A Logical Order of Topics for Honors Chemistry and AP Chemistry
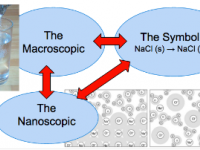
In this post, a veteran AP Chemistry teacher explains his rationale for the course's topic order. By using an empirically driven sequence, students build deep conceptual understanding and rely less on memorization of facts. It’s a must-read for any educator aiming to make chemistry more meaningful—and more memorable—for students.