Chemistry Magnets - Making Chemistry Visible
This past July, I had the opportunity to present “Making Chemistry Visible With Magnets” at ChemEd 2019. Additionally, through the creativeness of some fellow attendees, particularly Katy Dornbos, Ariel Serkin, and Kristin Gregory, I gained some additional materials that have enhanced the presentation.
Editors' Note: See the recording of Doug Ragan's 2022 ChemEd X Talk related to this post.
In my presentation, I mentioned using 2-inch colored magnets purchased from FLINN Scientific to represent everything from subatomic particles to atoms for balancing equations, to showing molecular geometry and even representing the atoms of organic molecules by placing the colored circles on top of a superimposed structure of an organic molecule (figure 1).
Figure 1: Overlay of colored magnets on top of an organic structure.
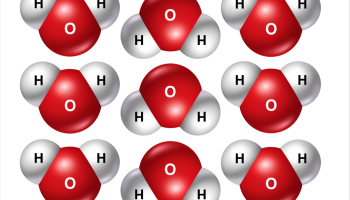
The magnets that I purchased are available in several colors, blue, red, green, yellow, and white. In order to get carbon and hydrogen, I had to print out 2-inch circles, laminate them, cut them out and tape them to some of the magnets. However, after arriving home from ChemEd, I received a Twitter notification with an alternate way of creating my magnets. A link was provided with beautiful images (*PDF files can be found in the supporting information with a complete list of other colored images that can also be printed) showing labeled carbon atoms, sodium and chloride ions, to name a few. There are also water molecules (figure 2). A big thank you goes to Katy Dornbos for putting the time into designing these. Katy also introduced me to printable magnetic sheets that can be fed into any inkjet printer allowing these images to be printed easily.
Figure 2: Examples images of the water molecules that can be printed on printable magnet sheets. (K.Dornbos)
I purchased five sheets of printable magnet paper for a reasonable price after a quick Amazon search. Using Katy’s images, I printed 9 water molecules, 15 Carbon atoms, 15 Oxygen atoms, 15 Hydrogen atoms, multiple bars representing single, double, and triple bonds, and multiple sodium and chlorine atoms and ions.
With the addition of these new images, I plan to ask my students why tap water bends toward a balloon after it has been rubbed on a students head. This idea comes with a common misconception and I hope to fix that by showing them what happens on the particulate level with my water molecules. Next, with the water molecules, my students and I will discuss why ice is less dense than water and then show the arrangement of the water molecules representing ice, water, and steam.
I have many other uses for the magnets and will continue to update followers about how I use my chemistry magnets. I would also love to hear from you regarding how you are using these magnets to enhance your lessons. Please share what has worked or what may not have worked. I would love to hear what your students think of using them. You can follow me on twitter @dragan39 and I will be using the hashtag #chemmagnets to post pictures, ideas, etc.
Editor's Note: This post has been updated with a few related tweets - 8/2/21
At @ChemEd2019 @dragan39 shared magnetic atoms.
I want magnetic molecules too, so:
5 printable magnetic sheets = $17
1 doc of water molecules, ions, atoms
120+ students to help cut them out
= many magnetic molecules (and atoms)!https://t.co/rj6oP51e4ahttps://t.co/rrJgU2I8oM— Katy Dornbos ??♀️ (@KatyDornbos) August 1, 2019
No more pencil drawings! Assessing student models with @Classkick. Then students ask can we use the magnets? Me: absolutely! #chemmagnets pic.twitter.com/k8gVtQyW5h
— Doug Ragan (@dragan39) October 17, 2019
In honor of ?? thanksgiving! It’s Tryptophan for #moleculemonday @aserkin #chemmagnets pic.twitter.com/bUCjrtbXbu
— Doug Ragan (@dragan39) October 15, 2019
Check out Ariel Serkin's post, Molecule Monday, for more ideas for highlighting specific molecules!
Today’s been kind of awful for a whole host of reasons, but I decided to get small magnetic whiteboards and printable magnet sheets to make some #chemmagnets for the Energizer lab and it made me really happy. pic.twitter.com/PrQUXUDRVO
— Heidi Park (@heidijpark) February 12, 2020
Check out our ChemEd X post Energizer Lab for more information about the activity and how to access other Target Inquiry activities. Heidi says that the cost "works out to about $4.55 per model ($3 for the whiteboard and $1.55 for the magnetic sheet)".
Make your own magnetic boards for Ss use #BestofChemEd @dragan39 pic.twitter.com/nSnQ2T60sG
— Kristin Gregory (@KristinGregory7) July 27, 2021
Doug presented about his magnets as part of Best of ChemEd hosted by AACT. Members of AACT can view the recording and download Doug's files HERE.