Mg Lab - A Lab(s) That is Great for Differentiation

What are we doing to help kids achieve?
It is always helpful to have a lab that can be adapted to meet the needs of students. The "Magnesium Lab" is one of these experiments.
This lab can be adapted a number of ways. It can also be "tweaked" to meet the needs of the number of students or time constraints. Here is the "jist" of the lab. Students obtain a small piece of magnesium. They pour a small amount of a strong acid, usually 6 molar HCl, into the bottom of a eudiometer or gas collection tube. They slowly fill the tube with water until it is completely full. The magnesium is placed in the tube with a stopper with holes (sometimes held in place with copper wire). The eudiometer is carefully turned upside down and placed into a tub of water without letting air enter the eudiometer. The acid goes down the eudiometer and reacts with the magnesium metal. Hydrogen bubbles up and it forces the water out the bottom. Students then can record the volume of gas. Michael Morgan discussed a version of this lab, Molar Volume of a Gas Lab that includes a great tip and a handout with instructions. There are many versions of this lab that can be found online or in old lab books. If you have never tried this before, you might want to consider one of the versions outlined below.
Calculating "R".
Students record the volume of hydrogen gas in the eudiometer. Students then record the atmospheric pressure and take into account the pressure due to water vapor. The mass of the magnesium can be converted to moles. The temperature of the water is recorded. It is assumed that the temperature of the gas is the same as the temperature of the water. Students use the ideal gas law to calculate the universal gas constant R and then compare to the actual value.
Calculating Moles
Students do the same experiment as described. They record the pressure, volume and temperature of the gas in the experiment. Students use the combined gas law to find the volume of the hydrogen at standard temperature and pressure. According to Avogadro, and gas at STP should equate to 22.4 Liters of volume for every mole of gas. Students then examine the volume they have and the moles of gas and compare.
Predicting the Volume of Gas
An AP teacher in Ohio came up with this idea that I did this week. Students were getting close to the end of the quarter and finals were approaching. Everybody was tired (including me). We had done some problems with gas laws, the ideal gas laws and Dalton's law of partial pressures. Students were told that they would be provided a piece of magnesium. They had to do the above experiment. Before they began, they had to predict the volume of gas that would evolve. Their grade depended on two parts. First they had to show their reasoning and calculations on a piece of paper. Second they had to predict the volume of gas. The closer the prediction, the higher their grade. See figure 1. Every lab group had a different mass of magnesium. Most groups solved the problem using the ideal gas law and subtracting the pressure of water vapor. The best part of the lab is that they received feedback within ten minutes of starting the reaction. The part about this lab that I liked the most is that students experienced a great deal of success and that it was easy to grade. Another aspect about this lab that was helpful was the number of concepts students had to use that were great review with exams around the corner. They had to predict products and balance equations. They also had to measure, use significant numbers, record data and work with gas laws. It also involved stoichiometry.
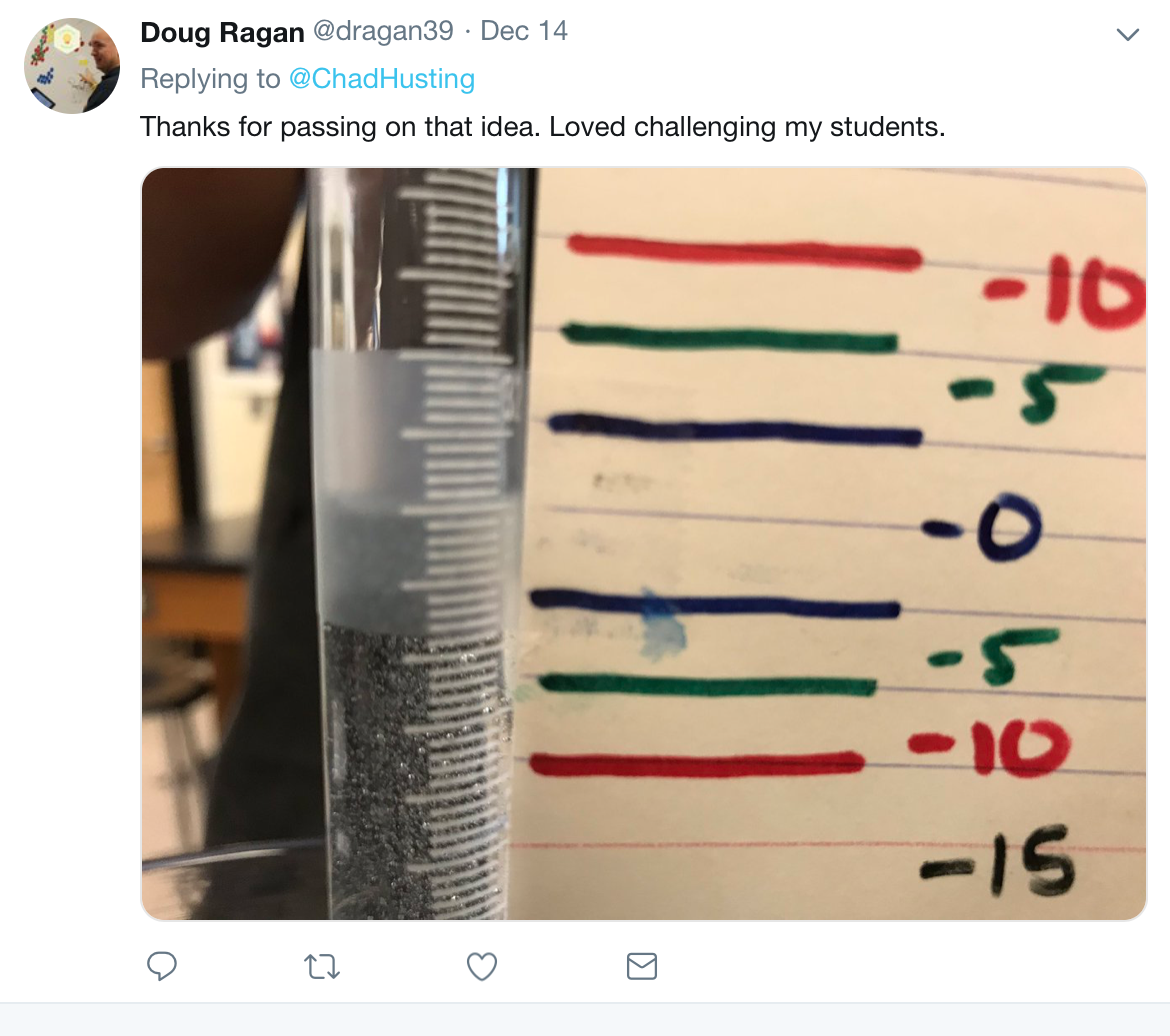
Figure 1 - Doug Ragan replied to my tweet about this lab activity with a grading tool.
Do you have a different way to do this lab? Do you have a great lab practical or demonstration that you use? Please consider posting or commenting. We would love to hear from you.