8. What affects chemical change?
“What affects chemical change?” is a question of identification of internal and external variables that affect the extent and rate of chemical processes. The extent to which reactants are transformed into products and the rate at which the transformation occurs depend on internal factors such as the composition and structure of the particles involved and their concentration in the system, as well as on external factors such as temperature, pressure, and the nature of the environment in which the reaction takes place (e.g., type of solvent, pH). Identifying these factors and their effects on reaction extent and rate allows us to design and control chemical processes for particular purposes.

The volcano probe formative assessment was developed by the ACCT team (indicated by the star), and the fire story and frying ice were developed by past ACCT cohort members.
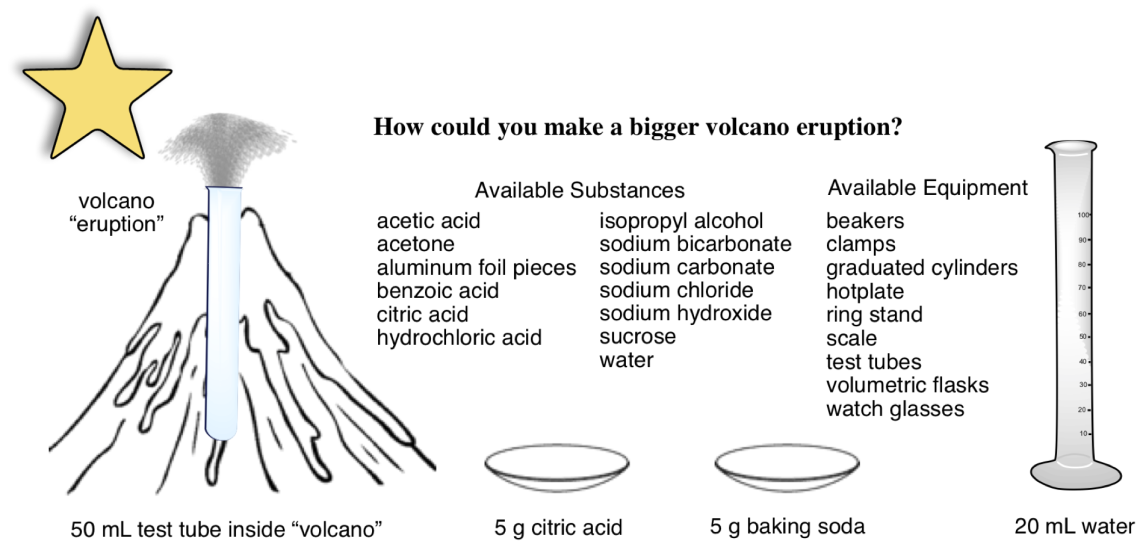
Volcano probe
The Volcano Probe is a formative assessment tool for learning about how students think about choosing reactants and reaction conditions based on how they think about chemical control. It has been tested in middle school, high school, and university chemistry classes.
The Fire Story
The fire story formative assessment is used to explore students’ thinking about the question “what affects chemical change?” through a real-world problem. This formative assessment specifically probes students’ understanding of energy and phase changes. There are several different explanations using different chemistry concepts that could be used to explain the phenomenon explained in the story.
Frying Ice
In the Frying Ice formative assessment students explore the three phases of matter. This formative assessment targets the question “What cues are used to differentiate matter types?” The target of this formative assessment was for students to demonstrate their understanding of the three states of matter and how the particles in each state react when heated.