Electroplating Copper

The electroplating copper activity is a formative assessment (FA) task that targets chemical mechanism. Thus, its aim is to give teachers opportunities for learning how students think about how chemical processes occur. In this FA task, students are asked to focus on how electrons “flow” and what this has to do with chemical changes in substances. They are asked to consider the properties of the different substances involved. Then they observe a chemical process occur and come up with ideas about what they think is going on.
This FA task was tested with high school chemistry students. In the class in which it was tested, students had recently started learning that atoms contain electrons, which can be added or removed (essentially the plum pudding model). Specifically, they had been exposed to the following:
- Atoms contain charged particles which can be added or removed.
- The periodic table can be classified into metals and non-metals (and metalloids).
- Metals conduct electricity because they tend to give away electrons (i.e., they let them “flow”), whereas non-metals do not conduct electricity because they hold onto their electrons tightly.
- Students did a matching activity where they read about how electrons are distributed in non-metal (covalent), ionic, and metallic substances, and then they decided which particle diagram matches which class of substances.
During the activity, students first do some initial work so they can remember learning about some key concepts: how to identify metals, non-metals/covalent compounds, and ionic compounds by their chemical formulas, and properties of these classes of substances. Next, groups of students receive the following materials:
- a 9-volt battery
- 2 alligator clip wires
- 2 nickels
- a beaker
- a small volume of copper (II) chloride solution
Students are asked to connect the nickels to the battery using the wires, and then dip them into the solution so that they don’t touch. Students probably will see a brownish color forming on the nickel that is connected to the negative battery terminal (this is metallic copper) and a greenish-gray color forming on the nickel that is connected to the positive battery terminal (probably a nickel oxide or nickel chloride). Do not tell students what these are. They should observe and think about it. Give the groups each a portable whiteboard to draw a diagram of what they think is going on, in particular what they think is making these color changes (and any other changes) to occur. Also give students the
Electrons and Chemical Change worksheet to write down their observations.
Teaching reflections
After reviewing student work with other teachers, the main patterns I noticed were that students were incorporating prior knowledge, recognizing that a chemical change was taking place (as evidenced by words like “rusting”, “corroding”, and “burning”), and attempting to incorporate moving electrons and/or charged particles to explain WHY these changes were taking place. In the future, I think I might think about doing this FA twice in the same unit – coming back to it at the end of the unit and trying to re-explain our observations using what we learn about electrons, “noble gas envy”, and chemical change over the course of this unit.
Student Group 1 whiteboard
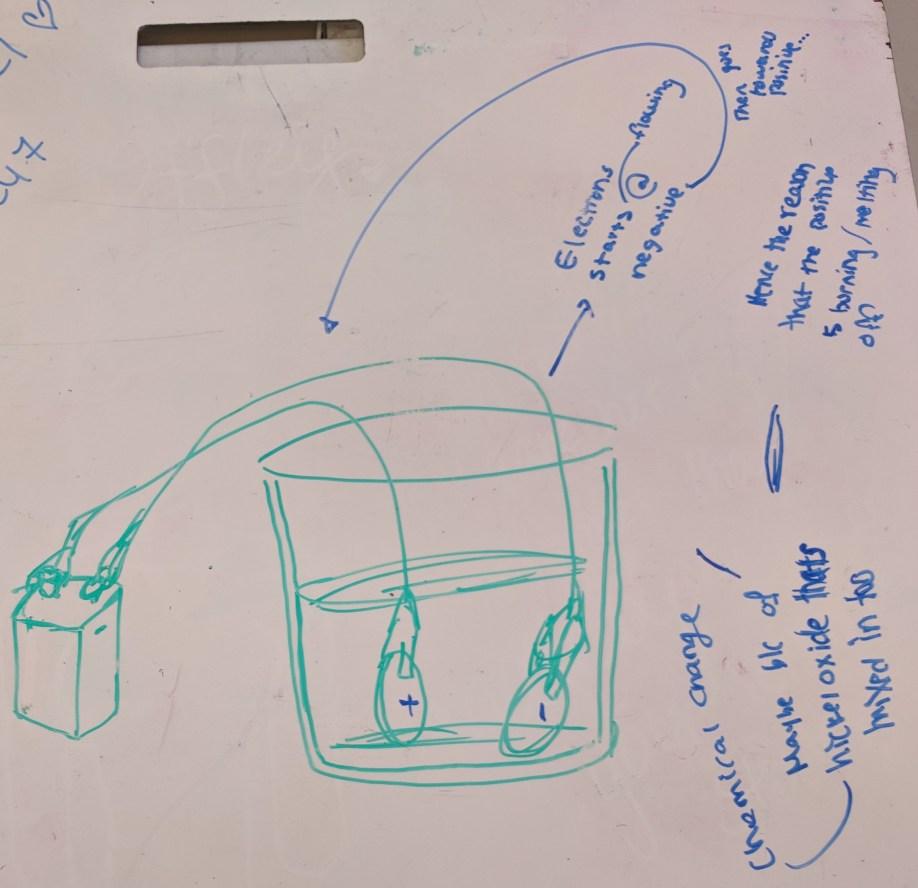
Student Group 1’s writing on this whiteboard (sideways) says:
- Drawing shows a beaker with liquid in it. Two nickels are in the liquid and are connected to wires. One nickel (on left) has + sign on it, and its wire connects to the + terminal on a battery to the left of the beaker. The other nickel (on right) has a – sign on it and connects to the – terminal on the battery.
- Chemical change maybe b/c of nickel oxide that’s mixed in too
- Electrons starts flowing at negative, then goes toward positive
- Hence the reason that the positive is burning melting/melting off
Student Group 2 whiteboard

Student Group 2’s writing on the whiteboard says:
What COULD be happening:
- The ions are leaving the neg. side causing the nickel to corrode in the water
- The ions are passing through the solution causing the other nickel to pick up the charge, and it’s also picking up pieces of the corroded nickel
Student Group 3 whiteboard

Student Group 3’s writing on the whiteboard says:
- Drawing at the top shows a beaker with a line at top of a small amount of liquid, two circles (labeled “Red” and “Green”) are standing up in the liquid and each circle has a wire attached at the top. The wire on the Red circle goes to the – (minus) terminal on a battery. The wire on the Green circle goes to the + (plus) terminal on the battery. Underneath the battery there are two arrows. Arrow pointing from the left side (below the +) says “ions”. Arrow pointing from the right side (below the -) says “electron”.
- Info: nickels have some copper in it; copper chloride ionic compound
- Nickel lost shine, copper crystal-like material on top forming, since copper chloride is an ionic compound maybe the electrons and solution created a copper coating on top of the nickel
- Green colour forming from the ions taking the copper inside and rusting it?
Student Group 4 whiteboard
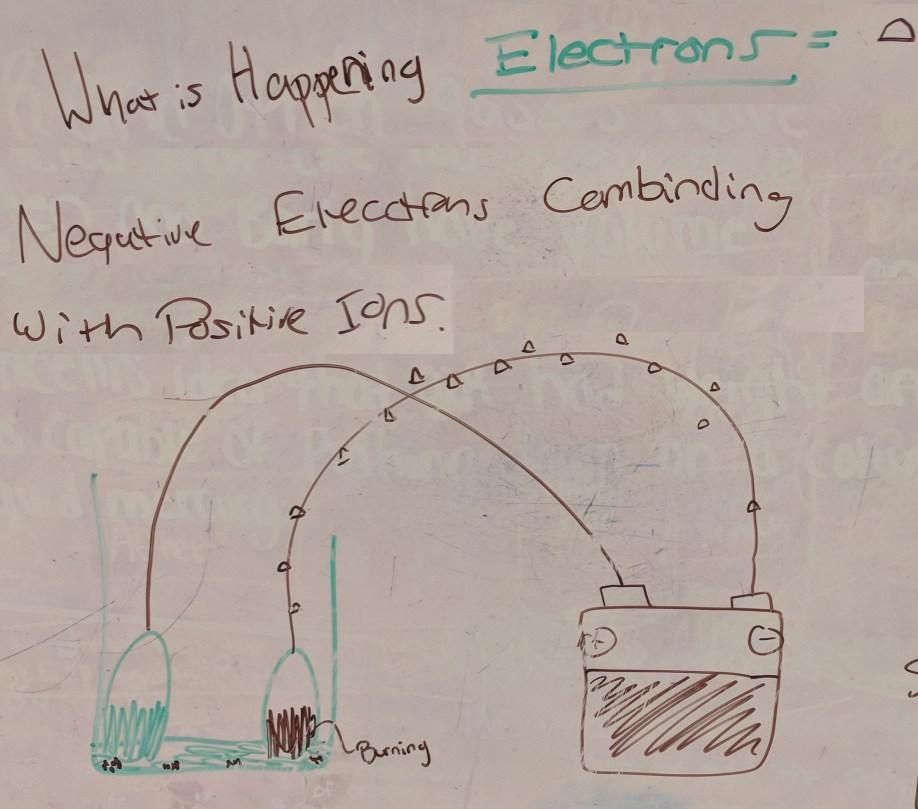
Student Group 4’s writing on the whiteboard says:
- What is Happening
- Electrons = (symbol of half circle)
- Negative Electrons Combinding with Positive Ions
- Drawing at the bottom shows a beaker with liquid at the bottom (shown as green) and some bunches of brown resting at the bottom of the liquid. Two ovals (nickels) are shown, each with a wire coming from the top. The nickel on the left is half colored green (bottom half). The nickel on the right is half colored brown (bottom half) and is labeled “Burning”. The wire from the left nickel goes to the + terminal on the battery. The wire from the right nickel goes to the – terminal on the battery, and has symbols of electrons (half circles) along it, kind of looking like ants crawling along the wire.