First Day Chemistry Engineering Activity

I just finished my first week of school, like many teachers in the Midwest. I work hard to get my Honors Chemistry students in a lab setting as soon as possible. It is difficult to find a perfect lab to do on the first or second day of school. In my mind, the ideal first chemistry lab would require no prior chemistry knowledge, involve interesting chemistry, address an NGSS standard, be relatively safe, not require expensive glassware or lab tools, and reinforce positive class norms. I have found engineering labs fit the bill! I don't know if I have found the "perfect" lab, but I have found something close I want to share!
For the last few years, I have been utilizing an engineering task as my introductory lab activity. During the engineering task, students work together in teams with POGIL-like roles (manager, recorder, presenter, and reflector) to solve an engineering problem. I use POGIL (pogil.org) and cooperative groups on a regular basis in my classroom so it only makes sense I start my class this way as well. Students conduct many inquiry labs in Honors Chemistry. During the engineering task, students get comfortable making their own claims based on evidence collected from the labs they design and scientific reasoning. Engineering design is one of the NGSS standards, so I am able to meet standards on the first day of class.
When I selected my engineering task, I was looking for a relatively safe and simple task related to chemistry. My search ended with slime. Making slime has become very popular over the years. The slime recipe using glue and borax is simple and can be modified to get different results. It is relatively safe and utilizes interesting polymer chemistry I discuss later in the school year. I decided to have my students engineer a superball that would bounce the highest in the class. This was an excellent lab that promoted great teamwork and discourse. My only problem was finding a way to quickly and accurately determine a winner. The superballs bounced, but often in all different directions. It was difficult to capture the bounce in a cellphone video or with a motion sensor. This past summer I was on the hunt for a new twist to my tried and true engineering task. Fellow ChemEd X blogger, Tom Kuntzleman, suggested that I have students engineer magnetic slime instead. The idea seemed very promising so I tried it out this past week in my Honors Chemistry classes.
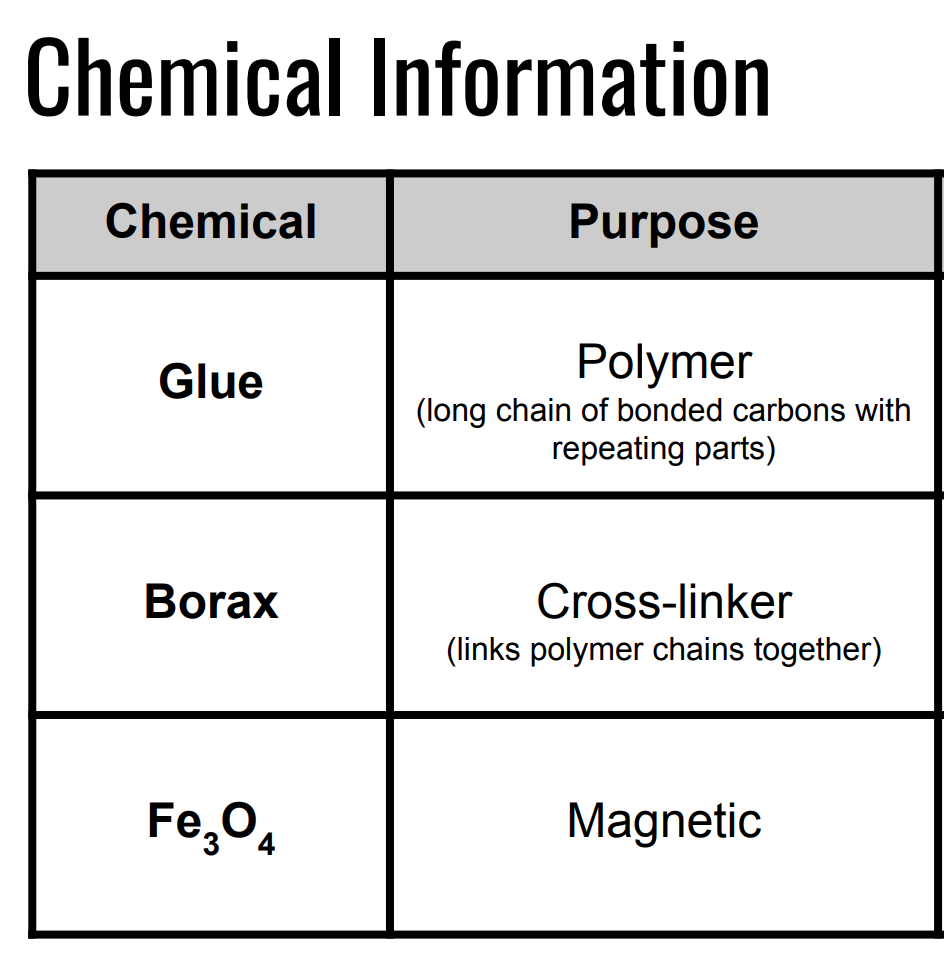
Figure 1: Chemical Information
Each lab group received a set of role cards that I use during labs and POGIL activities (see the end of the post for handout). I instructed them to read the cards and assign each person a role. After we briefly discussed why we use roles and what they are, I presented the engineering task (see the end of the post for a copy of the slides I used to present the lab). I told them their task for the activity was to engineer magnetic slime so that it climbs the highest when exposed to a neodymium magnet. I also told them I expected each group to report out the % Fe present in their slime. I gave them the rough recipe of slime, but did not tell them any specific amounts to use. We discussed the SDS for each chemical so they understood that they had to wear goggles and gloves during the lab. I also gave them a chemical information chart with the purpose and molecular structure of glue, borax, and iron(III) oxide so they could make informed chemical decisions and changes between trials (figure 1). On day one, students had 15 minutes and only one cup of glue and one cup of borax (with unlimited tap water) to run multiple trials and tests create a slime consistency they thought would be ideal for the challenge. On day two, they received a cup of iron(III) oxide and a magnet. I reviewed how to use an electronic balance and the % Fe calculation. Then students separated the slime they made the day before into different batches so they could test how different ratios of iron(III) oxide responded to a magnet. They had another 15 minutes to complete the task and we ran the class magnetic slime tournament to crown the winner. The winning group presented their recipe and % Fe used to the class. Afterward, groups reflected on the class norms: leave no one behind, group discussion is focused on class content, and everyone works together as a team (see figure 2).

Figure 2: Reflecting on the Challenge
The Magnetic Slime Challenge Engineering Lab was a success. It was much easier to determine a winner compared to the Superball Challenge I previously did. Students were engaged and I was able to pack a lot (chemistry content, hands-on lab, standards, class norms, and lab safety) into one activity. Reflecting on this activity, I did run into a few snags I will need to iron out for next year. Some groups, despite running multiple trials, ended up creating superballs and not classic slime. Overnight the borax had more time to react and everyone's slime was "tighter' than the day before. The superball-like slime did not combine with the iron(III) oxide very well. Early on the second day, it was evident that some groups did not have a chance at winning because of this. I will need to find a way to give them a chance at winning. I could run a second bracket for the best bounce. Or, I could allow for them to use more glue, borax, and iron(III) oxide if they want to remake their slime the second day. I could also possibly have them "purchase" the chemicals they need to help motivate students to be frugal with supplies. The winning slime would not only climb the highest towards the magnet but also "cost" the least.
What do you think? Do you have any ideas on how to make this engineering challenge even better?
I would love to hear about your first day or week activities! What do you do? Comment below!
Log into your ChemEd X account to join the conversation and comment. If you don’t have an account, register for one today - it is free! With a ChemEd X account, you can comment and download the attached documents.
Comments
10Awesome. Thank you for sharing I will have to give this a try as having used slow motion video to determine the height winner of the supper ball challenge. So I’ll let you know what I also Fe out! I caught what you did there!
In reply to Awesome. Thank you for by Doug Ragan
Great! I am excited to hear
Great! I am excited to hear how it goes and what improvements you made!
Some questions about amounts
What was your basic slime recipe you gave the students? For the magnetic iron was it granular or more of a fine powder?
Thank you!
In reply to Some questions about amounts by kbhetter
The basic slime recipe was on my handout attached to this post. It was a picture-based recipe that told them to combine glue and water in one cup and then borax and water in the other cup. Then they combine into one cup and knead into slime. They used the chemical information chart (also in the handout) to make educated decisions about how much glue, water, borax to add and what amounts to change as they run their trials. For example, groups that made "superball-like" slime the first trial, decided to decrease the amount of borax and increase the amount of water the next trial because they read on the chemical information chart that borax crosslinks polymer chains. I ordered in powder form from Amazon (black iron oxide natural). The reviews and description of the powder said it was great for magnetic slime - which it was!
iron compound?
Did you use Fe2O3 or magnetite (Fe3O4)? The Fe3O4 could be considered a 1:1 compoiund of FeO and Fe2O3 and might make a good "hook" to talk about oxidation states when the day comes.
I ask because Fe3O4 is (IIRC) the magnetic media used in ferrofluids.
In reply to iron compound? by Dave Blackburn
Yikes! Great catch! You are
Yikes! Great catch! You are right - Fe3O4! I blame being tired from the first week of school! I fixed that and updated the post.
I am very intrigued about your idea about oxidation states - could you elaborate on what you think you might do?
In reply to Yikes! Great catch! You are by Melissa Hemling
Since the usual way of figuring oxidation states for metal oxides MaOb is to assume O is -2 and solve for the charge of M, this is odd because they students would get a fractional oxidation number, +8/3. Then you can have them discuss whether that’s a meaningful idea. You could raise the question of is it Fe(+21/3), or maybe the oxygen is fractional? Then they work out, if they already know +2 and +3 states exist, what the ratio is. Finally, if you have samples of real Fe2O3 and FeO available, you can review pure substances vs. mixtures.
Share Your Thoughts