Using My Marbles: Connecting Surface Area and Calorimetry with Temperature Curves and Thermochromic Cups
by Dean J. Campbell*
*Bradley University, Peoria, Illinois
Adding solids at a known temperature to water at a different known temperature is the basis of some classic calorimetry experiments.1 In these experiments, the temperature of the solid and of the water converge on some intermediate temperature, and the differences in temperature are combined with some given information such as mass and specific heat to obtain a missing quantity, such as mass or specific heat. These calorimetry experiments rely on stable initial and final temperatures. However, if the temperatures of the components can be monitored as a function of time, the rates of heating or cooling can be explored. The size and/or shape of the solids influences their surface area, and solids with greater surface area have faster heat exchange with water. The simple calorimetry experiment modifications described here are designed to get students thinking about the importance of surface area, in this case, on the rates of temperature change. The rate of temperature change can in turn influence the timing of a chemical process such as a thermochromic color change. Increasing the surface area of reactants or catalysts provides more locations for reactions at surfaces, yielding faster reactions.
In these experiments, samples of glass or iron spheres of uniform sizes were either heated or cooled and then added to water near room temperature. The temperature of the water was measured with a Vernier LabQuest 2 and thermometer at 0.1 second intervals. For the heating experiments, the spheres were heated in a drying oven for at least two hours in polystyrene foam cups. When the cups were removed from the oven, the thermometer inside read about 74 to 71 °C. The spheres were added to samples of about 49 g of water at room temperature (21 °C in that lab) in polystyrene foam cups. For the cooling experiments, the spheres were cooled in a freezer overnight in polystyrene foam cups. When the cups were removed from the freezer, the thermometer inside read about -13 to -8 °C. The spheres were added to samples of about 49 g of water at room temperature (18 °C in that lab) in polystyrene foam cups.
For these experiments, it is desirable to clearly detect differences in the rate of temperature change as sphere sizes change. Materials with higher specific heat produce temperature curves of the surroundings (e.g., water) that have larger changes in the direction of the temperature axis. Materials with lower thermal conductivity more slowly transfer heat between the solid and the surroundings, so it will be easier to detect variations in the rate of temperature change in the direction of the time axis of a temperature curve. The exact compositions of the glass (probably soda-lime glass) and iron or steel in the spheres used in these experiments are not known. The literature shows variation in these values, but there is general agreement that glass has a higher specific heat than iron and conducts heat much more slowly than iron.1 One source lists soda-lime glass as having a specific heat of 0.88 J/g K and a thermal conductivity of 0.937 W/m K2. Another source lists iron as having a specific heat of 0.412 J/g K and a thermal conductivity of 80 W/m K.3
Approximately 30 g of glass spheres were used in each trial. The larger glass spheres were marbles with an average diameter of 1.58cm and a total surface area of 47 cm2. Six were used for each trial. The smaller glass spheres had an average diameter of 0.49 cm and a total surface area of 142 cm2. About 190 were used for each trial. Figure 1 shows the results of twelve trials with glass spheres, with three trials of each combination of heating or cooling with large or small spheres. The temperature curves were all shifted along the x-axis to all begin changing at the same time. Clearly, heat exchange with the water is faster for samples comprised of many small spheres rather than a few large spheres. It is important to note that thermal curves produced by samples of the same composition and mass had the same initial and final temperatures, independent of sphere size. This means that regardless of the rate of heat transfer, the total quantity of heat transferred (what we focus on in calorimetry measurements) did not change.
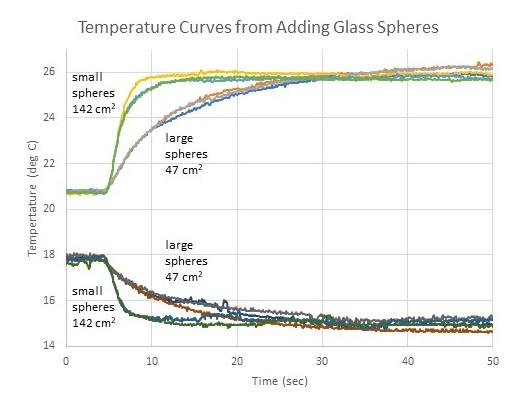
Figure 1. Temperature curves of 50 g of water with the addition of 30 g of glass spheres. Variations include heating or cooling with large or small spheres.
Approximately 33 g of iron spheres were used in each trial. The iron-based spheres were all deliberately rusted for use in other experiments, using an aqueous solution of sodium chloride, acetic acid, and hydrogen peroxide.4 Although rust is probably not necessary for these experiments, iron tends to rust over time in contact with water and Earth’s oxidizing atmosphere. The larger iron-based spheres had an average diameter of 1.25 cm and a total surface area of 20 cm2. Four were used for each trial. The smaller iron-based spheres were made from BB gun ammo that had their copper cladding removed by abrasion with sand in a rock tumbler. These spheres had an average diameter of 0.43 cm and a total surface area of 58 cm2. About 100 were used for each trial. Figure 2 shows the results of twelve trials with iron spheres, with three trials of each combination of heating or cooling with large or small spheres. The temperature curves were all shifted along the x-axis to all begin changing at the same time. Heat exchange with the water is still faster for samples comprised of many small spheres rather than a few large spheres, but the difference is harder to see than when glass was used. This is likely because iron’s greater thermal conductivity causes the temperatures to change faster and the resulting temperature curves are less distinguishable from one another under these experimental conditions. It should also be noted that a small amount of iron oxide comes off of the rusty spheres into the water, turning it reddish. As with the glass spheres, thermal curves produced by samples of the same composition and mass had the same initial and final temperatures, independent of sphere size.
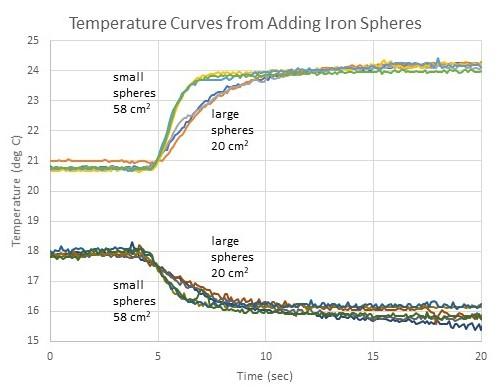
Figure 2. Temperature curves of 50 g of water with the addition of 33 g of iron spheres. Variations include heating or cooling with large or small spheres.
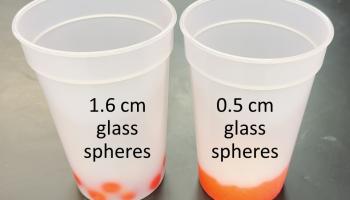
To demonstrate the relationship between surface area and heat transfer to an audience, one could display the temperature curves collected by a LabQuest module or similar device. Alternative demonstrations used glass spheres and a pair of thermochromic beverage cups, see Figure 3. The cups were designed to change colors, such as from colorless to red, when cool liquids were added. They likely used leuco dye chemistry, similar to that used by thermochromic paper.5 About 93 g of larger 1.58 cm glass spheres were added to the left side cup, and about 93 g of smaller 0.49 cm glass spheres were added to the right side cup, Figure 3 TOP. In one demonstration, the glass spheres were cooled in a freezer. The colorless cups at room temperature were placed upright on a surface and the cold glass spheres were added. For the cup at the left side of the images in Figure 3 MIDDLE, red spots appeared that corresponded to where the larger spheres touched and cooled the interior surface of the cup, causing the thermochromic reaction to take place. Because the spheres were larger, the relatively few points of contact between their surfaces and the cup interior surface were sufficiently far apart as to make the cooling spots visually distinguishable. For the cup at the right side of the images in Figure 3 MIDDLE, so many more red spots appeared that they became indistinguishable more quickly. The greater surface area of contact between the many smaller spheres and the cup interior caused the entire bottom of the cup to change color from the thermochromic reaction more quickly than when the larger spheres were added. Video 1 shows the demonstration described by Figure 3 MIDDLE.
Another demonstration essentially crosses the color transition in the other direction. The thermochromic cups were first cooled in the freezer to change them from colorless to red and then placed upright on a surface. This time, glass spheres were added that had been heated in a drying oven. For the cup at the left side of the images in Figure 3 BOTTOM, colorless spots appeared that corresponded to where the larger spheres touched and cooled the interior surface of the cup. For the cup at the right side of the images in Figure 3 BOTTOM, so many more colorless spots appeared that they became indistinguishable more quickly. Video 2 shows the demonstration described by Figure 3 BOTTOM.

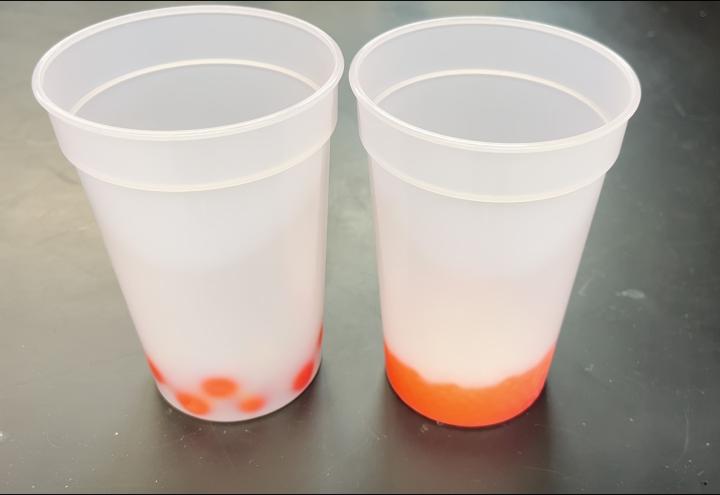

Figure 3. (TOP) ~93 g of cold 1.58 cm glass spheres added to the left thermochromic cup and ~93 g of 0.49 cm glass spheres added to the right thermochromic cup. (MIDDLE) Glass spheres removed from a freezer and added to room temperature thermochromic plastic cups to turn them from colorless to red. (BOTTOM) Glass spheres removed from a drying oven and added to cold thermochromic plastic cups to turn them from red to colorless.
Video 1: 93 g of cold 1.6 cm (left cup) and 0.5 cm (right cup) glass spheres removed from a freezer and added to colorless thermochromic plastic cups. The many smaller spheres turn more area of the colorless cup to red faster than the fewer larger spheres, because the many smaller spheres make greater contact with the interior of the plastic cup. The spheres themselves are shown at the end of the video. Note that there is also a similar video where warm spheres change the color of cold thermochromic cups. ChemDemos YouTube Channel (accessed 7/25/2022)
Video 2: 93 g of 1.6 cm (left cup) and 0.5 cm (right cup) glass spheres removed from a drying oven and added to colorless thermochromic plastic cups that have been turned red in a freezer. The many smaller spheres turn more area of the red cup to colorless faster than the fewer larger spheres, because the many smaller spheres make greater contact with the interior of the plastic cup. The spheres themselves are shown at the end of the video. ChemDemos YouTube Channel (accessed 7/25/2022)
The “greenness” of these demonstrations can be considered from the perspective of the Twelve Principles of Green Chemistry.6 The greenest aspect of these activities are their reusability. Production of metallic iron and glass are energy-intensive processes, but these materials can be used repeatedly for these and potentially other activities. The plastic thermochromic cups are likely sourced from non-renewable petrochemicals, however, they can also be used repeatedly. Even the thermochromic reactions inside the cups are reversible. From a safety standpoint, glass spheres can potentially crack due to physical or thermal shock, but no other glassware was used, so there were no issues with hard solid spheres accidentally cracking glass containers.
Energy usage can also be considered. The calorimetry labs at this University have traditionally used boiling water baths over Bunsen burners or hot pates to heat metal samples in test tubes before adding the metal samples to water. The activities described here heated samples using an oven or cooled them using a freezer. It would be an interesting study to figure out which temperature-changing process (oven, hot-plate, Bunsen burner, or freezer) would be most-energy efficient and produce the least amount of greenhouse gases. The results of those studies could very much depend on the number of experimental trials run, the number of students running the experiments, or even how much the oven or freezer is open or closed during its use. Regardless of the process used to heat or cool the solids, thermal curves can be added to calorimetry experiments to introduce the importance of surface area in processes such as heat transfer. The thermochromic cups use chemistry to provide simple but very visible illustrations of surface area concepts.
Safety Goggles should be used when working with the activities. Glass spheres can potentially crack due to physical or thermal shock. Always wash your hands after working on laboratory activities.
Acknowledgements This work was supported by Bradley University and the Mund-Lagowski Department of Chemistry and Biochemistry with additional support from the Illinois Heartland Section of the American Chemical Society, the Bradley University BEST Program, and Beyond Benign. The material contained in this document is based upon work supported by a National Aeronautics and Space Administration (NASA) grant or cooperative agreement. Any opinions, findings, conclusions, or recommendations expressed in this material are those of the author and do not necessarily reflect the views of NASA. This work was supported through a NASA grant awarded to the Illinois/NASA Space Grant Consortium.
References
- Ellis, A. B.; Geselbracht, M. J.; Johnson, B. J.; Lisensky, G. C.; Robinson, W. R. Teaching General Chemistry: A Materials Science Companion, 1st ed.; Oxford University Press: Oxford, 1993.
- Continental Trade. Soda-lime glass properties. https://www.continentaltrade.com.pl/soda-lime-glass, there is an excellent data sheet available for download here. (accessed July 22, 2022).
- Nuclear Power. Iron – Specific Heat, Latent Heat of Fusion, Latent Heat of Vaporization. https://www.nuclear-power.com/iron-specific-heat-latent-heat-vaporization-fusion/ (accessed July 22, 2022).
- MrDiyDork. How to rust metal in minutes! https://www.youtube.com/watch?v=RjAPyFQGYp4 (accessed July 22, 2022).
- Campbell, D. J.; Lojpur, B.; Liu, R. “Thermal Paper as a Polarity and Acidity Detector.” ChemEd Exchange. October 28, 2021. https://www.chemedx.org/blog/thermal-paper-polarity-and-acidity-detector (accessed July 23, 2022).
- American Chemical Society. 12 Principles of Green Chemistry. https://www.acs.org/content/acs/en/greenchemistry/principles/12-principles-of-green-chemistry.html (accessed July 23, 2022).