NGSS Periodicity Lesson
The periodic table that most of us use in our classrooms was originally developed by Dmitri Mendeleev, but thousands of alternate versions of the table have been devised over time. In this lesson, students are offered a variety of alternative versions of the periodic table. Students will identify trends that are consistent from one table to the next in order to understand why the tables they are working with and Mendeleev's version are organized in the manner that they are. Students will be prompted to use trends in subatomic particles, valence electrons, atomic radius and metallic properties. Students will also work on their process skills including critical thinking, asking questions, constructing explanations, and engaging in arguments from evidence. This lesson was designed to fit the NGSS performance expectation HS-PS 1.1 but can be used for any first year chemistry course or modified at your discretion.
Prior to this lesson, my students learned ways to identify types of elements in terms of metallic character, reactivity, group and period numbers, and atomic radii in class and then watched video tutorials at home that I created. To warm up and engage the students in this lesson, students individually answer the two “Do Now” questions at the top of the handout (this student document is available in the Supporting Information below) to define periodic in their own words and then turn to a partner to share ideas. Have volunteers report out a few of those ideas to the class.

In the exploration phase of the lesson, students are grouped in teams of 3-4 students. Each team is assigned an alternate periodic table to explore. (See figure 1 and 2 for two examples. A wide variety of others can be found by following the links in the Reference section below.) Students fill in the “Observe Your Table” section of their handout; identifying how their table is organized, how their table is different or similar to our current periodic table, and the properties that make their table unique. One student from each team will explain the trends in their periodic table to the class.
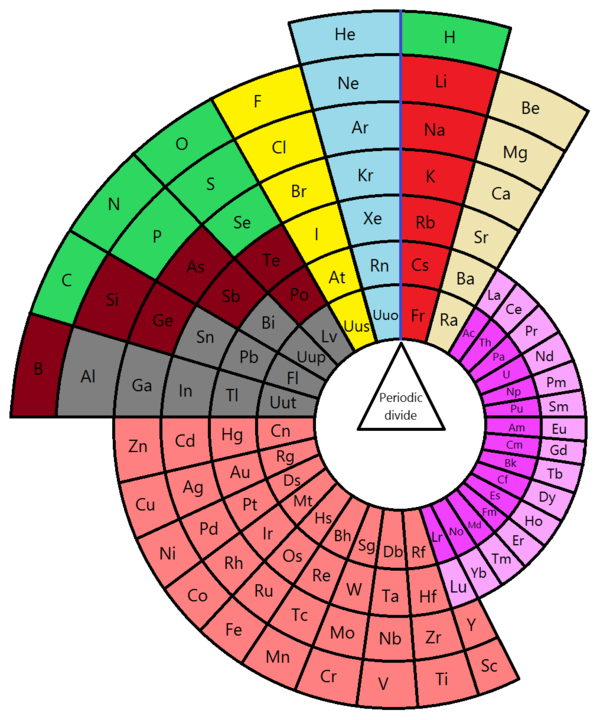
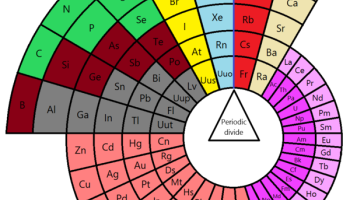
Figure 1: Alternative Circular Periodic Table, by Marco Piazzalunga - Own work, CC BY-SA 3.0, Link1
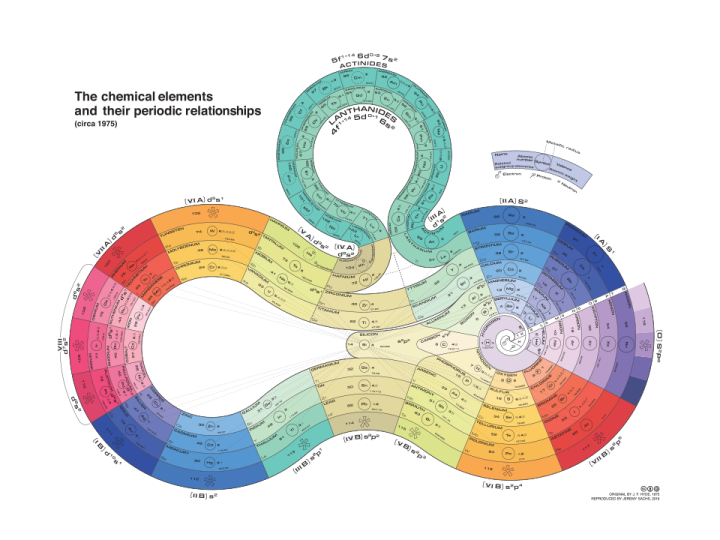
Figure 2: Curled Ribbon Periodic Table, by Rezmason - Own work, CC BY-SA 4.0, Link1
Observe Your Periodic Table
|
How are the elements arranged on your table? What properties were used to organize the elements? |
How is your table different than the standard periodic table? Give specific examples. |
|
How is your table similar to the standard periodic table? Give specific examples. |
What makes your table unique? Give specific examples. |
In the explain section of the lesson, each team will take turns explaining the commonalities and unique properties of their table. While the speaker is explaining their table, all other students should take notes in the first two sections “Patterns Found in All Tables” of their handout; including the properties that are commonly used to organize the elements and how similar elements are grouped. Each team will need 1-2 minutes to report out.
Patterns Found in All Periodic Tables
|
What properties are most often used by the tables? Provide examples. |
Are similar elements grouped? How so? Provide examples. |
|
How can these alternative tables help students learn the periodic properties of elements better than the standard tables? |
Why do you think the standard tables are preferred by most scientists and educational faculty? |
In the elaborate section of the lesson, as a class, list some answers to the first two sections of the “Patterns Found in All Tables” to ensure all students have identified the common characteristics before moving on to the next questions. In teams, students will then answer the last two sections “Patterns Found in All Tables” asking how the organization of different tables may be beneficial to students and why Mendeleev's version is the primary table used around the world. A few students may be asked to volunteer ideas they have that may answer these questions.
To close and evaluate the lesson, students turn their page over to answer the "ticket out" question, “If you were to design your own table, how would you arrange the elements?” Students should be encouraged to draw a mock representation of their ideas. Including organizational ideas and labels.
I ran this lesson in three separate classes this fall and it was met with fascination and engagement of my students. They loved seeing all of the different ways the elements could be arranged. As each team presented their table, I projected an image of that table on my white board for the class to see. Some really fun table concepts came up in our closure question such as creating a three-dimensional cylindrical table where noble gases lined up to the alkali metals in atomic number order, or spherical shapes similar to ones we looked at that included more information or different arrangements. Most students chose to draw their design. Some less creative students chose to take the table we currently have and add modifications to it such as adding the abundance percent on Earth, or other information not currently on our reference sheet, which I found to be perfectly acceptable. It was definitely a fun and engaging lesson that led to a lot of student to student conversations about what normally is presented as a dry and directive lesson on periodicity. This lesson also really helped in later lessons and assessments when students chose to define element properties based on their location on the table (opposed to a more acceptable answer using subatomic particle arrangements) because I would refer back to the many arrangements we had seen in this lesson and ask, “is that true on all tables?”
NGSS Three Dimensions Connections2
Performance Expectation: HS-PS1-1. Use the periodic table as a model to predict the relative properties of elements based on the patterns of electrons in the outermost energy level of atoms. [Clarification Statement: Examples of properties that could be predicted from patterns could include reactivity of metals, types of bonds formed, numbers of bonds formed, and reactions with oxygen.] [Assessment Boundary: Assessment is limited to main group elements. Assessment does not include quantitative understanding of ionization energy beyond relative trends.]
Disciplinary Core Idea: Each atom has a charged substructure consisting of a nucleus, which is made of protons and neutrons, surrounded by electrons. The periodic table orders elements horizontally by the number of protons in the atom’s nucleus and places those with similar chemical properties in columns. The repeating patterns of this table reflect patterns of outer electron states.
Cross Cutting Concept: Different patterns may be observed at each of the scales at which a system is studied and can provide evidence for causality in explanations of phenomena.
Science and Engineering Practice: Use a model to predict the relationships between systems or between components of a system.
Notes and References
- Wikipedia contributors. (2019, December 4). Alternative periodic tables. In Wikipedia, The Free Encyclopedia. Retrieved 00:24, December 7, 2019, from https://en.wikipedia.org/w/index.php?title=Alternative_periodic_tables&…. The Alternative Circular Periodic Table can be seen in figure 1 and the preview image above. The Curled Ribbon Periodic Table is seen in figure 2.
- As seen on the Next Generation Science Standards website (accessed 12/06/2019)
- Another resource that includes a variety of periodic tables is the Internet Database of Periodic Tables housed on the Chemogenesis Web Book compiled by Mark R. Leach. (accessed 12/4/2019)
- Quick Links to several tables found on the sites mentioned above:
Comments
5Great Activity!
This was a great activity to help students see some of the trends in the periodic table, and I loved seeing the organization of the other tables "click."
In reply to Great Activity! by Shana Ball
Periodicity
Thanks so much! I hope you enjoy it and if you think of any modifications I'd be happy to hear them! :)
Share Your Thoughts