Energizer Lab with Virtual Options
NEW virtual options below!
In this electrochemistry lab, students proceed through a prior knowledge activity, practice creating and using a voltaic cell and use a model designed to simulate the particulate level activity within that cell. The teacher checks for student understanding at specific points as groups work together. A discussion follows to help clarify ideas. A student document can be found in the supporting information below.
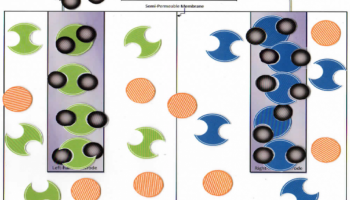
This lab was originally created and published as part of the second cohort of Target Inquiry at Grand Valley State University. Teachers can find the Teacher Document including the cell and particles that can be printed and distributed to groups of students (see figure 1). Those that don't have an account can register for one (for free) and gain access to over 80 activities for chemistry and other sciences. The Teacher Document also outlines common misconceptions that are addressed in the lab.
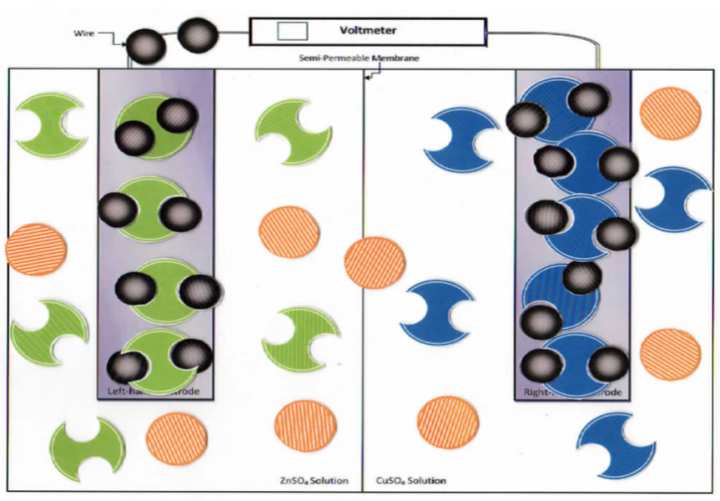
Figure 1: Students move the pieces on the in-class laminated paper model
Melissa Hemling created virtual options for manipulatives used in this lab and has generously agreed to allow ChemEd X to add them here. (Check out Melissa's ChemEd X Blog!)
This Google Slides link provides the cell and manipulative particles (see figure 2). The manipulative particles are stacked in the margins and students simply slide them onto the model.
Melissa also created a Classkick manipulative for teachers using that platform (Note that Classkick works best in the Chrome browser.)
Both of these options can be used with the original student document shared in the Supporting Information below. The teacher can demonstrate the prior knowledge activity and the working cell live or by creating a video as their situation allows. Alternatively, a quick search of YouTube or Vimeo yields a large assortment of videos that can be used.
Additionally, Kelly Burleson contributed her own modification of the Energizer Lab assignment in her ChemEd X blog post, Stop Motion Animation Galvanic Cell Process, published 8/12/19 that could be combined with the virtual manipulatives above.
Figure 2: Virtual manipulatives created in Google Slides by Melissa Hemling
Materials
- 0.10 M CuSO4
- 1.0 M CuSO4
- 1.0 M ZnSO4
- 2-10cm pieces of zinc wire
- 1-10cm copper wire
- test tube
- voltmeter
- electronic balance
- Voltaic Cell Kit OR you can set up a cell using a 400 mL beaker, a porous cup and alligator clip wires.
Background
In the prior knowledge section, students place a zinc paperclip in dilute copper II sulfate solution. After they let it sit for 5 minutes, they carefully remove it from the solution and observe. This provides an opportunity to revisit oxidation and reduction and check for understanding of those concepts before moving in to the lab.

Figure 3: Prior Knowledge activity prior to Energizer Lab procedure.
Procedure
Part A: Students may use beakers and a salt bridge or a Voltaic Cell Kit as seen here. I often have at least three different versions for student groups to choose from. If only one type is available, videos of other types, including microscale versions, are easy to find. Be sure that students unhook the voltmeter while the cell sits unattended to help ensure a maximum change in mass. Note: Notice that both half reactions from the Prior Knowledge Section are present. They are separated in this cell so that there can be no direct interaction between zinc atoms and Cu2+ ions. The porous cup keeps the Cu2+ ions from coming into contact with the zinc strip.
Part B: Check to make sure students have set up the model per the diagram provided in the student document before they work through the procedure. While students work through the model, check for understanding of each group. Get students to say that the Zn2+ goes into solution. Ask them what happens at this point to keep the charge within each cell neutral. What happens at the cathode with the electron? Have students explain the electron flow, ion flow through the semi-permeable membrane, and cation/ anion change at the electrodes. Initial Check Point at end of Part B.
Part C: Students should observe the electrode. If students have trouble seeing a mass change within the lab period, keep one cell running for a longer time period (several hours possibly) until a change in size can be seen and then show to students the following day. It can be difficult to measure the actual mass but students only need to visually see that one electrode gains mass and the other losses mass. The mass of the zinc electrode should decrease and the mass of the copper electrode should increase. The zinc electrode may seem dull because it is losing zinc atoms. The copper electrode may look shiny because it is gaining copper atoms.
See the Student Version provided in the Supporting Information for the Complete Procedure.
Questions
See the Student Document provided in the Supporting Information for associated questions as they are found throughout the document.
Concepts
- The student will draw and label the parts of an electrochemical cell.
- The student will identify and explain the reactions occurring at the anode and cathode.
- The student will explain the flow of electrons in relation to the cell and the resulting voltage.
- The student will explain the movement of cations and anions in the cell.
Time Required
About one 60 minute class period for lab and manipulative procedure.
Preparation
Prep cupric sulfate and zinc sulfate solutions. Set up voltaic cell kits or alternative equipment.
Attribution
The model in this lab is adapted from the following article:
Huddle, Penelope Ann and White, Margaret Dawn, Using a Teaching Model to Correct Known Misconceptions in Electrochemistry, Journal of Chemical Education, 2000, 77 (1), p. 104- 110.
Research related to the Energizer Lab activity is discussed in the following article:
Cullen, Deanna M. and Pentecost, Thomas C., Model Approach to the Electrochemical Cell: An Inquiry Activity, Journal of Chemical Education, 2011, 88 (11), 1562-1564.
Credits
General
Items tagged with the safety:general tag have some safety concerns that may be also described more specifically by additional safety terms
General Safety
For Laboratory Work: Please refer to the ACS Guidelines for Chemical Laboratory Safety in Secondary Schools (2016).
For Demonstrations: Please refer to the ACS Division of Chemical Education Safety Guidelines for Chemical Demonstrations.
Other Safety resources
RAMP: Recognize hazards; Assess the risks of hazards; Minimize the risks of hazards; Prepare for emergencies


Comments
5Question on Part A
Deanna - this lab is great. I really like the virtual options. One quesiton about part A...Why is it necessary to unhook the voltmeter to ensure a maximum change in mass? Why wouldn't the mass change with the volt meter connected?
In reply to Question on Part A by Stephen Pulliam
Voltmeter
Thank you! Some teachers weren't seeing a mass change so I asked a mentor. I was told that the resistance in the voltmeter could slow down the chemical reaction so my advice is based on their direction.
I appreciate Melissa's virtual options too!
Share Your Thoughts