One teacher's attempt to use BCA Tables for Stoichiometry

There has been a fair amount of discussion on ChemEd X within the last two years about how to teach stoichiometry. (See Stoichiometry is Easy, Conceptual Chemistry, and Rethinking Stoichiometry.) I've been teaching chemistry for 22 years now, and while I've made small changes to my method of teaching stoichiometry, I haven't tried anything drastic, until this year. Inspired by the discussion going on here about stoichiometry, I decided to give BCA Tables (a method borrowed from the "Modeling Instruction" movement) a go with my introductory Chemistry class.
I have traditionally taught stoichiometry using what is commonly called the "algorithm method" of using conversions. I have a PowerPoint (attached below) and a Notes Handout (also attached below) for students. This takes them through the concept of what the balanced equation represents first. So on some level, I was attempting to go beyond the mere algorithm with my introduction to stoichiometry. Then I take them through a couple problems to practice the newly found skill of using the balanced equation as a conversion.
After the notes, I give them some practice problems (also attached below) and wander around helping students as they work. Any problems not finished are completed as homework. I give students access to a set of answers (attached below) and a set of worked solutions (attached below) so they can check their work as they go - even from home.
Following this pen-and-pencil attempt at stoichiometry, I run a stoichiometry lab the following class. I've typically used a variation of the lab provided by David in Stoichiometry is Easy but have struggled with this as I had the students evaporate the water to actually SEE the NaCl that is made. This has never given good results. Last fall I searched for an easier one-day lab that wouldn't require the same level of water evaporation. By asking #ChemChat on Twitter, I discovered an alternative: Using the thermal decomposition of calcium carbonate. (This is where I should give credit to the person that gave me this idea. I'd love to do this, but I couldn't find the conversation in a search of my history. Sorry!)
The reaction itself has an interesting color change, from the greenish copper II carbonate basic to a very stark black of copper II oxide. If you'd like to see the reaction, you can watch a video clip from a student lab last fall posted below. The lab handout requires students to go step-by-step through the process of stoichiometry - with the goal that they see the possibilities. Ideally, they will get close to 100% yield (most groups in the fall were in the 90s) and say something like, "Wow, we predicted 3.2 grams of product, and there IS 3.2 grams of product."
So this spring, I basically did all of this the same. However, I had some extra time and a group of students keen to discuss a different way of looking at stoichiometry. Attached below you'll find the PowerPoint I used along with the notes handout I created. Some of the material for the BCA notes and PowerPoint came from the previous blog posts mentioned above. I deliberately used the same sample problem as the original "traditional" set of notes so the students could focus on the process rather than the outcome. I also went beyond what I've seen with the typical BCA process by adding a bit of emphasis to the conversion to and from grams.
After my notes, I polled my students (anonymously with a Google Form) using the following questions:
Question 1
On a scale from 1-5, how confident do you feel in your ability to use the “algorithm” method we’ve already learned. (5 is most confident, 1 is not confident at all)
Question 2
Justify your answer to question 1. In other words, explain your rating. What do you like about the “algorithm” method? What confuses you about the method?
Question 3
On a scale from 1-5, how confident do you feel in your ability to use the BCA Table method we’ve just learned. (5 is most confident, 1 is not confident at all)
Question 4
Justify your answer to question 3. In other words, explain your rating. Did the BCA Table method help you understand the concepts behind stoichiometry calculations? What do you like about the BCA method? What confuses you about the method? Do you plan to use it again or stick with the “algorithm” method?
While I recognize that student opinion data isn't as valuable as the results from assessments, I do like to ask my students for feedback to see how they are processing things and where I can help. Below are some of the results and discussion of the survey results.
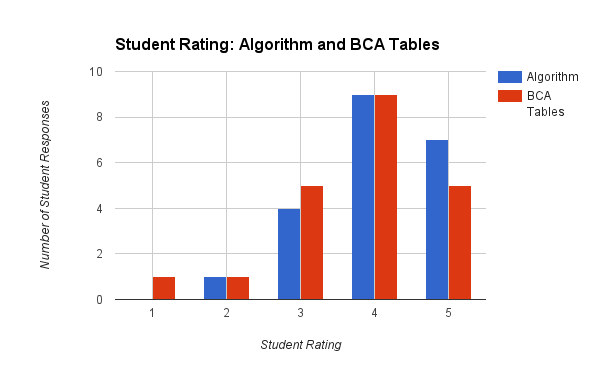
The average rating for the algorithm method was 4.05, while the average rating for the BCA Table method was 3.76. With only one class of 21 students, I certainly didn't run a statistical analysis to see if there is significance to this difference. Overall students had general confidence in both methods - and this was certainly evident based on one formative assessment the day after the notes on BCA tables. Interestingly enough, only two of the 21 students actually used BCA tables on the quiz. Most students simply used the algorithm method.
When comparing the ratings of the two methods, the numbers bear out (qualitatively) that more than half the students had either the same or increased confidence in using the BCA Table method. However, 9/21 is a relatively large number of students that didn't have as much confidence in using the BCA Table method.
Student Confidence in BCA Tables compared to Algorithm Method |
Number of Student Responses |
Decreased Confidence in Method |
9 |
Confidence in Method Stayed the Same |
7 |
Increased Confidence in Method |
5 |
The main reason for this decreased confidence may not have been the method itself, but the lack of practice. To be fair to the discussion, it is true that I didn't have as much time to dedicate to this as I would have liked for a full comparison. For many of the students that had decreased confidence in the BCA Table method, a lack of practice was mentioned as a reason. The following comment is indicative of these comments, "I understand the concept and ideas behind it, but I think I need more practice using the concept to get it right all the time and feel confident about it."
For students with confidence in both methods, two comments stood out:
"This is also confident for me because this is using concept of stoichiometry. I like that in BCA, we can find every mole of reactant and product as reaction occurs. I think I can both use BCA and algorithm method but I think BCA show me better understanding of stoichiometry while algorithm method is used for faster conversion."
"I understand the BCA method in full, but compared to the other way of solving, it is a lot more time consuming which could be helpful if one was looking at a more in depth analysis as why the problems are solved as they are."
While admitting that my use - and teaching - of the BCA Tables method is limited to this lesson, I do think the algorithm method is more efficient. However, the BCA Table method can lead to a better conceptual understanding of how stoichiometry works.
For the students that had increased confidence in the method, they generally thought BCA Tables provided a better understanding. One student said, "It is a simple method that allows us to see the process and it helps to give a clearer understanding of Stoichiometry."
This is certainly not intended to be a complete discussion of which method is "better." My sample size is 21 students, with one small attempt at using BCA tables. I also think that as teachers we should be open to some methods being more usable by some students, while other methods will work better for other students.
Having seen them in action now, I can certainly see the benefit on a few levels. While I don't think the algorithm method prevents conceptual understanding, it can often be done by students that don't really "get" the underlying reason the method works. The BCA Table method does seem to provide a more solid understanding of the mole relationships of the reaction in question. I can also see real benefit to BCA Tables for limiting reagents and equilibrium problems.
Update: After I'd written my first draft of this blog post, I was reviewing the making of buffers to my IB Chemistry students and found another scenario where BCA tables might prove helpful: Explaining why titrating 1/2 the amount of a weak acid with a strong base can give a suitable acidic buffer. The picture below shows my whiteboard from class - where a "de facto" BCA table is being used.
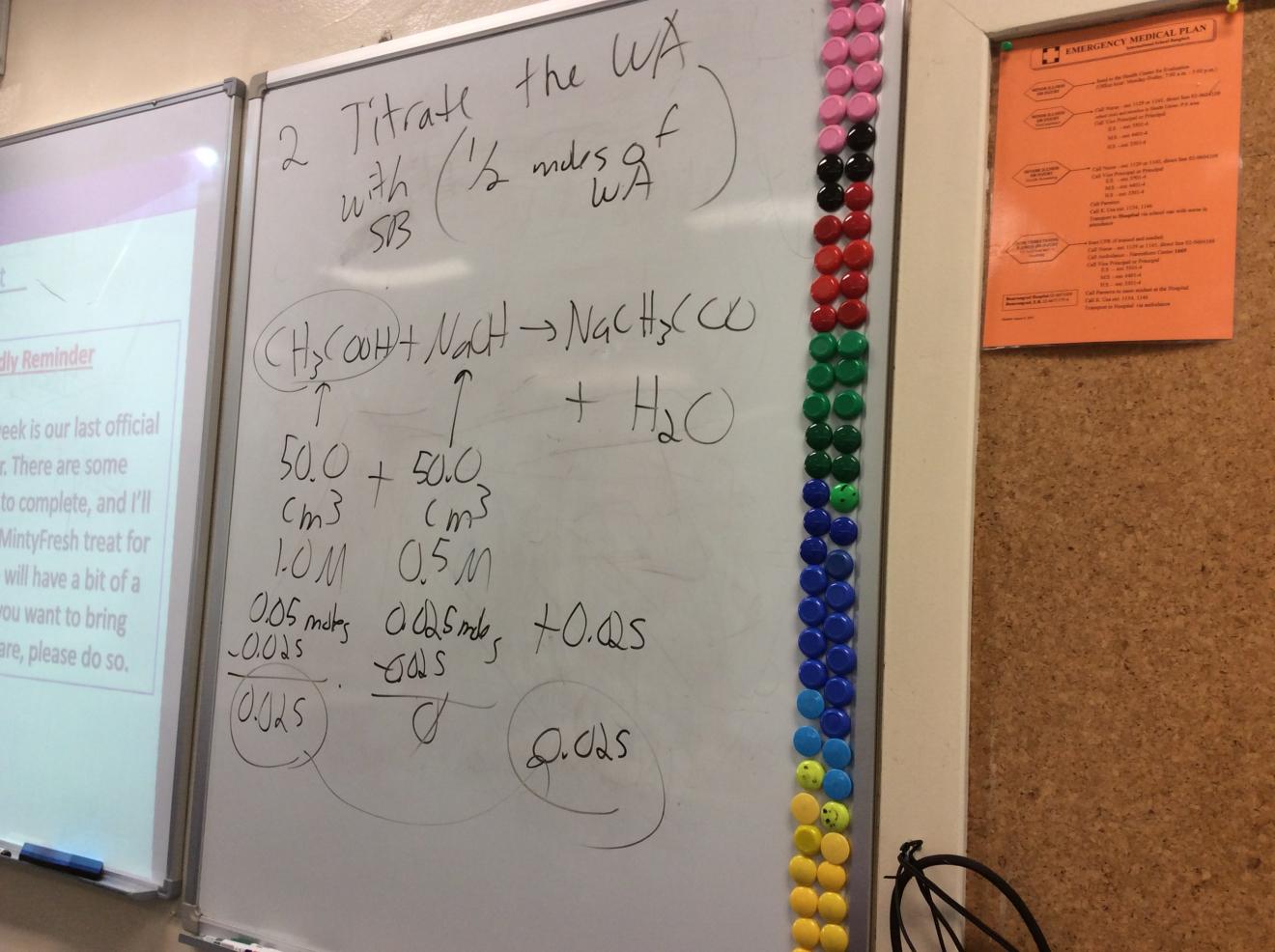
I will likely try BCA Tables again. I have not yet decided if I want to go all in with this method or not. But adding more tools to my teaching toolkit is always something I enjoy.
For those of you that use BCA Tables, I do have a question: How do you tie in the grams to moles conversion? Do you use "typical" dimensional analysis/conversions? And how do you teach students to use the mole ratios to complete the "C" row of the table? I found that students struggled with the ratio a bit (and maybe this means I struggled to teach it correctly) in trying to decide 2/3 or 3/2 for a ratio, for example. How do you address this?
Comments
9BCA is great!
I replaced my traditional method of teaching stoichiometry with more of a modeling-based approach this year in my regular chem classes. This included the use of BCA tables. Prior to introducing the BCA tables my students had extensive experience with the conceptual meaning of a balanced equation as well as a strong background in the connection between mass and conversions (including mole conversions). My students used either a proportion method or a factor-label method to convert between grams and moles, whichever made the most sense to them. So, we didn't explore BCA tables until we had built the idea that there's a connection between the counts of reactants and counts of products (Much thanks to the Argument Driven Inquiry book by NSTA http://www.nsta.org/store/product_detail.aspx?id=10.2505/9781938946226). Then, we used BCA tables over several days to formalize how we relate reactants to products. This was my first time using this approach, but not only did my students demonstrate a significant improvement in their ability to answer a stoichiometry question, but they had a vastly improved conceptual understanding. They understood the mole ratio conceptually and understood how the mole conversions were something different, not just another step in a calculation. They also understood that we started with set amounts of reactants and those substances became the products. This is the first time I've ever enjoyed teaching stoichiometry to this population. I'd love to talk about it more as it's really been eye-opening for me. Thanks for bringing it up!
Wondering about Argument Driven Inquiry
Thanks for the response, Chad. I appreciate the dialogue.
I'm quite interested in how you used the Argument Driven Inquiry to build their understanding of the connection between the counts of reactant and the counts of products. I think I'll need to take a look at the book you reference here. Can you provide a bit more discussion on this?
As for the BCA tables, did you find students moved easily into and out of the BCA table (from grams to moles to get into the table, and converting the "answer" back to grams moving out of the table)?
Thanks.
Lowell
Anybody use BCA tables for titration calculations?
Just yesterday I was teaching my class about the calculations required for titrations, and wondered if anybody uses BCA tables for this?
I also use BCA tables in my class (I wrote the article, "Rethinking Stoichiometry" http://www.chemedx.org/blog/rethinking-stoichiometry) and I swear by them. My students use traditional dimensional analysis or proportional reasoning to convert between mass to moles and do not have a problem with it because it is an entirely separate step. Students know that only moles can go into the BCA table and by the end of the unit they can do stoichometry with mass, gas volumes and solutions (molarity). I call everything outside the BCA an "add-on" and we learn each "add-on" separately so students have ample practice.
I have found the key to getting students to understand the "change" line is driving home the concept of ratios. All year we talk about ratios (thanks to Modeling Instruction). Students are very familiar with the language, "for every blank moles of reactant, you can produce blank moles of product" or vice versa. By thinking about the reaction in terms of ratios, students know if they need more product or more reactant and the numbers in the BCA table start to have meaning.
I love how well some of your students were able to articulate the differences between the alorithmic method and BCA tables!
Titrations
Lowell,
I have found them to be very helpful in titration problems. I have used different names for the approach over the years but it is the same concept. I like to use millimoles in them when doing titration problems. I find them very helpful and my students follow along well.
Cheers,
Michael
Why I Choose to Use BCA Tables
"It is a simple method that allows us to see the process and it helps to give a clearer understanding of Stoichiometry."
When I read this student quote in the blog it reminded of the conversations I have heard in my chemistry classes. I have been using BCA tables for a few years now however this is the first year my new school was introduced to them. Overall the students have mixed opinions on them however I will say I actually hear group discussions explaining how the mole ratio is used rather than the memorization of which variable goes in which 'spot'.
This year I have included them in my acid/base unit and the students have transitioned with ease. Yes they will have to complete a conversion before they can use the BCA table however they have expressed that it helps to continue with the same idea. So far in this acid/base unit I have only used them for neutralization reactions however I expect my students who will continue on to AP Chemistry will see the benefit once they reach equilibrium content. Athough stoichiometry will always be a challenging unit for students I am pleased to hear that the dialogue has shifted from a memorization strategy to an explanation of the reaction, which I consider a win for BCA tables.
Share Your Thoughts