Bonding Interactive Notebook

Hang on to your hats, folks! We are about to enter my longest notebook unit of the year: Bonding. This unit is massive with 6 two page spreads. Read on to see my discussion of pros and cons of splitting it into two units versus keeping it as one big unit.
Before I get into the details of how I teach bonding with interactive notebooks, I want to let you know that you can go back and watch a ChemBasics Talk that I gave on Bonding last year: Chemical Bonding and Naming with Nora Walsh. In that talk I discussed my entire bonding unit, including how I fit interactive notebooks into the rest of my activities.
Bonding is a unit that can be taught in a variety of ways. You can teach it as one big, continuous unit or you can break it into smaller units. I have done all of these formats: ionic bonding and then covalent bonding; principals of bonding and then naming; and finally, one big unit with ionic flowing into covalent (using polyatomic ions as the link the between ionic and covalent). I have found that when I teach ionic bonding and covalent bonding as separate units, my students then want to put them in neat little separate boxes and get confused when I explain polyatomic ions in terms of covalent bonds. They also push back against the final task of classifying mixed examples of compounds because they start out with each type being isolated. When I teach principals of bonding first and nomenclature second, the students do well with the principals of bonding, but the nomenclature seems forced and isolated and a drag. I now teach Bonding as one big unit with multiple formative assessments throughout. In each topic I teach the principals of the bond and the nomenclature as integrated concepts. I then build onto each concept throughout the unit, and find that this flow works best in my classroom.
By the time I get to bonding, I have already taught students about ions in terms of subatomic particles (in my Atomic Structure unit). I teach ions again at the beginning of my Bonding Unit, focusing on the gain or loss of valence electrons (previously we had examined ions in terms of the balance of protons and electrons). I have students produce different models that show comparisons between neutral atoms and ions, really driving home the concept of valence shells and the gain or loss of valence electrons to fulfill the octet rule. This leads naturally into a lesson on Binary Ionic Compounds. I take several days with Binary Ionic compounds, starting with compounds of fixed charges and then introducing cations of variable charge. Next comes learning about polyatomic ions and how they combine with other ions to form ionic compounds, and then I use polyatomics as a bridge between ionic and covalent compounds. Students fill in a Covalent Compounds page as a guided reading – I try to use a variety of methods when filling in notebooks (for more on this, see my blog post A Classroom View of Using Interactive Notebook Pages). Forming and naming Covalent Compounds has its own spread, followed by a page with Lewis Structures and Lewis Structures practice. I end up the unit with a lab on Ionic, Covalent and Metallic Solids. I will have students come back into class the day after the lab and link the macroscopic characteristics of the three types of bonds (Ionic, Metallic and Covalent) with symbolic and submicroscopic representations (for an article on the value of students examining all three representations see Ben Meacham's ChemEd X post on Johnstone's Triangle: Why Do You Make Us Draw so Many Particle Diagrams?). There is a LOT going on in this unit – again, please strongly consider using many, many assessments throughout this unit and ancillary activities outside the notebook to make sure your students master the application of the material that is summarized in the notebook.
Materials
Background
For backstory about how Nora uses interactive notebooks, view a recording of her ChemEd X Talk: Integrating Interactive Notebooks into Chemistry Courses with Nora Walsh and read her previous post, Interactive Notebook Unit on Stoichiometry. Nora wrote the following in response to questions she received after her ChemEd X Talk: A Classroom View of Using Interactive Notebook Pages.
Nora's interactive notebook units published so far: Scientific Reasoning, Science of Matter, Atomic Structure, Periodic Table, Bonding, Stoichiometry, Gas Laws, and Thermochemistry. She plans to publish all of her units by the end of 2022.
Procedure

Spread 1: Ions and Valence Electrons
Rightside – All About Ions
This is a page layout that I have students glue directly onto the page, and then paste a hamburger foldable at the bottom that compares cations and anions. They define valence electrons at the top of the page, and then label the columns of the periodic table with the number of valence electrons as well as the corresponding charge. This comes quickly if you have already done an activity with ion formation. They fill in a bit of information in the middle of the page about the octet rule, and then at the bottom of the page they compare cations and anions side-by-side with the foldable. Inside the foldable, information is recorded about the name of the ion (it stays the same for the cation, but with the word “ion” after it, versus adding the “-ide” suffix for anions), Bohr models of atoms, and then some basic information about the formation of ions relative to valence electrons.

Leftside – Models of Neutral Atoms and Ions
I have always done some kind of activity here that has students compare atoms and ions, and you may already have a favorite activity you would like to use. This year I modified a previous activity that only used Bohr diagrams to include multiple representations of electronic structure. I did this so that students could continue to think about strengths and limitations of various models, and also to provide a spiral review of previous content. If you choose to use this output, it is a tab book – print each page onto a single page of paper and then staple it together at the top. If you are looking to decrease your paper use, you could eliminate the cover page, and try shrinking the pages so that you have two pages per 1 sheet of paper.

Spread 2: Binary Ionic Compounds
I have students do a drag and drop activity, either by using paper dots (quickly make them using a hole punch and construction paper) or using these Google Slides to help them see how electrons are gained/lost to fulfill the octet rule. I do naming right away with these binary compounds – I have found that naming is easier for students if it is introduced in bite-size chunks and closely tied to the examination of the formation of the bond.
Rightside – Ionic Compounds
This page is an overview, and I use it as a summary after we do a variety of activities. The top part of the page is very general information about how ionic compounds are formed, and also very general information about how they are named. The middle of the page shows the Criss-Cross method, but I specifically chose an example in which the students need to understand that charges reduce because it’s a proportion and not just a direct transcription. Next to this is a place for students to consider transition metals in naming. At the bottom of the page is a table to practice naming. Because of the wide variety of content on this page, I fill it in over several class periods.
Leftside – Ionic Bonding Activity
After doing the drag and drop activity, I have students do an Ionic Bonding Puzzle Activity (this version1 is available on AACT, a digital version can be found here, and many others are available via Google search). You can have students paste the puzzle pieces onto a piece of construction paper that you can fold and either tuck into a pocket, or glue down on the left page.
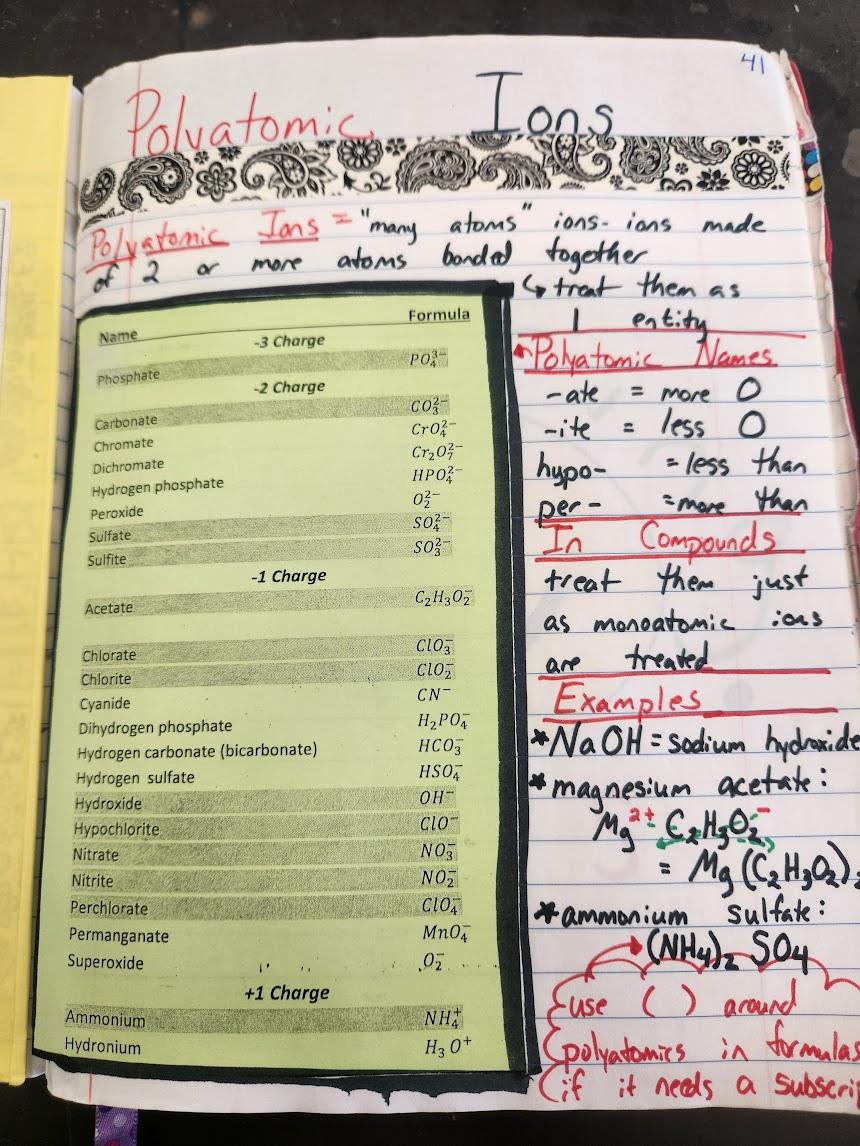
Spread 3 – Polyatomic Ions
Rightside – Polyatomic Ions Glue-down
This glue-down is pretty straight forward. It doesn’t take me long to have students fill it in. I have them identify patterns they observe in the polyatomic ions table, and we then fill in some notes about polyatomic names and how they relate to the structure. We wrap up the page with a few examples.

Leftside – Polyatomic Practice
This output can be anything that has students do some kind of polyatomic ions practice. I have included two options for you: a table of ions that they use to write the formulas, and a game that facilitates practice of the same skill using a ball toss.
The table is pretty straight forward – I have students fill in both the name and the formula of the compound that is formed by each combination in the table.
The ball toss is a highly interactive and fun activity. It takes a bit of set up the first time you run it, but once it’s made it will last for years. Get a couple of cheap balls – beach balls work well because you can deflate them. Make one ball the Cations ball, and the other one the Anions ball. Using a Sharpie, write names/formula of cations scattered on the surface of one ball, and then scatter anions on the other ball. Break students into small groups (the number of groups you have determines the number of ion balls you will need to make). Have one group GENTLY toss the cation ball and then the anion ball to the other group or partner. The ions closest to the catcher’s right index finger on each caught ball are the ions they use to write a neutral formula. Play several rounds, and have students record the ions and the resulting formulas on this page.

Spread 4 – Covalent Compounds
Rightside – Covalent Compounds
As mentioned in the introduction above, this is a glue-down page that it is a guided reading from ChemLibreTexts (for more on this, see my blog post A Classroom View of Using Interactive Notebook Pages). Students use a posted article to read and apply knowledge of bonding to covalent bonds. It also has the rules for naming covalent compounds. This reading is the initial instruction on covalent compounds. This initial information will be fleshed out by the application of the facts on this page to Lewis Structures on the next two-page spread.
Leftside – Ionic vs. Covalent
There are a wide variety of practice options here. I have a coloring page that I got from another teacher that I use. A card sort would work really well – I would highly recommend checking out Chad Husting’s Ionic Covalent Compounds and Card Sorts post for an idea on what this could look like. The categories he talks about could be written in the notebook, and students could either write or paste the card prompts into the correct categories.

Spread 5 – Lewis Structures
Rightside – Lewis Structures
This page has a few layers. The top of the page has a split flap that illustrates bonding and lone pairs by relating them to a Lewis structure. Next to that is a periodic table flap for students to color the diatomic elements. I always tell them to remember the Rule of 7: Start at element 7, trace over to column 7, trace down to draw a 7, and then add H to remember the 7 diatomic elements. Underneath those pieces of information are the rules for drawing Lewis Structures. I have played with this being a flap or a foldable, but in the end decided to leave it as a fill in section. I have students use different colors when noting each step, and then use the same color as they apply each step to their Lewis Structure example.
At the bottom of the page is a mini flip-book to compare types of bonds (single, double and triple). I use the examples of ethane, ethene and ethyne as required by my (former) state standards. You are welcome to change those examples or add more (diatomics work well here). Students fill in information about the relative length and strength of these bonds and draw the Lewis structure of the example(s) on the leftside of the flipbook (the blank backside of the previous layer).
Leftside – Lewis Structures Practice
This is a simple foldable (I call it a spine foldable) that gives students work space for Lewis structures. I have selected 4 different molecules that require varied Lewis structures. Students keep their valence electron tally on the flap and then draw the Lewis structure directly on the page. You can certainly add more examples to this if you wish, but I do lots of Lewis structures practice outside the notebook and have decided that 4 examples on this page for review later is enough.
An alternative to this foldable would be to have students physically build Lewis structures. I do this activity outside of the notebook because I have students take pictures of their models and upload them to Google Classroom for scoring purposes, but it is highly interactive and would work well here. I usually use marshmallows, pretzel sticks and mini chocolate chips to represent atoms, bonds and lone pairs (respectively). If you don’t want to use food, toothpicks/straws, playdough or Pom Poms, and small beads work well.

Spread 6 – Types of Bonds Comparison
Rightside – Properties of Bonds Foldable
As mentioned in my intro, this foldable sums up the entire unit. It links together the chemical basics of the bond with formulas, names, particle view diagrams and macroscopic properties. I have them fill in this foldable AFTER we do the lab on ionic, covalent and metallic solids. (Remember: Activity Before Concept). This is a simple tri-fold sheet that pastes onto the last page of the unit.
Leftside – Comparing Solids Lab
The leftside of this page is where I have students insert their lab handouts. They either make a pocket to tuck it into, or you can shrink it to 75% and have them tape the page down the side or across the top so it lifts. If you choose to instead have your students analyze the lab digitally, this would be an excellent place to have your students create a word cloud, one pager, or cartoon that relates the 3 types of bonds and their solids.
It might be helpful to view video 1 to help visualize what these pages will look like in action.
Video 1: Bonding INB pages, ChemEd X Vimeo Channel, 10/21/2022.
- M. Bratsch, Ionic Bonding Puzzle, AACT Classroom Resource, March 2020.
Questions
Concepts
Time Required
Preparation
Provide copies of the following for each student. Note that I have grouped the components by page. Please preview them before printing as some pages have multiple copies to minimize paper waste.
Spread 1:
Spread 2:
Spread 3:
Spread 4:
Spread 5:
Spread 6:
Attribution
*Webinars Referencing Interactive Notebooks: ChemEd X Chem Basics, October 2021 – Chemical Bonding and Naming and AACT , February 2020 – Teaching Chemistry in a Textbookless Classroom
**I have three PLC colleagues that have supported this work as we always review the pages and talk through content together. Many thanks to Tom Jankowski, Lori Young and Mihir Paranjape.



Comments
7Notes
Hey Nora! You inspired me to try interactive notebooks in chem, so I'm trying it with my ELL students this year. I love it so far, and I'm so grateful for all of your shared resources. Do you by any chance have a photo of the completed notes for this unit? Specifically the binary ionic compounds page - I'm not sure about the wording for the special considerations section. Thank you so much!
In reply to Notes by Jessica Pittarese
Notes Key
Hi Jessica! I'm so glad you are enjoying notebooking! I apologize for the delayed response - I was sick at home but my notebook was at work. Here you go!
keys
It would be great to try this out as our textbook does not have the greatest presentation of bonding. However, it would be really helpful to have full keys so I can make sure I have pre-taught all needed skills and understand where each section is going.
Thank you for sharing this!
In reply to keys by Dedra Hopper
Keys
Hi Dedra! I'm so glad that you're interested in trying these out! I don't publish the full keys because I don't want students to be able to find them. I'm very happy to communicate with you (or anyone) directly who would like keys and provide them to you privately. Feel free to email me: nora.walsh@evsck12.com.
Share Your Thoughts