The Blue Butterfly Effect

The June, 2018 issue of the Journal of Chemical Education contains an article that describes a simple, yet fascinating experiment that you and your students are going to love! It involves the use of butterfly wings from the genus Morpho.1 The wings of these butterflies display the most beautiful blue color I’ve ever seen. I purchased some of these wings2 so I could observe them and also to see what happens when some methanol is dripped onto these wings:
What gives these wings their vivid blue color, and why do the wings change color when methanol is dripped onto them?
First, let’s discuss why the wings are blue. Remarkably, the stunning blue color is not due to chemical pigments, but rather from millions of nanoscopic structures called lamellae that are embedded on the wings (Figure 1). The lamellae contain several alternating regions of ridges and air gaps. The ridges and air gaps are nanoscopic in size: small enough to diffract visible light. Any color of light – except for blue – that interacts with the lamellae is cancelled out due to destructive interference. However, when blue light strikes the lamellae it is amplified via constructive interference. These effects cause the wings to appear an intensely blue color. Color that arises in due to structural elements rather than pigmentation is called structural coloration.3
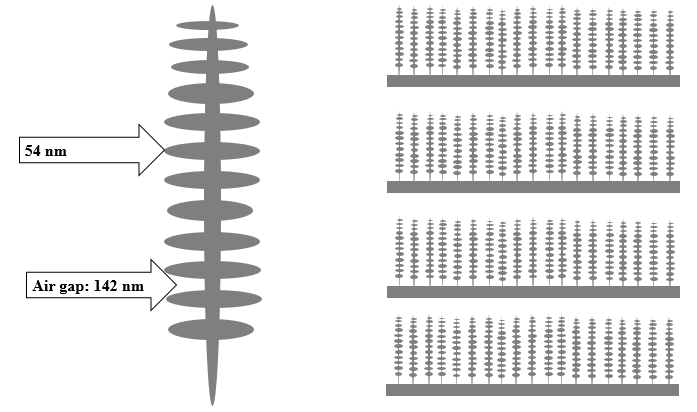
Figure 1: Schematic of lamellae. (Left) a single, nanoscopic structure. (Right) Several lamellae.
The authors of the J. Chem. Educ. article note that the central wavelength of light,
λ = 2( n 1 d 1 + n 2 d 2
Where d1 and d2 are the dimensions of the air gaps and ridges, while n1 and n2 are the refractive indexes of the air gaps and ridges, respectively. Plugging in the known values for the dimensions (54 nm for ridges and 142 nm for the air gaps)1 and refractive indexes (1.56 for the ridges and 1.00 for air)1 we calculate that the scattered light is expected to be centered at 452 nm, which is blue light:
λ =2[( 1.00)( 142 nm) + ( 1.56)( 54 nm)] = 452 nm
We can also use Equation 1 to describe the color change that occurs when the wings are wetted with other substances such as methanol. When this is done the gaps between the ridges become filled with methanol instead of air, which changes the refractive index of the gaps. Using the value for the refractive index of methanol (1.326)1 rather than air in Equation 1, we obtain the result that the center of the wavelength of scattered light should be 545 nm, consistent with a greenish color:
λ = 2[( 1.326)( 142 nm) + ( 1.56)( 54 nm)] = 545 nm
After I observed the color changes that occur on these wings upon adding methanol and other alcohols, I began to wonder what would happen if the wings were soaked in liquid nitrogen. Here’s what I observed when I did so:
Taking into consideration the refractive index of liquid nitrogen (1.200)2 and using Equation 1, a central wavelength of 509 nm is obtained:
λ =2[( 1.200)( 142 nm) + ( 1.56)( 54 nm)] = 509 nm
This value harmonizes well with the brilliant green color observed when these wings are dipped in liquid nitrogen. The fact that the wings appear somewhat yellow-green in color when soaked in methanol but more blue-green when soaked in liquid nitrogen is consistent with the calculations above.
Finally, these wings are very difficult to wet with water:
This effect results from a combination of the high surface tension of water and the very large surface area the millions of lamellae impart to these wings. I like to think of surface tension as the energy required to get a liquid to “spread out” over a certain area. Therefore, because water has a high surface tension (72 mJ m-2),4 to it resists “spreading out” on surfaces – it tends to “bead up” instead. The lamellae on the Morpho butterfly wings mean there are hundreds of millions of crevices on the wing surface, which gives these wings enormous surface area. Because water has such a high surface tension and therefore does not spread out easily, it cannot penetrate all the nooks and crannies introduced by the lamellae. Thus, it cannot wet the wing. This probably comes in handy for the Morpho butterflies, because they live in rainy tropical regions.3 On the other hand, methanol (22.5 mJ m-2)4 and liquid nitrogen (8.9 mJ m-2)5 have considerably lower surface tensions, and they therefore easily wet these wings.
I have found that people young and old enjoy viewing experiments with the blue butterfly wings. In the video below you can view some of these experiments explained and explored in a bit more detail. Let me know if you try experimenting with blue Morpho butterfly wings – especially if you learn something new. Happy experimenting!
Acknowledgement: I wish to thank Bruce W. Baldwin for helpful discussion.
References
1. B. Bober, J. Ogata, V. Martinez, J. Hallinan, T. Leach, and B. Negru, Investigating Nanoscopic Structures on a Butterfly Wing To Explore Solvation and Coloration, Journal of Chemical Education, 2018 95 (6), 1004-1011.
2. I was able to purchase 50 wings for about $50 at The Butterfly Company. When handled carefully, the wings can be used several times. See: https://www.thebutterflycompany.com/product-category/butterflies/morphidae-blue-morphos-others (Accessed 1/29/19)
3. P. Vukusic and D.G Stavenga, Physical methods for investigating structural colours in biological systems, Journal of the Royal Society, January 2009.
4. G. Vazquez, E. Alvarez, and J. Navaza, Surface Tension of Alcohol Water + Water from 20 to 50 .degree.C, Journal of Chemical & Engineering Data, 1995 40 (3), 611-614.
5. Dortmund Data Bank, http://www.ddbst.com/en/EED/PCP/SFT_C1056.php (Accessed 1/29/19)