Developing a Model for Enthalpy of Solution—Reframing the Approach

There has always been something I have loved about calorimetry. Waiting for that transfer of energy within the system to match my prediction while simultaneously trying to prevent this invisible stuff from escaping is a fun challenge. Trying to teach it can be a fun challenge as well. Since this year created numerous opportunities for us to rethink how we do things, it was only a matter of time before my class reached the topic of enthalpy and a different approach would be needed.
Due to the variation of hybrid/distance learning over the past year, part of my pedagogy that I feel has improved involves creating opportunities for students to continue playing a large role in the development of a concept. This means reframing many of the topics covered to exhibit a data-to-concepts approach rather than the other way around. In a classroom that is still predominantly virtual, this takes a bit more work, but the challenge to engage students in authentic scientific practices is one that is worthwhile. To provide some clarity on this idea, I wanted to share an example enthalpy activity my class recently did that helped me rethink how to overcome logistical barriers in an effort to provide a quality of science education that I can be proud of when I leave school. What I ended up with was something that not only improved my traditional approach to enthalpy, but also provided me with greater confidence in the role that students can play when developing new ideas when provided with the appropriate support.
During our unit on solutions, we have traditionally done some kind of investigation involving calorimetry, like designing an effective cold pack. Whatever it is, students must have some idea of how to apply ideas related to the exchange of energy between solute and solvent. While the investigations, demos, and other activities I have done throughout the years surrounding this topic certainly have value, they all inevitably expected students to utilize an equation or two that seemingly had no origin. Why do we use this equation? Who cares—it helps you get the right answer doesn’t it? While that has never been my actual response, it is not that hard to imagine students feeling something similar. That did not sit well with me. So, I decided to alter my approach and focus on getting students to first develop the mathematical model for enthalpy of solution so they could eventually apply it to make predictions for different solutes being dissolved. At least this way I knew they were more likely to have an increased awareness of why they needed to use this specific equation to solve their problem.
To do this, I proposed the following question to students: How can we develop a mathematical model (equation) that describes the amount of energy released/absorbed for every one mole of solute dissolved so that we can predict the temperature change for any quantity of solute dissolved? Not the most concise question, I know. But we deconstructed it and started talking. I asked them to consider any variables that might be useful in helping us answer this question. Here are a few example responses they provided:
- temperature
- amount of stuff being dissolved
- the container holding the solution
- amount of water
- temperature of water
- energy needed to break bonds
- energy released when bonds form
- how much you stir, etc.
Not every response was relevant or something we were actually planning to measure, but that was not the point. The goal was to get them thinking and developing that skill of experimental design. A brief back-and-forth ensued for nearly every variable suggested until we eventually reached a consensus on what variables we could easily measure and would be most likely to help us reach our goal. This left us with the initial/final temperature of water, volume of water, and the mass of solute dissolved as our primary variables.
Great, so what do we plan to do with these values once we actually measure them?
At this point, students’ lack of prior knowledge on topics like specific heat and energy meant that I needed to facilitate this a bit. To address specific heat, I asked them simple questions about their own experiences with different objects heating or cooling more quickly than others. Without giving it a label, several students quickly referenced events typically involving metal objects or walking on asphalt compared to grass during the summer. After this minimal background primer, I provided the definition of specific heat to give them a label for what they were describing. We eventually agreed that this too would likely be important to consider, so I provided them with the specific heat for water.
With all these variables taken into consideration, I showed them the following slide (figure 1). This was kind of a cool moment because nearly every single variable either came from them or the result of our conversation.
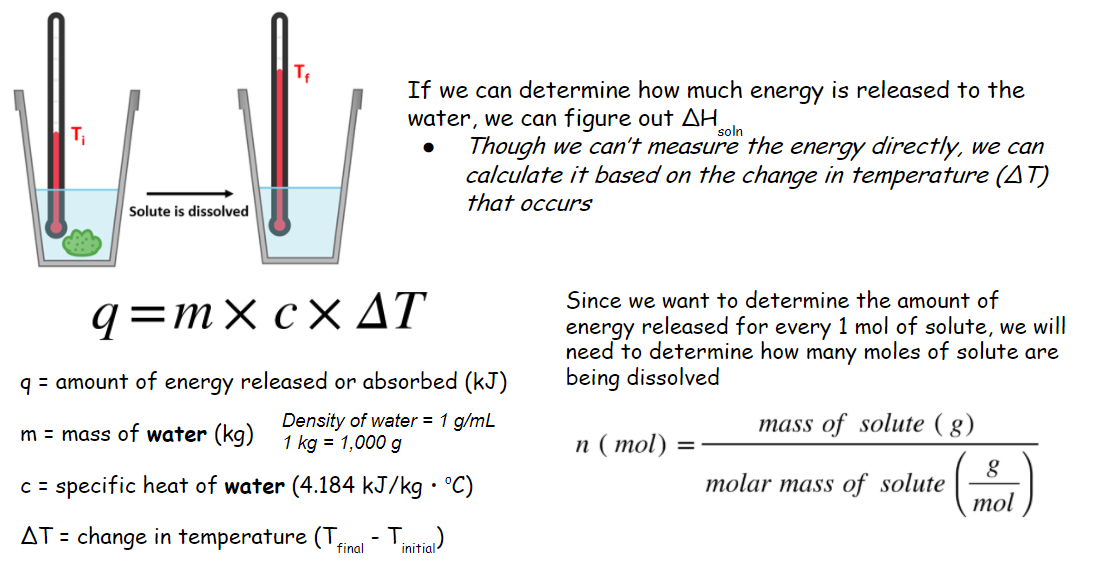
Figure 1: Google Slide summarizing enthalpy variables
Okay, so we know the things we are measuring will allow us to calculate energy (in kJ) and moles of solute. But what do we do with that information? Let’s try graphing it! What should go on X and Y? What variable are we changing and what variable will likely change in response? This line of questioning eventually led to an agreement that energy (q) would go on the Y-axis and moles of solute on the X-axis. Looks like we are ready to go.
I gave them access to two links:
1) The first link took them directly to a Google Sheet (see figure 2) that contained areas for the experimental and calculated data we had already discussed. Though I had originally planned to have them do the calculations manually and then enter the values, I thought this may be a useful opportunity to teach them how to program a spreadsheet to make it do what you want. I went over a quick tutorial on how to make the spreadsheet calculate things like energy (q) and moles of solute on its own once the appropriate data were entered. The time saved from lack of manual calculations easily compensated for the time spent teaching them how to do this. Additionally, their spreadsheet already included a graph. However, it was completely blank. I showed them how the graph would auto-generate once energy and mole values were placed in their table. They liked this and so did I. This ensured me that an appropriate graph would be produced, assuming appropriate data were used, and it saved them the time from navigating Google’s chart-making features—a skill better suited for another day.

Figure 2: Image of tables from the Google Sheet students used to record data from video
2) The second link took them directly to the experimental video that I had filmed earlier that week (see video 1 below). I do not have any fancy camera equipment (or knowledge) but a typical smartphone these days has a good enough camera that can easily film an experiment with sufficient quality.
Video 1: Ben Meacham's experimental video, ChemEd X Vimeo Channel (accessed 3/16/2021)
Since the video was only about five minutes long and a spreadsheet had already been setup to auto-generate certain calculations as well as the graph, I gave students between 10-15 mins to input data from the video, produce their graph and copy/paste it back into their lab document. From there, the mathematical model for enthalpy we were searching for could be developed.
After dissolving various amounts of NaOH, students eventually produced the following graph (see figure 3).
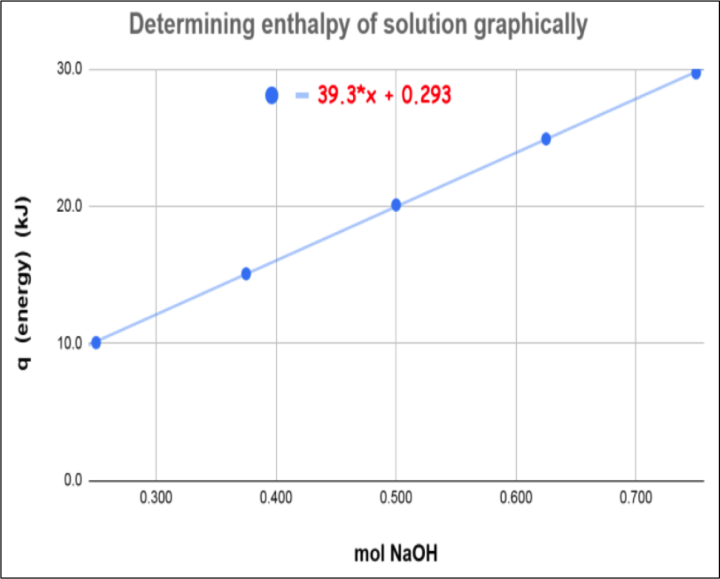
Figure 3: Enthalpy graph created with data collected from experimental video above
I asked them a few questions focused on the slope of the graph.
What does the slope of the graph seem to describe in terms of the variables in the experiment?
How many kJ of energy released for every one mole of NaOH dissolved?
What is the value (and units) of the slope?
39.3 kJ/mol
On a graph of energy (kJ) vs. amount of solute (mol), the slope gives us…
Enthalpy of solution (∆Hsoln) – how much energy is released/absorbed for every 1 mole of solute dissolved
Awesome! We have now at least found out how to determine the enthalpy of solution graphically. Now let’s focus on developing the mathematical model.
What kind of relationship does your graph appear to follow?
Linear
What is the generic equation for a linear relationship?
y = mx + b
Rewrite this equation, replacing “X” and “Y” with your respective variables “q” and “n” as well as what you now know the slope (m) to represent (∆H)—you can ignore the y-intercept since no energy should be released/absorbed if no moles of solute are dissolved.
q = ∆H · n
Rearrange your equation so that ∆H is by itself.
∆H = q/n
There it is—our mathematical model for enthalpy of solution that can be applied to any solute! In science, when a model is developed, the only way to see whether it is a good model or not is to test it against reality. Let’s see how this does in the wild.
It was at this point that I asked them to use their new model to predict what the final temperature of the solution would be if I dissolved 7.74 g of NH4Cl in 250 mL of water. I gave them the accepted ∆Hsoln for NH4Cl (14.8 kJ/mol) and asked them to determine their answer while in breakout rooms.
After roughly five minutes or so, I brought them back and asked for answers. With the exception of a couple groups that made simple math errors, the rest told me the temperature would decrease to 17.7 °C. Now that they had used their new model to make a prediction, the only thing left to do was see how their prediction compared against reality. I pulled up a video I had previously filmed of me carrying out this exact process. See an image of the final temperature in figure 4. Well would you look at that…off by 0.1 °C—BOOM!

What I enjoyed most about this was that students had been given an opportunity to gain a sense of what it is like to construct knowledge in a way that allows for meaningful predictions to follow. In the past, I would have given them the enthalpy equation and asked them to make a similar kind of prediction. That too would have resulted in an accurate prediction. However, the journey taken to arrive at such a prediction would have been entirely different and void of any feelings of ownership from students in the model they used to make their prediction. Does this difference really even matter—YES!
The construction of new knowledge, developed by both student and teacher, is the type of thing that makes me excited to teach. While creating these opportunities is certainly more logistically challenging from a teacher’s point-of-view, it is not impossible; regardless of the limitations placed upon us. This activity alone was only possible because of the skills I have been able to learn as a result of these same limitations. Though we are likely to return to the classroom in full by next schoolyear, I am more confident now than I was before that I can provide these kinds of opportunities of creation for students if I truly want to. So, I will continue with this mindset and develop the necessary skills to make it happen.