Science of Matter Interactive Notebook

Once the school year has gotten into full swing, and procedures in lab and with interactive notebooks (INBs) have been established, I begin the chemistry content in my class with learning about matter and how we describe it. This unit is a great way to continue to introduce the notebooking concept to students and teach them different kinds of components that will be used throughout the year. There is a lot of flexibility and opportunity in this first unit for both input and output. As always, I hope you can use my materials as a starting point and edit them to work in your classroom and with your standards. Keep in mind that, when possible, it is helpful to give the students opportunity for creative expression and personalization so that the content in the notebook is expressed in a way that helps them to make connections and feel ownership over the material.
I also use this unit to start to introduce some inquiry/exploration on the students part when we begin pages. We have a focus in my school on Students Do The Thinking, and this unit (which is mostly familiar to students from middle school) is a great place to start to lead students toward higher order thinking skills. I talk about how I facilitate this with my properties of matter introduction activity and my physical and chemical changes lab below.
One thing I should note about INBs: not all of your students’ work will go into the notebook. Think of the INB as the content reference. What goes into the notebook is really up to you. I find that what helps me in making these decisions is thinking about what I want the students to see again when they are reviewing for the unit test and the semester exam. You will also have assignments, labs and activities that students do not complete in the notebook.
Materials
Composition notebooks, printed copies of each page, colored paper or construction paper for making flipbooks/foldables, scissors, tape or glue
**A note on my manipulatives: If you see a dashed line, that is where the item should be cut. If you see dotted lines, that means fold.
Background
For backstory about how Nora uses interactive notebooks, view a recording of her ChemEd X Talk: Integrating Interactive Notebooks into Chemistry Courses with Nora Walsh and read her previous post, Interactive Notebook Unit on Stoichiometry. Nora wrote the following in response to questions she received after her ChemEd X Talk: A Classroom View of Using Interactive Notebook Pages.
Nora's interactive notebook units published so far: Scientific Reasoning, Science of Matter, Atomic Structure, Periodic Table, Bonding, Stoichiometry, Gas Laws, and Thermochemistry. She plans to publish all of her units by the end of 2022.
Procedure
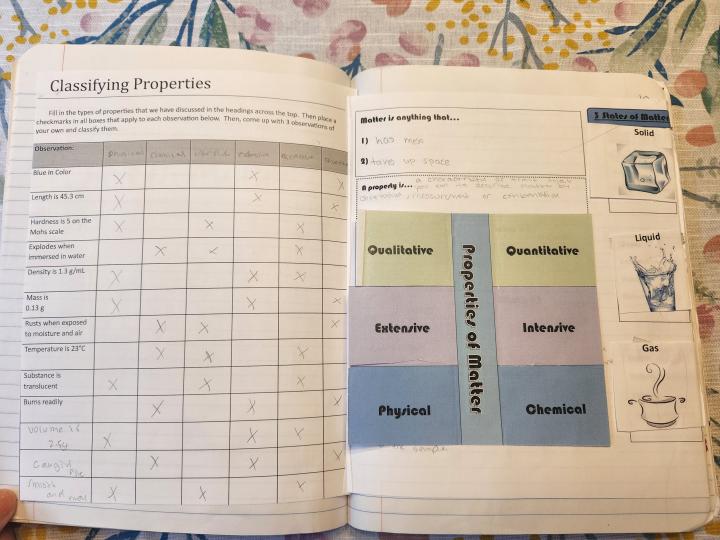
Spread 1 – Describing Matter
Right Side – Describing Matter
There are many teaching strategies that could be used to complete this page. Last year, I created a simple activity that provided an inquiry approach to the ways we describe matter. We began the class with a reminder of the two requirements for a substance to be considered matter, which students were able to do quickly using choral response as this is a definition they have used in previous science classes. We recorded this at the top of the INB page. Then I assigned students a partner and each pair was given an unknown substance. All substances were household items – sugar, borax, salt, crushed chalk, etc. – and students were asked to describe their substance in as many ways as they could. They were given some guiding requirements:
- They had to have at least 5 descriptions
- They had to use:
- At least 1 number
- At least 1 color
- At least one way to change the substance (and what happens after that change)
- They had to use at least 1 instrument to obtain a measurement (triple beam balances, metersticks, thermometers and graduated cylinders were provided)
After allowing some time to make these observations, I then directed pairs to combine with another pair and work to sort their pooled observations into categories of their choosing (an open sort). They were told to be prepared to the defend their choices. Then these groups combined with another group of 4 (to make groups of 8) and they again pooled and categorized their observations. This time they were asked to assign titles to their categories. Then we had a class discussion. Through the process of sharing as a whole class I guided them to make groups that were labeled with our 6 terms.
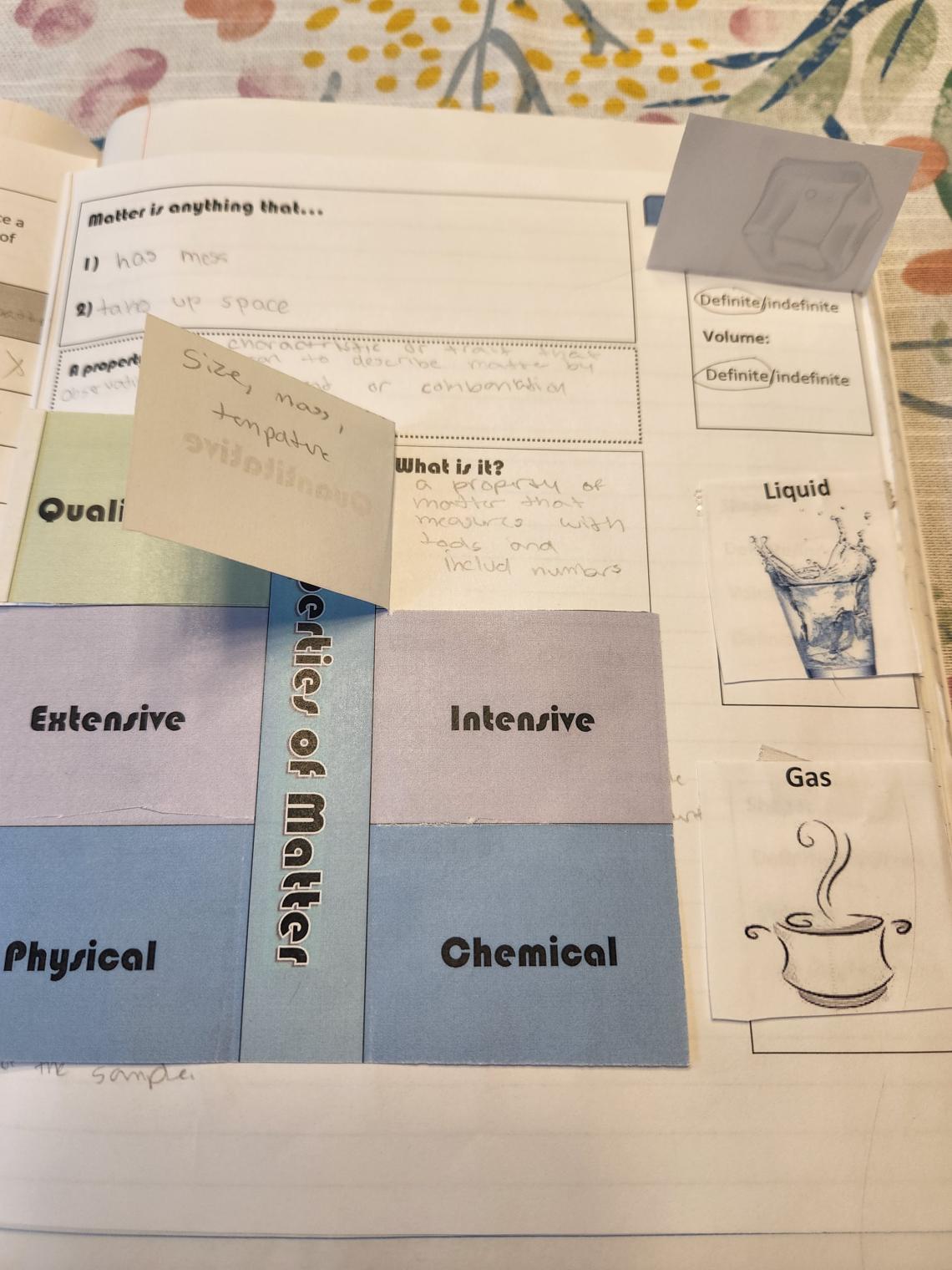
After walking through this activity, we then proceeded to fill in the definition of property box and the foldable on properties of matter.
Finally, down the side of this page are the common three states of matter under flaps, with students describing the shape and volume of each as definite or indefinite. Students do this while I do a very simple demo of pouring ice and liquid water into different containers, and then show them a balloon. You could modify these flaps to be more student-created by having them sketch images on the front. I also have them draw a particle view diagram of each state of matter under the flaps while examining the PhET States of Matter: Basics simulation.
In terms of pacing, I found that this spread takes me two class days: one to do the inquiry activity and fill in the properties of matter information/foldable, and a second day to do the states of matter demo/flaps and the output.
Left Side – Classifying Properties
A variety of activities would work here to have students practice classifying properties. I have created a teacher guided activity in which students label the columns at the top of a table with the different property descriptions, and then use checkmarks to classify each description in the rows. At the bottom of the page, students add on three of their own descriptions and classify them. One way to do this would be have them switch materials with another group and come up with new descriptions. Alternatively, you could distribute a blank version of this page and “crowd source” descriptions from the activity that students use to fill in the rows and then continue with the classifying as described above.
Spread 2 – Physical and Chemical Changes
Right side – Changes and Reactions
This input page is simply a barn-door foldable that also contains a cut-and-paste activity. The top two flaps are the typical side-by-side comparison of physical and chemical changes. Inside the foldable students include a description and then cut and paste several images onto the side flaps that depict examples of changes. (I used the excellent infographics from ThoughtCo as my image sources, and used a drawing program to snip and mix up parts of the images so that students had to evaluate the images to determine if they were physical or chemical changes.) Then, students work either in small groups or pairs to brainstorm 3 additional examples for physical and chemical changes beyond those that are depicted in the cut and paste.
At the bottom of the foldable are flaps for indications of a chemical change and driving forces of a chemical reaction. By having these flaps side-by-side, I’m attempting to head off a common misconception among students. I find that students often struggle to differentiate between indications and driving forces – they either think they are interchangeable terms, or when asked to state the driving force they instead cite the indication of change.
I first introduce the driving forces of chemical reactions, and I like to include a demo for each. We discuss observations during the demos and then after this is finished the students open the flap for the indications. I have included pictures on that side (in part to support students who are virtually learning and may miss the demos) and students label each picture with the indication. On the driving force side of the foldable, they denote the driving force to which each indication points. By having students make side-by-side correlations between indications and driving forces, they more clearly see how these two terms are related but different.
Left side – Physical and Chemical Changes Lab
The output for this spread is a lab. We do stations at our school and while this lab is not necessarily an inquiry-based lab, the students are provided options which leads to increased interest and thoughtfulness. We find that when we require students to make a choice in lab they take time to think through and discuss with their partner the outcome because they want to choose the “cool” station. In this subtle way we are having them make predictions about what will happen which engages higher order thinking.
This lab (like all of our labs) has evolved over time at our school, and we review and tweak it each year to meet our standards and our learning outcomes. The current iteration of the lab is included below. Please note that the first two pages are the student handout. The remaining pages are the directions for the stations – we print out the directions and post them at the individual stations.
Spread 3 – Pure Substances and Mixtures
Rightside – Pure Substances and Mixtures
On this page, students complete a matchbook foldable comparing the terms atom, molecule, element, compound, homogeneous and heterogeneous. A matchbook foldable works well here because the side fold allows the division between mixture and pure substance to be clearly demonstrated. Students include the definition, as well as examples, for each term. We included particle view diagrams to show the terms, but you could certainly have your students create these themselves. We had already done a POGIL activity that had students examine particle view diagrams, so we didn’t feel that we needed to do more than that. We had students utilize an online text (from ChemLibre) to fill in this foldable. You can see video of me introducing this foldable in my class in Video 1 below.
Video 1: Filling in Pure Substances and Mixtures foldable, originally published in A Classroom View of Using Interactive Notebook Pages, accessed July 26, 2022.
Leftside – Matter Flowchart
For this output I have students create a matter flowchart, and require students to use all of the terms from the matchbook foldable. I leave it very open ended, but ask students to clearly show how the terms are related to each other.
If you are having any trouble visualizing what these pages will look like in action, check out the video below.
Video 2: Science of Matter INB pages, ChemEd X Vimeo Channel, 7/25/2022.
Questions
Concepts
Time Required
Preparation
Provide copies of the following for each student. Note that I have grouped the components by page. Please preview them before printing as some pages have multiple copies to minimize paper waste.
Spread 1:
Spread 2:
Physical & Chemical Changes (Please note, for spread 2 the first two pages should be printed front-to-back with one paper per student. The third page is the images for the cut-and-paste and there are 4 sets per page to reduce the amount of paper required.)
Physical / Chemical Changes Lab with Instructions (Please note that the first two pages are the student handout. The remaining pages are the directions for the stations – we print out the directions and post them at the individual stations.)
Spread 3:
Attribution
*Webinars Referencing Interactive Notebooks: ChemEd X Chem Basics, October 2021 – Chemical Bonding and Naming and AACT , February 2020 – Teaching Chemistry in a Textbookless Classroom
**I have three PLC colleagues that have supported this work as we always review the pages and talk through content together. Many thanks to Tom Jankowski, Lori Young and Mihir Paranjape.


Comments
7Physical and Chemical Changes
Hi Nora,
I was wondering if you could tell me which demos you use when talking about indicators and driving forces. I can kind of see but not fully. Thanks Nora. Loving my INBs !
In reply to Physical and Chemical Changes by Rathsas
Driving Forces Demos
Oh, I'm so glad! I use demos like the following - there are lots of options - and this year I didn't do demos, I had them talk about the results they saw from doing the Physical and Chemical Changes lab first.
-Some kind of very obvious precipitation (like lead(II) nitrate and potassium iodide) - formation of solid, color change
-Something that bubbles and forms a gas (mossy zinc in HCl perhaps) - formation of gas, bubbles
-some kind of redox, like putting a penny into acid and seeing the acid change colors - transfer of electrons
-formation of water is harder to "show" - I sometimes show an acid-base reaction with an indicator - students often have experience from middle school that allows them to understand a little bit about pH indicators
Slides for your units
Do you have slide decks you use for these units?
In reply to Slides for your units by Nicole Martin1
Slide Decks
I make slide decks to show the students how to assemble the notebooks, and I often have them do the construction on test days before the next unit begins. I don't use slide decks very often in my lessons - I have them learn the content in other ways (activities, videos, card sorts, etc).
Share Your Thoughts