6. What determines the outcomes of chemical changes?
“What determines the outcomes of chemical changes?” is a question of dynamic behavior and probability. When the particles that make up different substances interact with each other, their interactions may lead to a variety of structural changes (e..g., some atoms may change positions or separate from a molecule). Which of these random changes are more likely to occur depends on the relative potential energy between the original particles and the new particles that are formed. More stable particles (with lower potential energies) are more probable to form and will determine the path and outcome of the reaction. Understanding the relationship between structure, stability, and reaction mechanism allows us to predict, explain, and control the products of chemical processes.

The Cool off Challenge formative assessment was developed by the ACCT team (indicated by the star). The Balancing with Legos, Acids, bases and equilibrium oh my! and What Makes Volcanos Erupt? formative assessments were designed by past ACCT cohort members.
Cool off Challenge
The Cool off Challenge is a formative assessment tool for learning about how students think about chemical causality. It has been tested with middle school and high school students, and with preK-12 science teachers. It provides participants the opportunity to think creatively about a science challenge, and then test their predictions in the lab.
Balancing with Legos
The Balancing with Legos formative assessment task asks students to create the product substances given the reactants. Students have to figure out what they did to make it work, and that gets them to talk about how they think about balancing chemical equations. The formative assessment targets chemical mechanism, and provides an opportunity for students to use a hands-on model to realize that, in order to conserve matter, the atoms need to rearrange into the new compounds in specific ratios. The goal of the formative assessment is to elicit students’ thinking about how compounds behave. The learning goal is to have students understand that only full compounds can be introduced on the reactant side for balancing in order to make complete compounds on the product side.
Acids, bases and equilibrium oh my!
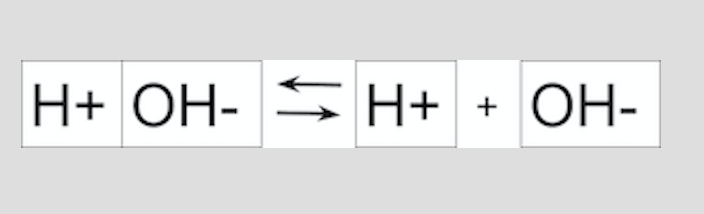
The Acids, Bases, and Equilibrium, Oh, my! formative assessment asks students to model the effect of a strong acid on the dissociation equilibrium of water. This formative assessment targets Chemical Mechanism by examining how students interpret LeChatelier’s principle as it relates to pH. The central idea of this formative assessment is that the dissociation of water is based on equilibrium and looks at how adding more H+ ions or more OH- ions would affect that equilibrium. The questions in the assessment are predominantly eliciting questions, in that they ask students to narrow in on one aspect of acids and bases reacting with water. There is plenty of opportunity during the formative assessment for teachers to use advancing questions to get students to explain their thinking.
What makes volcanoes erupt?
The What Makes Volcanoes Erupt? formative assessment asks students to draw and write what causes volcanoes to explode and then choose from three options of movement in Earth’s mantle to select the one that showed convection currents accurately. This formative assessment targets chemical mechanism, which focuses on how different processes occur. The formative assessment focuses on how volcanic eruptions occur and helps to uncover students prior knowledge of plate tectonics.