publications/clark_automated_knowledge_2006.pdf
This does NOT mean that the activities in POGIL and Modeling are not useful. What cognitive experts say (see the first link above) is this:
“Small group and independent problems and projects can be effective – not as vehicles for making discoveries, but as a means of practicing recently learned content and skills.”
For practice to help students learn when to apply new knowledge, active learning is vital.
My thinking would be: When science in our field changes, we must change. How the student brain works is science we need to know, because it is central in our work.
But check out what cognitive experts say, and if anyone thinks I’ve mis-interpreted, please reply.
-- Eric (rick) Nelson
In reply to The Necessity to Plug and Chug by Eric Nelson
Interesting
I didn't go read the articles but I find it very interesting. I recently started working for a new school and in the interview I said that I know that they use modeling in the chemistry classes and that I'm not a modeler and don't want to be...if thats a problem let me know and I'll look elsewhere. They told me it wasn't a problem and I could teach more "traditionally" if I wanted to. Well at the end of my first year, I was registered for a modeling class and told that I needed to teach modeling the next year (last year). I was told this after I signed my contract of course. The funny thing is that my students did better on the district end of year assessments, which had many modeling type questions on them, than the teacher who had been doing modeling for years. After doing modeling for a year, I've seen no evidence that the students understand the concepts "more" or "better" after doing the modeling. If anything, I see that we ask them the same questions over and over and over and over again, so that by the time they get tested on it...if they can't answer it...than they really haven't paid attention. But i've seen no evidence of better long term understanding or learning.
However, the reason I replied to your comment...it made me think of a conversation I had with the teacher that has been modeling for years. It was about using formulas. He doesn't give kids formulas, like density or specific heat, he teaches them to do it using dimensional analysis. (even though I pointed out specfic heat doesn't work using dimensional anlaysis...lol) But the specific heat discussion is the one that sticks out in my mind. We have a lot of students who sign up for honors chemistry and engineering 1 at the same time. He said, "Its always a race with specific heat to get to it before the engineering class does. Engineering teaches them the formula and how to use the formula, so if they get to it before we do then the kids know the formula and they just want to use that, but I don't let them use it. So if I get to it before engineering does, then I can teach them my way without them being poisoned with a formula" And I couldn't help but think to myself....why are we fighting with the engineering teachers? And what happens if they get there first and the kids use the formula...do you mark their work wrong? That seems absurd.
Examples
Could you provide a few examples of what it would look like to teach unit conversions using proportions? Thanks!
In reply to Examples by Jessie Martin
Further Reading for Example Problems
Hi Jessie! I think you might find Gary Abud's article interesting: The Two Words Every Chemistry Student Needs to Learn. This strategy helped my students recognize proportions in stoichiometry and beyond. As far as working problems, I think that Lauren's post, Rethinking Stoichiometry, does a nice job of going through some stoichiometry problem examples. She uses the two words, "for every", that Abud supports in the first article link. Another good article walking through what problems would look like was written by Melissa Hemling: Using Visual BCA Tables to Teach Limiting Reactants. You can find other examples (besides stoichiometry) in Ben Meacham's article: Gas Laws and the Over-Reliance on Algorithmic Thinking.
Thanks for your interest,
Deanna
Share Your Thoughts