Immobilization of Lactase Enzyme on Alginate Beads - A Quick Test

I came across an interesting Journal of Chemical Education1 article that explains how it is possible to crosslink sodium alginate, leading to the formation of calcium alginate beads. Sodium alginate is commonly used as a thickener in food such as ice cream and fruit-filled snacks.2 It’s a non-toxic and versatile material; not only is it used for food technology purposes but also in drug-delivery and wound dressing systems.
Calcium alginate beads are hydrogels which are a class of cross-linked polymers whose formation is represented in Figure 1: by cross-linking the polymer chains (long lines), we get a network (long lines connected by junctions) that is able to contain plenty of water molecules in its structure.

Figure 1: Schematic representation of cross-linking process
The crosslinking process can be achieved either by chemical or physical methods, depending on the interaction among the macromolecules of the final network. Chemical hydrogels feature strong interaction in the structure due to the formation of covalent bonds (permanent junctions), whereas in physical hydrogels macromolecules are connected by hydrogen bondings, ionic interactions etc (transient junctions). In any case, either of these types of junctions is able to make the hydrogel insoluble in an aqueous system.3 Biological fluids can be included in the structure as well.
A cool possibility that hydrogels offer, is the immobilization of enzymes in their structure; that is carried out by actually trapping the enzyme in the hydrogel network. In that case, the final result is a heterogeneous catalyst that can be easily separated from reaction mixtures by mechanical methods (such as filtration). Because the enzyme is a catalyst, it is found unchanged at the end of such a procedure. As a result, the hydrogel/enzyme system can be used for multiple runs. Therefore, immobilization of enzymes can be economically suitable for industrial applications; in addition, the immobilized enzymes are more stable than the free enzyme and has activity in several denaturing agents.4
I thought it would be cool to immobilize some lactase enzyme onto calcium alginate beads and investigate its ability to hydrolyze lactose. In the digestive system, lactose is normally broken down enzymatically into glucose and galactose (Figure 2) the latter of which can easily be detected by using glucose test strips. These monosaccharides are subsequently metabolized by the body. If lactase enzyme is not produced by the organism, lactose cannot be digested causing diarrhea and intestinal problems. It seems that lactose intolerance is a widespread problem all over the world (here in Italy, over 50% of the population suffers from lactose intolerance).
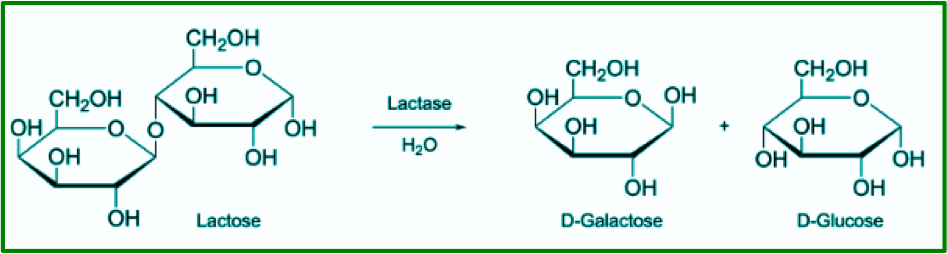
Figure 2: Hydrolysis of lactose by Lactase enzyme
By using glucose test strips the concentration of the monosaccharide can be quickly estimated by looking at the color of the strip after its contact with the processed milk.
EQUIPMENT
- Glucose test strips
- Lactase enzyme
- Milk
- 2% sodium alginate (2 grams of that slowly added and stirred into 100 ml of water)
- 1% calcium chloride (1 gram of that mixed into 100 ml of water)
- Plastic pipette (alternatively, a syringe is perfect)
- 60 disposable syringe, a flexible pipe and a clip
Figure 3: Milk, lactase enzyme and glucose test strips
EXPERIMENTAL RESULTS
In this experiment, a physical cross-linking process was conducted. The procedure I followed doesn’t involve the use of any hazardous chemicals.
Calcium alginate beads were obtained following the procedure described in the JChemEd article I cited previously.1 In general, a 2% sodium alginate (2 grams of that slowly added and stirred into 100 ml of water) and a 1% calcium chloride (1 gram of that mixed into 100 ml of water) solution are adequate for conducting the experiment. I’ve been quite lucky to have these chemicals (from Sigma-Aldrich) already available in the lab but you can use any commercial version of them. They are often sold together since they are used in the field of “molecular gastronomy” in order to carry out a process known as Spherification.5 Lactase enzyme and glucose test strips were purchased online (see Figure 3). In some procedures, even an antacid such as Gaviscon® is suggested as a source of sodium alginate.6
Of course, you can use any kind of lactase enzyme supply (I ran a similar experiment in the past and I used a different brand with no qualitative differences in terms of glucose production). In Italy, all these substances, except for Gaviscon®, are not normally available in regular supermarkets; it is very easy to get them online though and I think they are easily found locally in the United States.
The preparation of the sodium alginate solution is a quite long procedure. To do that, I added 2 grams of sodium alginate powder into 100 ml of distilled water. At least 3 hours of vigorous mixing are required to get a proper solution that results in a dark-yellow kind of color. Once the solution was ready, I added the content of 4 pills (they correspond to 20000 enzyme units) of lactase enzyme to that. Since the pills contain other things along with the enzyme (fillers and excipients), the resulting mixture was quite cloudy. I do think that a commercial lactase enzyme solution would be even better (I would use Lactaid® Enzyme Drops or something like that) but I used the pills because of their minor cost. The mixture was vigorously stirred for 30 minutes; that was added drop by drop (by using either a pipette or a syringe) to a previously prepared calcium chloride solution (prepared by adding 1 gram of CaCl2 in 100 ml of distilled water). The contact between the two substances, immediately gave rise to calcium alginate-lactase beads (Figure 4, left). That procedure allowed to trap and immobilize the enzyme in the beads that became, as said before, a catalytic support the lactose contained in the milk can interact with. When the addition of sodium alginate was completed, I moved to the second step which involved a simple filtration and washing with distilled water of the obtained beads (Figure 4, right). That step was necessary to remove the calcium chloride, fillers/excipients contained in the pills as well as the enzyme that was not trapped in the beads.

Figure 4: From left to right: Calcium alginate beads before and after filtration/washing step
The beads were used to pack up a 60 ml syringe (a procedure which reminded me the packing of a chromatography column). See the final set-up in Figure 5.

Figure 5: Syringe packed with loaded enzyme/calcium alginate beads
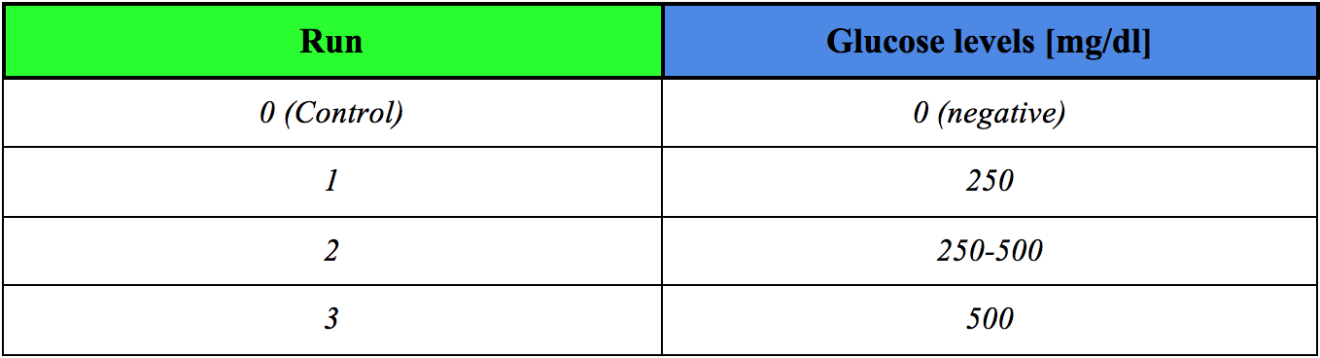
The procedure I followed afterward was very easy: the milk was poured in the column from the top of that, keeping the clip closed. When all the beads were covered by the milk, I let the system sit for 90 seconds in order to make sure there was a good contact between the beads and the liquid. After that period of time, the milk was drained into a beaker by opening the white clip. The final glucose level was recorded by dipping the glucose test strips in the milk for 60 seconds; after that, the collected milk was poured again into the column and the procedure was repeated. I ran the process three times (see Figure 6 and Table 1 for results).

Figure 6: Left: Glucose test strips before (Control) and after (Runs) lactose hydrolysis. Right: Relation between colors and glucose levels
By looking at the color of the strips, we can say that the glucose levels eventually increased. The color of the Control strip, indicated that there was no glucose in the fresh milk. On the other hand, the colors of the First, Second and Third runs strips are different from the Control one. That means that hydrolyzation of lactose took place as the milk reacted with the beads in the syringe.
Table 1: Glucose concentration in the milk depending on the performed run
I’m quite sure that the final glucose levels can be increased by keeping the milk in contact with the beads for a longer period of time as well as stirring the mixture. Although I was curious to, I didn’t taste the processed milk but I thought it would taste sweeter because of the higher glucose levels (I don’t really suggest you taste the milk either!) Of course, this experiment is just a quick and qualitative evaluation of how lactase can be immobilized and used; in any case, I think it may be a good introduction to enzymes and how they can be used in an industrial kind of setting.
If you’re not interested in completing the immobilization described, just the formation of alginate beads is still a cool demonstration to do.7 Very nice results can be achieved by adding some drops of food dyes into the alginate solution. The reaction with the calcium chloride will give rise to colored beads and a stirring bar would create a nice “colored beads tornado”.7
As usual, I’m open to comments and suggestions about this experiment. I hope you will let me know how it goes if you try it out in your own chemistry lab!
ACKNOWLEDGEMENTS
I wish to thank the reviewers of this manuscript for their suggestions and advices.
REFERENCES
1. Pignolet, L.H., Waldman, A.S., Schechinger, L., Govindarajoo, G., Nowick, J.S., Labuza, T., The Alginate Demonstration: Polymers, Food Science, and Ion Exchange, Journal of Chemical Education • Vol. 75 No. 11 November 1998, 1430.
2. Dziezak, J. D. Food Technol. 1991, 45(3), 115–132.
3. American Journal of Polymer Science 2014, 4(2): 25-31.
4. React. Funct. Polym. 71: 104-108.
5. Sperification, Wikepedia, https://en.wikipedia.org/wiki/Spherification (accessed 1/6/19).
6. Steven Spangler TV, Gaviscon Worms - Cool Science Experiment, https://www.youtube.com/watch?v=vAbCEYMfM5M (accessed 1/6/19).
7. Chemical Snakes Worksheet, Dynamic Science Website, http://www.dynamicscience.com.au/tester/solutions1/chemistry/chemicaldemos/alginatewksht.htm (accessed 1/6/19).
Comments
9Lactase enzyme Immobilization, Apparatus Used
Hi Andrea,
Just a quick question on the immobilization of lactase. I have access to powdered lactase enzyme, and am using that for this procedure. I made a 3% solution of sodium alginate, and another 2% solution of calcium chloride. However, for the lactase enzyme, I am a bit confused. Do I simply add the powder lactase enzyme into the sodium alginate solution and stir until the lactase dissolves as well? After that, is it the same process? but then, won't the lactase enzyme be trapped inside the beads? how would it form substrate complexes with lactose samples?
Other than that, I want to know if it would be feasible/practical to use glucose spectrophotometry, along with a calibration curve, to measure the concentration of glucose generated? Data for the calibration curve could be obtained using Qualitative Benedict's Reagent. (I'm asking this because from what I've learned so far, Lactose is a reducing sugar, which would give a positive result when tested using benedict's reagent. But when it breaks down into glucose and galactose, its 'reducing' abilities increase [I assume], which would allow me to produce a large enough range of quantitative data)
Besides that, I would also like to know the technical name for the black and white clip you've attached to the 60ml syringe, and if there are any alternatives to using that?
Thanks a lot, all this information would be really helpful for my investigation!!
In reply to Lactase enzyme Immobilization, Apparatus Used by Darsh G
Lactase enzyme Immobilization
Hello Darsh G,
Yes, you have to add the powdered lactose enzyme into the sodium alginate mix - the latter should be prepared first (sodium alginate solution is quite thick, therefore adding all the substances together would create a suspension which would not be easy to dissolve).
Regarding the first part of your question - yes, the enzyme will be trapped inside and on the beads via crosslinking technique. The principle behind that it's to immobilize the enzyme so that it can used multiple times without dispersion of it in the substrate matrix. Of course, variations on temperature, sodium alginate concentration, pH and other parameters, would improve the entire process (more lactose would be exposed to the action of the enzyme) but for our purposes it's ok to carry it out with the prescribed instructions - it is important the students figure out how the lactase enzyme can be immobilized in order to hydrolyze lactose multiple times without dispersion of the enzyme in the substrate (which would be less convenient in terms of industrial process, since we are adding an extra step to it)
I find the second part of the question really interesting - I didn't try that out, but the UV-Vis spectrometry would be an exciting experiment to carry out. I would collect the absorbance of glucose in its UV range in order to get a more specific data.
Let me know if I answered your question and feel free to add other comments to that!
Andrea
In reply to Lactase enzyme Immobilization by Andrea Amato
Name of the black and white clip
Hi Andrea,
Thanks a lot for your response! I understand the principles of immobilization now!
Yes, you have answered my questions, and thanks again for that, but could you please tell me the name of the 'black and white clip' used to block the syringe? My school's lab assistant can't seem to find anything similar to that, nor have I been able to order it online. I did try out a DIY remedy, but it wasn't nearly as efficient as the clip.
Thanks a lot, Andrea! Your paper has been really helpful and detailed!
In reply to Name of the black and white clip by Darsh G
Name of the clip
Hi Darsh G,
Glad you enjoy the article!!
It's actually an article I am proud of!
I'm sorry but I don't know the name of the clip...when I wrote the article, I was a chemist in a lab which was part of the mechanic section of the industry I was working in. I found the white clip somewhere in the lab and I used that to adjust the flow of the liquid in the syringe (the black part is a portion of a small black plastic tube). It was extremely efficient actually!
Have you tried this solution? (I think the website is in Italian language but the picture is what I would use as an alternative to the white clip)
https://www.fishersci.it/shop/products/hoffmann-stainless-steel-pinch-c…
Hope that helps!
Andrea
White Clip Darsh
Link for clamp at Fisher Scientific: https://www.fishersci.com/shop/products/pinch-clamp-5mm-tubing-100-pk/5…
clip
Industrial Flow Control Hose Clamp
Also at: https://www.amazon.com/Plastic-Resistant-Adjustable-Laboratory-Industri…
Share Your Thoughts