Chemistry of Brewing Part I: The Malt
Humans discovered the agriculture of cereal grains, developed communities around their fields, and promptly began brewing. Currently, the earliest evidence of barley fermentation dates the process to at least 5,400 B.C.E in the Zagros Mountains of present-day Iran.1 Loosening the criteria to include other grains pushes the date back to approximately 7,000 BCE.2 The fact that brewing spans nearly the entirety of human civilization is quite impressive, but even more so when one realizes that humanity had little or no idea of what we were doing at all until the late 1850s, when Louis Pasteur discovered the role of yeast in fermentation .3,4

Unless of course, one adopts the belief that humans don’t really make beer. If a perfect pint is considered a work of art, humans are not the artists. They are at best the essential workers that make sure that there is always paint, brushes and canvases at the ready and that the refrigerator is filled with snacks. The actual artists are a gang of enzymes and fungi whose work we can’t even hope to mimic. In this series, I’ll try to describe the chemistry of this gang.
In the most general terms, grain fermentation is the sum of two chemical transformations:
- the enzymatic breakdown of starch into simple sugars, and then
- the conversion of simple sugars to alcohol by yeast
The actual beer brewing process consists of the following steps:
- malting
- mashing
- sparging
- boiling
- fermentation
- aging and carbonation
The breakdown of polymeric starches to smaller sugar units occurs in the first two steps. The first step, malting, is the topic of this article. This step is unique in that the chemistry occurs completely within the grain itself.
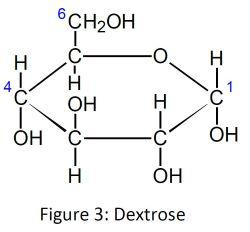
The starches found in mature barley (Hordeum vulgare) grains are mainly polymers of the monosaccharide dextrose, also known as glucose.
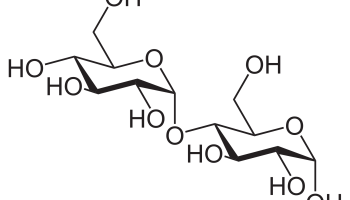
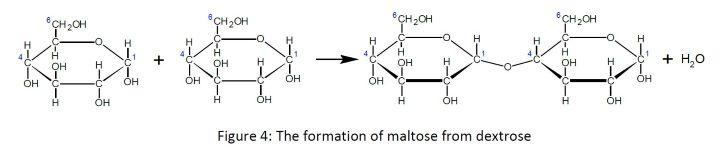
The dextrose units are linked (bonded) to form the polysaccharide starches in two ways. Both involve the removal of a water molecule and leave a bridged oxygen (technically, an ether linkage and specifically, a glycosidic bond). If the bridged oxygen is between carbon 1 of one dextrose molecule and carbon 4 of an adjacent dextrose molecule the result is a 1,4 linkage (= glycosidic bond). The dissacharide maltose is shown below.
Longer chains are called amyloses, and may be depicted in the following manner, with a horizontal bar representing the 1,4 linkage and the lowercase letter d = dextrose:
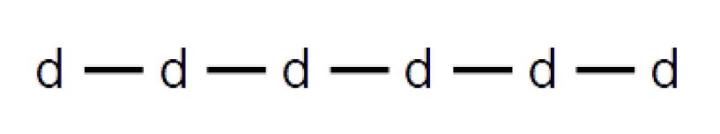
As shown below two dextrose units also may be connected via carbons 1 and 6 to give (not surprisingly) a 1,6 linkage:
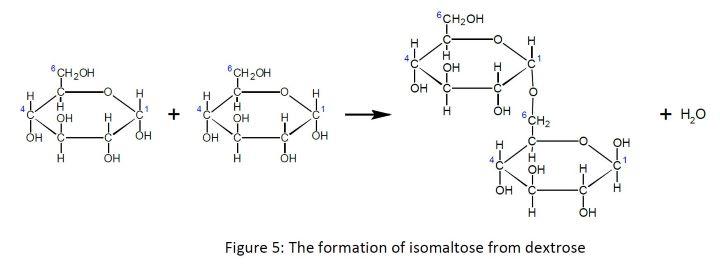
In the shorthand notation using the lowercase letter d and horizontal bars, a 1,6 linkage is represented by a vertical arrow. The disaccharide formed is commonly called isomaltose. Longer chains containing both 1,4 and 1,6 linkages are called many names by many different fields, including dextrins, α-glucans, and amylopectins. They can be represented in the following manner:
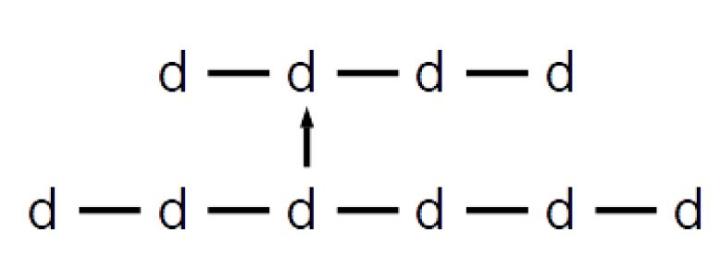
Brewer’s yeast cannot metabolize starches with more than 4 saccharide units, with disaccharides preferred for ethanol production.
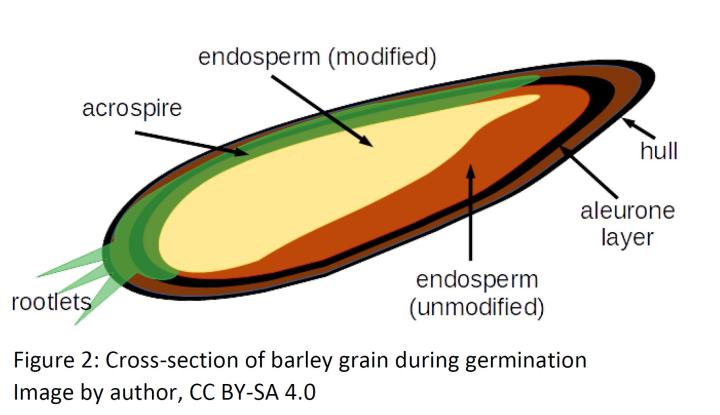
The breakdown of the endosperm begins in the malting step, where the grains are steeped in water over the course of 30-50 hours5 to raise the moisture content in the grains to 44-46%.6 The grain then begins to germinate, which triggers the activation of several enzymes already present and the production of others. Brewers refer to the conversion of the endosperm by these enzymes as modification. While maltose production is the end goal, the most important process that occurs during the malting step is actually protein degradation.6 The unmodified endosperm is a protein-glucan matrix which blocks the α-glucans from the amylase enzymes that perform most of the starch degradation. The protein component is broken down by a number of proteolytic enzymes including endopepditase, carboxypeptidase, aminopeptidase, and dipeptidase.7 The amino acids produced by the action of these enzymes is used by the embryo to support further germination. The starch granules in the unmodified barley grains are a mixture of 70–75% α-glucans and 25–30% amylose.8 The α-glucan component of the matrix is broken down by limit dextrinase. Limit dextrinase is a “debranching” enzyme, that selectively cleaves 1,6 linkages:
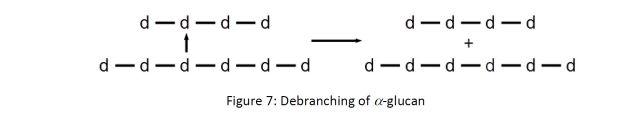
This transforms the α-glucans into amyloses, where they are further broken down in the grain by the enzyme β-amylase. β-amylase preferentially breaks 1,4 linkages near the end of the polymer chain, producing the dissacharide maltose (d-d in Figure 8 below):
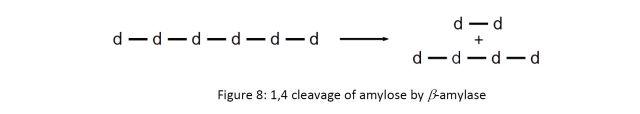
Once the desired level of modification is achieved, the grains are dried to a moisture content of 4% to stop the germination process. The dehydration and heating destroys or deactivates the enzymes and halts all other biological activity. Lager malts are heated to approximately 85°C, and ale malts to 100°C.6 These malts provide the base of most, if not all, beer styles. Further heating above 100°C is used to create specialty malts, which are used to impart flavors distinctive to certain styles (stouts, porters, etc.). Further starch and protein conversion occurs in the next step, mashing.

1 McGovern, P. Barley Beer https://www.penn.museum/sites/biomoleculararchaeology/?page_id=84 (accessed 27 June 2020)
2 McGovern, P.; Zhang, J.; Tang, J.; Zhang, Z.; Hall, G.; Moreau, R.; Nuñez, A.; Butrym, E.; Richards, M.; Wang,C.; Cheng, G.; Zhao,Z.; Wang, C. Fermented beverages of pre- and proto-historic China. Proc. Natl. Acad. Sci. U. S. A. 2004, 101, 17593–17598.
3 Vallery-Radot, R. The Life of Pasteur. Constable & Company: London, 1919; pp. 79. (Translated by Devonshire, R.)
4 Barnett, J.; Barnett, L. Yeast Research : A Historical Overview. ASM Press: Washington, DC, 2011.
5 Mosher, M.; Trantham, K. The “Food” for the Brew. Brewing Science: A Multidisciplinary Approach, Springer: Cham, CH, 2017; pp 132-141.
6 Fix, G. Principles of Brewing Science; Brewers Publications: Boulder, CO, 1989; pp 101.
7 Wiesen, E.; Gastl, M.; Becker, T.. Protein changes during malting and brewing with focus on haze and foam formation: A review. Eur. Food Res. Technol. 2011, 232, 191-204.
8 MacGregor, A.; Fincher G. (1993) Carbohydrates of the barley grain. In: Barley: chemistry and technology. MacGregor A.; Bhatty R., Eds.; American Association of Cereal Chemists: St. Paul, MN, pp 73–130.