Scientific Reasoning Interactive Notebook Unit

Ah, sweet summertime. The time when teachers are torn between relaxing and recharging, and beginning to do planning for next year in hopes of feeling more prepared for the school year. If you find yourself looking for a way to refresh your teaching next year while supporting student ownership and creativity during the learning process, then interactive notebooks may be what you’ve been looking for.
During the spring of 2022, I began sharing my interactive notebook units. I started where I was in the content, and I have already shared my units covering stoichiometry, gas laws, and thermochemistry. I still have three units left from my second semester curriculum (solutions, acids and bases and nuclear), but I’m going to leave those for now and shift to my semester one content.
This post will be about what I call Unit 0 – Scientific Reasoning, and a bit about how I work with notebooks from the beginning of the year.
The first thing I should tell you is that my students do not receive a supply list over the summer (I suspect many high school students don’t). I tell students on day one to purchase a composition notebook, and recommend that they have it by Monday of week 2. In my experience, composition notebooks are BY FAR the best thing for students use for their interactive notebook (INB), for several reasons:
- They are cheap and widely available at many stores.
- Students can choose color/pattern to express their own style and differentiate their notebook from others.
- They are bound in a way that will last.
This last point is the biggie: any time I have had students use spiral bound notebooks or anything that has glue binding instead of stitched binding, the notebooks have fallen apart within weeks. One year I had this happen myself when I discovered that some composition notebooks are glued instead of stitched. (Have students look for a composition book with a curved spine instead of a flat spine – the curved spine is stitched.) I like to structure the first several activities of my curriculum to give them time to purchase a composition notebook. Like most of us, my first two or three days of class revolve around school start up tasks– we go through the syllabus, we create our own set of class rules, we build class identity, and we talk about the importance of chemistry in their lives and why the course is valuable for them as citizens of the modern world.
If you want all of this information in your INB, no worries – just format your activities in a way that it will be easy for students to paste them into their notebooks later. Here are some ideas:
- Format your syllabus as a pamphlet
- Give students a composition notebook sized-page for them to illustrate the class rules
- Shrink your getting-to-know-you handout so that it fits on the composition notebook page
- Make flipbooks or foldables that will be a fast glue-in later
I use this last tip when I do my lab safety lesson, which occurs in the first week of class when many students do not yet have a notebook. Students create a flipbook of lab safety by cutting and pasting the labels and matching them to the rule sections outlined in the document. This is a subtle, unstated introduction to a right side - “input” activity. Then they walk around the room to create a lab safety map – their first left side - “output” (see figure 1). When we assemble the notebook on Monday of week 2, they paste these two items into their beginning pages – after the table of contents but before Unit 0. The title of the page is, fittingly, Safety First. (For more about input and output and some backstory on my interactive notebooks, check out my first post on the subject: Interactive Notebook Unit on Stoichiometry.)
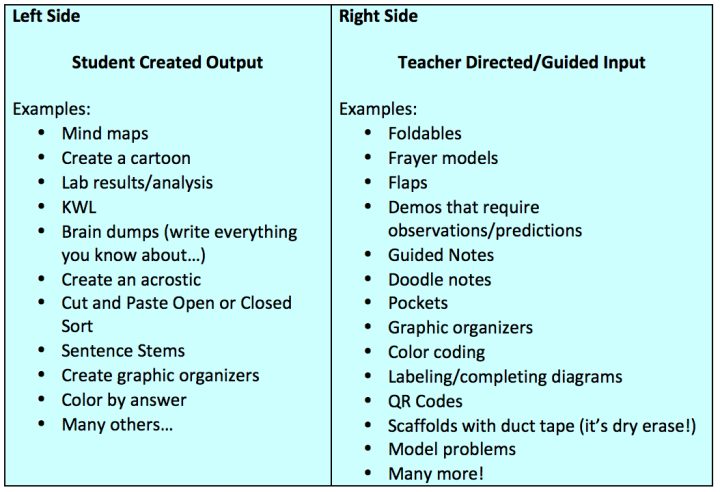
Figure 1: For every page of teacher directed “input” there should be a corresponding student created “output”. Ideally the left side should be very open ended and allow for lots of student interpretation.
If you have questions or comments, please log into your ChemEd X account and add a comment below the post.
Materials
Composition notebooks, printed copies of each page, colored paper or construction paper for making flipbooks/foldables, scissors, tape or glue
**A note on my manipulatives: If you see a dashed line, that is where the item should be cut. If you see dotted lines, that means fold.
Background
For backstory about how Nora uses interactive notebooks, view a recording of her ChemEd X Talk: Integrating Interactive Notebooks into Chemistry Courses with Nora Walsh and read her previous post, Interactive Notebook Unit on Stoichiometry. Nora wrote the following in response to questions she received after her ChemEd X Talk: A Classroom View of Using Interactive Notebook Pages.
Nora's interactive notebook units published so far: Scientific Reasoning, Science of Matter, Atomic Structure, Periodic Table, Bonding, Reactions, Stoichiometry, Gas Laws, and Thermochemistry. She plans to publish all of her units here on ChemEd X.
Procedure
Before Unit 0

Spread: Safety First
Right side (see above) - Students do a cut and paste activity to organize the lab safety rules into a flipbook for reference. This serves as their lab safety contract. I have provided the contract that I currently use, with dashed lines indicating to students where they should cut. You could also have them dissect any other lab contract you choose to use to create a flipbook. (Just remember to print one sided only!) Creating a flipbook here is really great for all of your students, but especially those who don’t yet have a composition book. It organizes the material into one place, and it’s very easy and fast to paste into the notebook once they have it.
Left side - Students are given a bare map of the room, and label it with various safety features. This is then pasted into the notebook once they have one.
Unit 0 – Scientific Reasoning
Spread 1 – The Scientific Method
Right Side - Doing and Communicating Science/Chemistry
If you cover "areas of chemistry", this is where I include them. A simple, small hot dog foldable does the trick – the areas are labeled on the outside (analytical, biological/biochemistry, chemical engineering, inorganic, organic, physical), and then the inside shows definitions and examples of applications. I don’t have a template for this, because I just have students create a simple foldable. I like to direct students to the Areas of Chemistry webpage on acs.org and have them use the sections and short readings to complete the foldable. This also brings some literacy into your notebook, as well as addressing NGSS crosscutting concepts. (Note: Last year, I had students do this activity as a Google Slides presentation instead of a foldable, as our district tasked us with having students submit a digital assignment within the first week of school to introduce them to Google Classroom. So you will not see a place for this foldable on my layout below. However, you can dissect and rearrange my page as you wish to make the layout work for you, or you can have this foldable be its own stand-alone two page spread.)
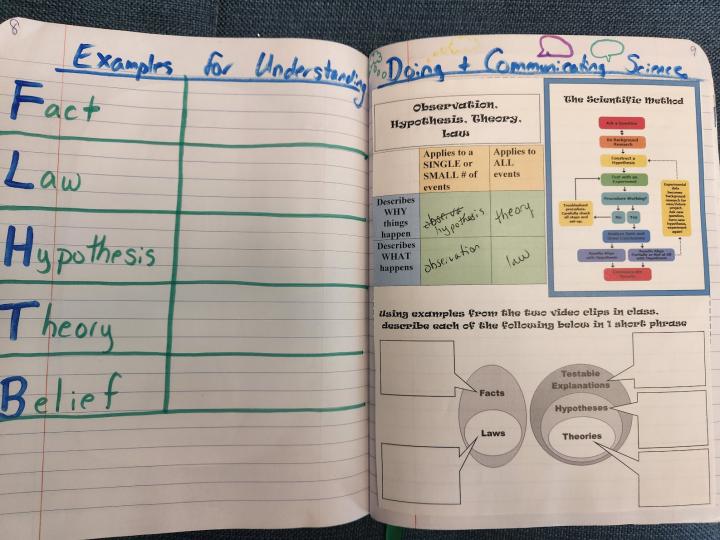
In surveying and questioning students, I found they have covered the scientific method in every prior science class. This is great but it feels repetitive for me to do the same kind of activity they have previously done several times. So I adjusted my lens, and the main focus of this page is to address common misconceptions that exist around some of the terms we use in the scientific process. I show two video clips (TED-Ed: Theory vs. Law What’s the Difference and It’s OK To Be Smart: Theory vs. Hypothesis vs. Law) and have students use the information presented in these videos to fill in a couple of graphic organizers that help them differentiate the terms.
Left side - This year, I had students create a table where they paste/copy in examples of Facts, Law, Hypothesis, Theory, Belief taken from this NASA created activity.

As an alternative activity, in previous years I have had students paste a clipart picture or sketch an image of a scientist, and then do a brain dump of words/phrases describing what they think a chemist does (see above).
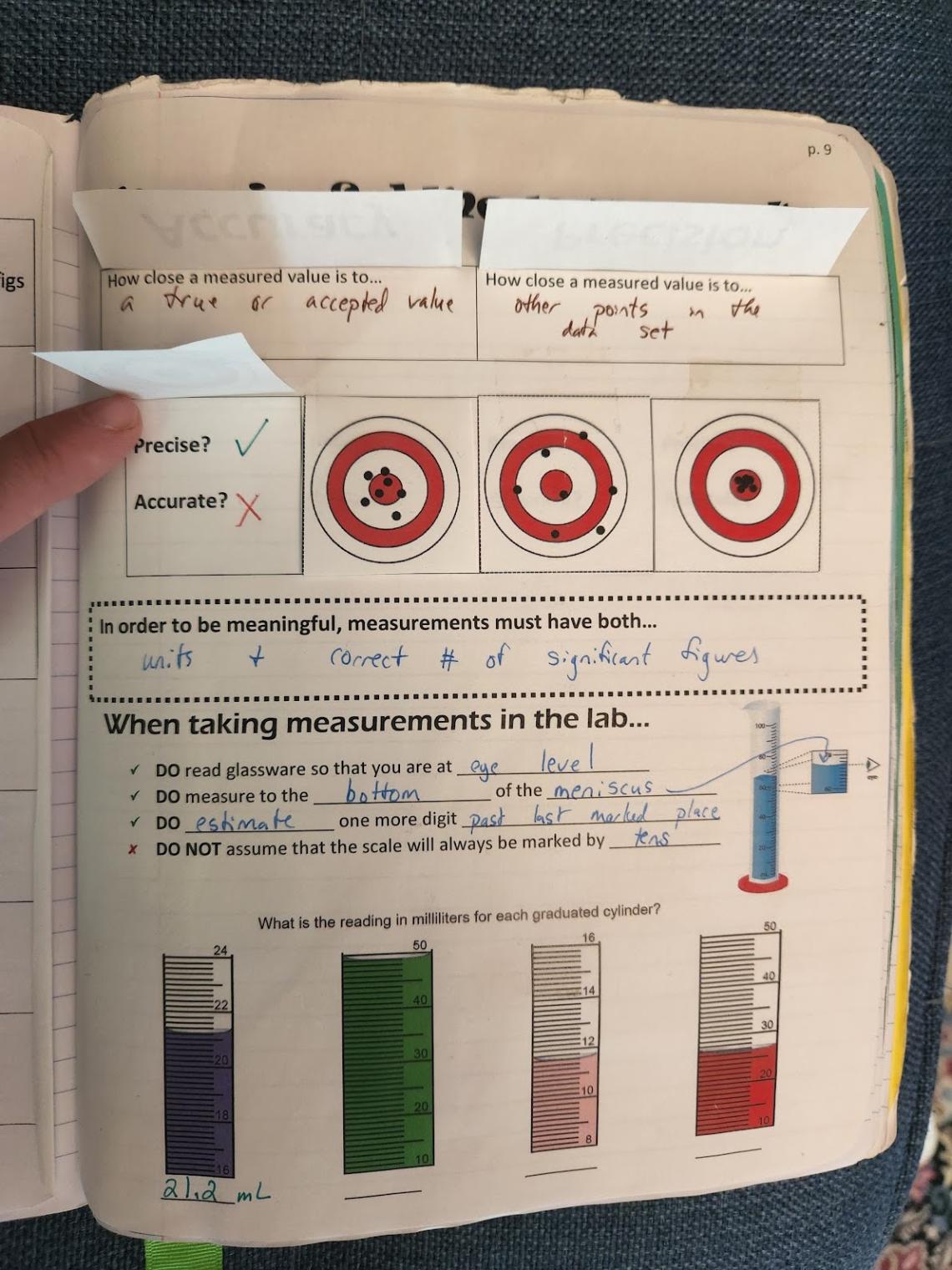
Spread 2- Meaningful Measurements
Right Side - On this page, I cover all things related to measurements in the lab: accuracy, precision, reading glassware, and the components of valid data. Flaps are created that define accuracy and precision, which students fill in while watching TED-Ed: What’s the Difference Between Accuracy and Precision. The classic example of targets are used with various data sets represented as points, which are flaps that students then lift and classify as accurate/precise. They fill in a statement about what meaningful measurements must have, and then fill in information about accurately reading glassware and practice with four images of glassware (created using the worksheet generator from Math-Aids.com).
Left Side - There are several options for output here, depending on what you want your focus of the page to be and how you want to deliver it. You can nicely bring in some NGSS Science and Engineering Practices here. I have provided two options, and I’m sure you can find many others online.
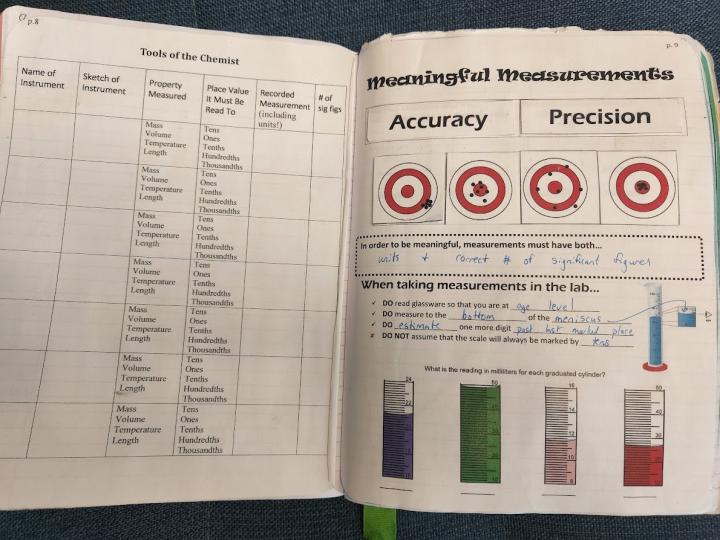
Option 1: Tools of the Chemist Lab (see above)
In previous years, we have had the output be a lab activity we called Tools of the Chemist, where students rotate through various glassware and measuring tools setup in the lab, and then record the values represented on the instruments. We still do this lab, but instead of having students do it on paper, we now have them enter their answers on the computer. The paper version is attached.
Option 2: Accuracy and Precision Data Set
Last year, because we made Tools of the Chemist a digital submission, I had students analyze molar mass data sets for accuracy and precision. This is also shared for your use. If you would like to up the rigor of this activity, remove data set 5 and have them research and create this own data set, and then go through it with a neighboring group.
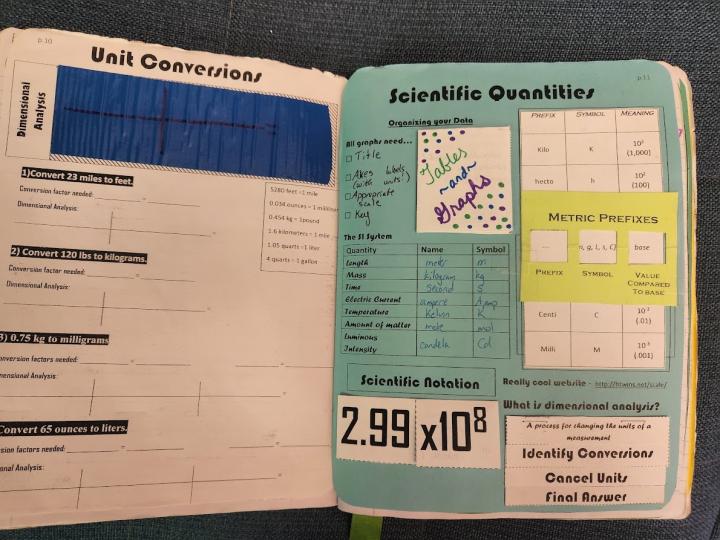
Spread 3 – Scientific Quantities
Right Side - This page is a catch-all of the math crossover we have in the lab: metric prefixes, the SI System, Scientific Notation and (optional) significant figures. If you do an early intro to dimensional analysis, that could also go on this page. I used to introduce dimensional analysis at the beginning of the year, but in the last two years have decided to wait because we don’t use it heavily until Chemical Quantities (which is a winter topic for my district).
Students compare types of graphs on this page, usually by examining some graphs themselves (there are lots of online resources for activities with graphs), and fill in a small accordion-style foldable. Then they create a slider that highlights the metric prefixes (if you need to save time, just make this a glue down) and then fill in a table with the SI units. The slider holder is a separate document, and should be printed on cardstock – you need the rigidity of a heavier paper so that it doesn’t rip as you slide the table through it. Finally, we review the format of scientific notation. If you teach significant figures, I have also included a foldable for identifying sig figs (see Video 1).
Video 1: Significant Figures Foldable, ChemEd X Vimeo Channel, 7/5/2022.
Left Side - My practice here is usually to include some type of worksheet. As much as possible, I try to make my output open-ended, but I have found that this page needs a more concrete set of practice problems to identify student-specific stumbling blocks. On the last page of the layout, you’ll see that I’ve included a set of practice problems. At the top of the page students paste a strip of duct tape (duct tape is dry erase). They draw the scaffold in sharpie, and then use the duct tape scaffold to structure their work. I don’t always use this page of problems for practice – it really depends on how well my students are doing with the algebra.
It would also work really well to do a pre-quiz on the topics addressed by this page the day before, and then tailor your left side output practice to the greatest needs indicated by that data, or even have different output pages for individual students as demonstrated by the data.
Spread 4 – Density
Right Side - A few years ago, I was looking for inspiration to freshen up my density page, and I stumbled across this free foldable from Science and Math Doodles on Teachers Pay Teachers. I use the density foldable from the bundle, and then add a few extra notes around the edge.
Left Side - The output on this side is either a lab or a set of practice problems. In building algebra skills, we have been using practice problems the last few years, and then have them do the lab as an online submission. But either could work well, as could using an online SIM or CK-12 activity. We also found a fun, free density digital escape room that was a hit with our students.
If you are having any trouble visualizing what these pages will look like in action, check out the video below.
Video 2: Scientific Reasoning INB pages, ChemEd X Vimeo Channel, 7/5/2022.
Questions
Concepts
The first week or two of my course seems to alter slightly every year, depending on timing, school requirements, and changes to our standards or district curriculum map. But generally, in Unit 0, I cover most of the following topics:
- Areas of chemistry
- The Scientific Method and related vocabulary
- Theory vs. Law
- Accuracy vs. Precision
- Reading glassware
- Types of glassware
- The SI System
- Scientific Notation
- Graphing
- Significant Figures
- Density
I have shared components that you can use on your page for each of these items. I’ll discuss that in more detail below as I go page by page.
Time Required
Preparation
Provide copies of the following for each student. Note that I have grouped the components by page. Please preview them before printing as some pages may have multiple copies to minimize paper waste.
Interactive Notebook Rubric - Scientific Reasoning
Spread 1:
Spread 2:
Accuracy and Precision Data Analysis
Spread 3:
Metric Prefixes slider holder - print on cardstock
Attribution
*Webinars & Presentations Referencing Interactive Notebooks: ChemEd X Chem Basics, October 2021 – Chemical Bonding and Naming and AACT, February 2020 – Teaching Chemistry in a Textbookless Classroom
**I have three PLC colleagues that have supported this work as we always review the pages and talk through content together. Many thanks to Tom Jankowski, Lori Young and Mihir Paranjape.


Comments
18Fall Units and I can statements
What other units do you teach in the fall? Also, can you share your list of I can statements? Looking forward to Monday :)
Thanks, Penny
In reply to Fall Units and I can statements by Penny Shrum
Re: Fall Units and I Can Statements
I can't wait to talk to you on Monday!
In the fall, I teach:
I will try to remember to share my I Can Statements unit-by-unit moving forward. Here they are for Unit 0 - one year I had students paste in a checklist, which I liked, but I haven't kept up with that (I'm not sure why).
Unit 0 I Can...
Nora
Gratitude
Thank you for sharing this!
I learned about interactive notebooks only after leaving a 10-year stint teaching high school chemistry and moving to middle school science. If you have ANY suggestions about resources for creating IANs for grades 5-8, please share!
In reply to Gratitude by R2-N2
You're Welcome and Middle School INBs
Hello Tim! I think middle school is a GREAT age for INBs! Also, you're in luck - they are much more widely used in middle school so there are many great resources out there for middle school standards. Search for INB and ISN (interactive science notebook) on Google, and try both broad and specific terms. Pinterest is a great starting place for lots of INB materials - you may want to start with my board and branch out from there (https://www.pinterest.com/norawalsh/interactive-notebook/). I get much of my inspiration on Pinterest.
Middle school is such a critical age for keeping their interest in science, so ISNs can be a really fun way to keep the spark alive! Good luck, and please reach out if I can support you at all. Also, follow along with my units, especially the next few months - much of my early content may work in middle school.
Nora
Presentation
Logistically speaking, do you have a Google Presentation that goes along with all of this for giving the "Input" information ?
Yes, I do use a Google presentation to show them the layout and what goes on each page. They open it and reference it when assembling the page. When I'm having them work independently to find the information, I will also imbed requirements and/or suggested links, or any other procedural information they need to fill in the page (or part of the page).
In reply to Yes, I do use a Google by Nora Walsh
Presentations
Do you use presentations for lecture or do you not really lecture or? :) I'm just trying to get the whole picture for how you teach using the ISN. I have used ISN for several years and I am always changing them up and looking for ways to make them better for my chemistry classes. Thank you for sharing your work!
In reply to Presentations by Holly Solomon
Re: Presentations
I use presentations to show students the layout, and sometimes to post resources that they will use to fill in their notebooks. I push out that presentation and tell students to not make a copy - I want them to access it as the live presentation so I can update it as we move through a unit.
I try to not lecture very often or for very long. I do sometimes use presentations - other times I do a "chalk talk" and still other times we use the notebook as a way to summarize information that they gained independently in a prior activity (such as a PhET SIM or a POGIL activity).
For more in-depth discussion on the ways I share the content information in my INBs/ISNs, see my blog post: https://chemedx.org/blog/classroom-view-using-interactive-notebook-pages
Thank you for the comment - it's nice to hear that even experienced notebooking practitioners are finding these posts useful!
Nora
How to Make the Safety First Foldable
I was asked on social media about how to assemble the safety first foldable. This is called a layered book foldable, and in order to make this you use 3 pieces of regular printer paper. Simply stack the three pieces of paper, staggered so that you have about 3/4"-1" of edge showing (this is where you will put your category labels). Then you fold the stack over, so that you maintain the stagger, and then staple across the top fold. This should create 6 evenly spaced reveals. You will need to trim the sides of the paper to make it fit into the notebook. I keep scrap pieces of paper in a basket in my room - students use them later on to make flaps and other components on later pages. I hope this helps with some of the construction logistics.
Unit 0 – Scientific Reasoning Spread 1
I am excited to try your materials this year! I was wondering if you have a pdf of your handouts for Scientific Reasoning Spread 1 ver 2 (the right side)? Thanks!
In reply to Unit 0 – Scientific Reasoning Spread 1 by Emily Markovitz
Added to content
It's now been added to the rest of the downloads!
Unit 0 - Scientific Reasoning Notebook Resources Teacher KEY
Nora, I have used Interactive Notebooks in middle school science for 9 years but have moved into a high school Chem position. So, I am happy you are publishing your Chemistry notebook resources!! I was wondering if you have a KEY that shows a completed version of the notebook resources you provided here. Thank you for supporting high school Chemistry teachers!!
In reply to Unit 0 - Scientific Reasoning Notebook Resources Teacher KEY by Rachel Pedler
Re: Key
Rachel, I'm so glad you're excited to use interactive notebooks! No, I don't have a complete key. I filled in all of my notebook the first year I made one, but I evolve and tweak it every year and I have stopped creating a complete resource because I no longer need to. I still fill in some of the portions as we go through so I remember what I put in for consistency, but many other times it's information I have recorded elsewhere.
If there are particular portions that you are confused about, please feel free to post a message and I can tell you what I'm thinking. Also, if you watch the video flip through, most of the pages show at least some of the filled in material.
Thank You
I find your interactive notebook format and the resources extremely useful and thank you for sharing your hours of hardwork with everyone.
Just a quick question: Do you usually have these pages completed during class or given as homework?
In reply to Thank You by Rose Joseph1
When do we make pages?
Thank you so much! I'm glad that the resources are useful!
I usually do these pages in class. I do a lot of the time-consuming prep up front: I pre-trim the big cuts, and sometimes even do a rough pre-fold so that's obvious where they need to crease it. They use double-sided tape to put in the foldables, although glue sticks work well also. I usually give them a count timer of about 5 minutes for construction, and then we fill in the content and notes together.
I did try having them do the notebook at home, but it didn't go well. It required to many materials that students either didn't have or couldn't find at home, and so I ended up taking time in class for about a quarter to half of the class to put the pages together. In the end, that wasn't worth it so I just tightened up procedures and it's been fine!
Share Your Thoughts