Atomic Structure Interactive Notebook

By the time we get into the atomic structure portion of my class, things are well under way. The students know me and my expectations, I have learned their names, and we have our classroom dance well-choreographed. This unit is short in terms of the notebook – just a two page spread. But these two pages contain a lot of information.
While this may seem very dense, I think that the way the information is grouped helps students to see connections within the material. It does generally take me more than 1 day to do a spread in Unit 2, and I make sure to do a lot of spiraling practice with this content. I also find that this unit is a mixed bag in terms of prior content knowledge. Some students have practiced these skills before and quickly refresh while the skills are new for others. I try to have a variety of practice for outside the notebook so that I can differentiate, provide extra support and also have extensions.
Materials
Background
For backstory about how Nora uses interactive notebooks, view a recording of her ChemEd X Talk: Integrating Interactive Notebooks into Chemistry Courses with Nora Walsh and read her previous post, Interactive Notebook Unit on Stoichiometry. Nora wrote the following in response to questions she received after her ChemEd X Talk: A Classroom View of Using Interactive Notebook Pages.
Nora's interactive notebook units published so far: Scientific Reasoning, Science of Matter, Atomic Structure, Periodic Table, Bonding, Stoichiometry, Gas Laws, and Thermochemistry. She plans to publish all of her units by the end of 2022.
Procedure

Spread 1 – History of Atomic Structure
Rightside – Atom Timeline
Over the years I have collected a number of resources, including videos and external sources, into a large PowerPoint that I use to support my walk through of the history of atomic models. As I talk them through it, students fill in the timeline template. Our current state standards focus on what evidence caused the model to change, which is a skill that is great to build before students go into AP classes. On my timeline template I have already noted the scientists’ names, the date they are credited with the breakthrough, and a very brief description of what they did. There are two boxes for students to fill in that correspond to each model change -one to sketch the model of the atom as seen by that researcher, and then a second box to fill in with what caused that model to change. I also have students jot down Dalton’s Atomic Theory on the backside of the timeline. I let students know that it is a good starting point as we begin to think about compounds. (Over the course of the year, I periodically ask students if new facts support or refute Dalton’s Law, which leads to some really interesting conversations.)
The way I have always taught this lesson is very teacher-centered. My goal this year is to make it more student-centered, while still ensuring that students get to the correct information. I think a guided WebQuest or a jigsaw activity would work really well also. (I tried an open web search with my honors students one year with mixed results. I think guiding students to vetted sources would work much better.)
Students fold the atomic timeline into quarters and paste it on the top of the page. Below that is a fill-in table that compares protons, electrons and neutrons. This past year, I had students fill in this table as a warm-up the day after the atomic timeline lesson.
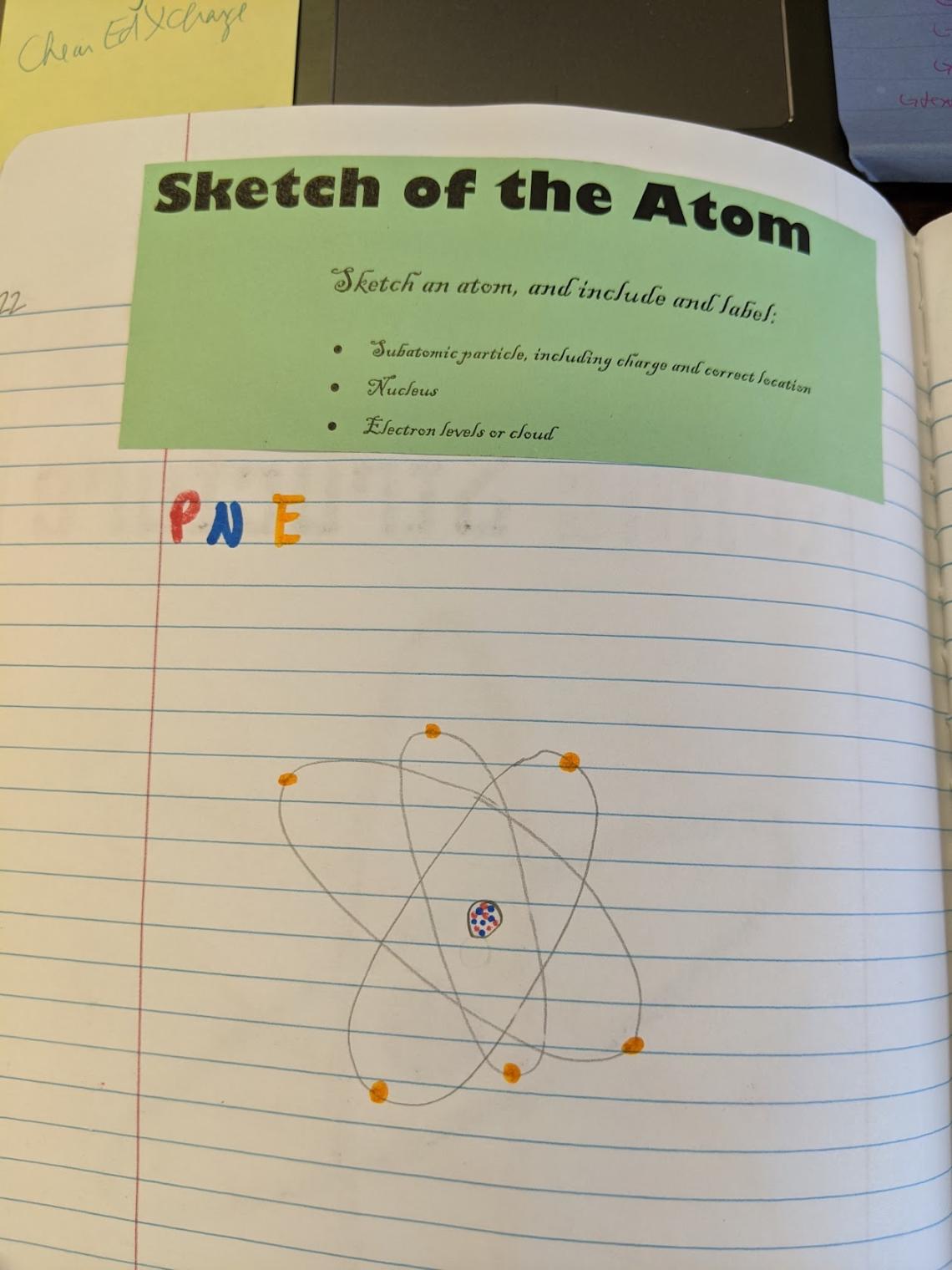
Leftside – Model of the Atom
For this output, I have students sketch an atom. I have a few requirements for them, and that is outlined in the guidelines that they paste at the top of the page. This model allows me to check for comprehension of the location and placement of the subatomic particles before we dive into the nuances of counting particles.
Spread 2 – Quantifying Atomic Features
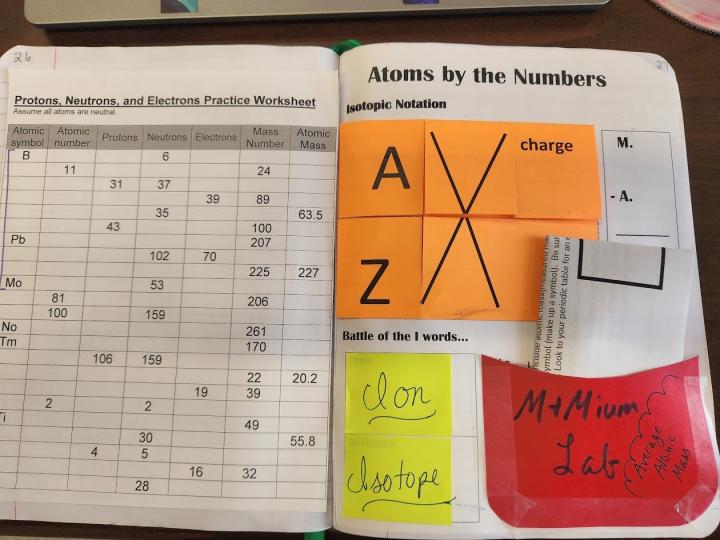
Right side – Atoms by the Numbers
This is a very information-dense page, and I break it into several days in my class. We first begin by defining isotope and ion – students write this at the bottom of the page in a space I call “Battle of the I Words”. These boxes aIso work well to have flaps covering the definitions. Have students tape the top edge of squares of paper over each box.
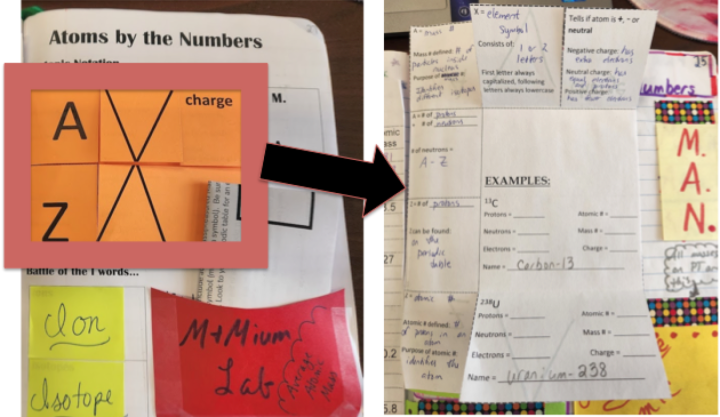
Next we fill in the isotopic notation foldable. I have always done this with direct instruction (as you can see in Video 1 of my Classroom View of Interactive Notebooks blog post). This year, my plan is to make this lesson more student-centered by having them examine side-by-side models for several atoms: the isotope notation, the corresponding periodic table tile, a Bohr model, and then statements of the atomic number, mass number and charge. Students would use the foldable as a guide for what information they should gather from the models. I presented this idea at BCCE in early August 2022, and was approached by a representative from POGIL, who told me there is a similar exercise that they have designed. I mention this to point out that there are a variety of ways for students to get the information that they will use to fill in the notebook components.
On the side of this page, I have students make a note of the M.A.N. memory aide for finding neutrons (Mass # - Atomic # = Neutrons). Underneath that, I have included a graphic of an example of calculating average atomic mass, but you can also have students paste in their own example. We do a PhET activity, Isotopes and Atomic Mass, to support understanding of average atomic mass and students also find the average atomic mass of M&Mium (an activity where I make baggies of mixed kinds of M&Ms, and students find the average atomic mass of this “element”). Because most of the learning of average atomic mass is done by activities outside of the notebook, this graphic is more of a reminder of the activity. If you wished, you could instead make a pocket here that could include a paper to go along with a class activity that students could tuck in for later reference.
Note: You can find an M&M activity that is very similar to mine published on ChemEd X: M&Ms as Isotopes.
Leftside – Protons, Neutrons and Electrons
Many teachers have some version of this activity that they use. This is the first time my students really do a deep dive into pulling information about atoms from the periodic table. I use the gradual release method (I Do, We Do, You Do) on the first 3-5 rows of the table and then students do the rest on their own and check with a partner. I also try to dispel a misconception on this page, which is the difference between mass number and atomic mass. I intentionally include several isotopes on this page, and I have students identify isotopes and group them by highlighting the sets in the same color.
I would suggest here that this page is an opportunity to do a blended notebook and incorporate some immediate, digital feedback. You could set up an online quiz on your LMS that matches this output page. After doing a certain number of problems (perhaps 10 lines), students could enter their answers and you can check their work. This would allow you to immediately and quickly find and correct existing misconceptions before they become ingrained in students’ routines for counting subatomic particles.
One final note, this output page has a large number of practice problems. I sometimes break this into day 1 and day 2, to spread out the practice and create deeper understanding with repetition. You could even break this large table into two (or more) parts and have students create layers in their notebooks.
If you are having any trouble visualizing what these pages will look like in action, check out the video below.
Video 1: Atomic Structure INB pages, ChemEd X Vimeo Channel, 8/17/2022.
Questions
Concepts
Time Required
Preparation
Provide copies of the following for each student. Note that I have grouped the components by page. Please preview them before printing as some pages have multiple copies to minimize paper waste.
Spread 1:
Spread 2:
Right side -
*Note about formatting of the printing for this page: Page 1 and 2 of Atoms by the numbers is the foldable. It should be printed front-to-back, flipping on the long edge. This will set up the foldable correctly once it’s cut and folded. Page 3 of the document is the page layout that gets pasted into the notebook.
If you are looking to limit the amount of paper you are using, you can omit the page layout from your printing, and simply use it as a template to display for students. Have them make the "I" words flaps out of Post-It notes or construction paper, and then write the M.A.N. memory aide on the page, and have them record an example on the bottom right page of the page for average atomic mass.
Attribution
*Webinars Referencing Interactive Notebooks: ChemEd X Chem Basics, October 2021 – Chemical Bonding and Naming and AACT , February 2020 – Teaching Chemistry in a Textbookless Classroom
**I have three PLC colleagues that have supported this work as we always review the pages and talk through content together. Many thanks to Tom Jankowski, Lori Young and Mihir Paranjape.
Comments
13Thank you!
So excited for my students to use an interactive notebook. Thank you for sharing all of your hard work.
In reply to Thank you! by Katiepabst
You're welcome!
You're very welcome! I love when people are excited about what I share.
Rubrics
Hi Nora
Thanks for sharing all your hard work. I am excited to try INBing again. Curious if have them glue the rubric in the notebook for each unit. I was thinking of doing that. Thanks,
In reply to Rubrics by Suzanne Irwin
Rubrics
I used to have them glue rubrics into their notebooks, yes. I would distribute them with the title page and have them glue it onto the leftside page of the unit title spread. Then during test day I would come around and complete the rubric. Sometimes I had them do a self-assessment first and then I would come with a highlighter and agree or change their score (in which case I would talk to them).
Now I don't do that anymore, but that's mainly because I changed my procedures during COVID and started having them submit photos of certain pages and spot check their notebooks in that way. Instead I use that first page for a list of learning objectives or we go back and put a study guide at the end of the unit.
There are lots of right ways to approach this - I hope that gives you some ideas to help you find what works for you!
One more question
Thanks for your response. One more question -- how do you create your pdfs? Do you use an Adobe program? I want to create my own/adapt some of yours and was wondering--especially for the docs where you rotated things to fit more on the page. Thanks!
In reply to One more question by Suzanne Irwin
Making templates
My favorite way to make templates is to make them in Word. It's easy to rotate images to fit more on the page. You do have to reason through rotating tables a bit more because you have to change the formatting of the table themselves and then change the direction of the text. I turn them into PDFs by telling Word to Print to PDF. Sometimes I find the easiest thing to do is create a table with normal orientation in word, use the Snipping tool to grab it as an image, and then paste it in and rotate it as an image.
If you have Adobe you should be able to turn my PDFs into docs pretty easily but you might lose some formatting. We are posting them as PDFs because we have learned they do not open the same on all computers so the formatting is lost unless they're posted as PDFs.
Hope that helps!
Average atomic mass foldable
I love your interactive notebooks and will be using them to support my notebook this year. I did notice that you have a spot on the Atoms by Number handout for calculating average atomic mass. Will you be sharing that resource in the future? Thanks for the time and effort you have put into these. They will be treasured and put to good use.
In reply to Average atomic mass foldable by Shatawn Tedesco
Calculating Average Atomic Mass
I'm so glad that you are getting good use out of them!
So, I put different things there depending on the year. This year I plan to just make a pocket out of a piece of paper, and have students tuck the M&Mium lab handout there. Other years I have had students paste or write in a worked out sample problem. You can certainly grab an image offline or create your own to create a paste-in.
MAN as acronym, confusing?
I am enjoying your INB pages, and I have been using them as a guide for my kiddos to make their notebooks. I wonder if with the big "A" on the nuclear symbol (mass number) if the kids would get confused that the A in the MAN acronym meant atomic number. Have you run into this?
Thanks again for sharing.
In reply to MAN as acronym, confusing? by Karen Collins1
MAN acronym
You know, that's an interesting point but no, I haven't had that happen. The students are usually ok with it. They are so used to acronyms being first letters of the words in other classes that I don't think they even stop to think about it being a symbol instead.
Share Your Thoughts