Water Tower Reservoir Analogy of a Buffer
In a previously published work1, I described an activity. In part it goes:
| Movement | Statement |
| Hold the object. | This is a proton. I am an acid. (InstH+) |
| Turn to student and say: | You are a base (Stu). |
| Hand off the "hydrogen ion". | Now you are an acid (StuH+) and I am a base. |
A Brønsted base—in the above case a student—has the capacity to be filled with a hydrogen cation, a proton. The base has "empty" whereas the acid has "full", and by transfer, they "exchange" the empty and the full. Restating a truism from an earlier blog post, "Empty is not nothing, it is the capacity to be filled." That is, empty has a boundary, and this boundary creates a capacity. The concepts "empty" and "filled" both have capacities, and both exist in their potential. Empty is not empty unless it can become filled, and filled is not filled unless it can become empty. In this way we see that both the Brønsted acid and base exist in their potential to become their conjugates.
After that short and active philosophical introduction to acid-base chemistry, we come to the Brønsted Acid-Base buffer system. Here we move from the molecular level acid/filled, base/empty to the macroscopic equilibrium level description. In a simple acid-base equilibrium, the proton donor and acceptor are a conjugate acid/base pair that exist together, and from this coexistence, we can take advantage of each characteristic—proton donation and accepting—to temper the effects of adding and removing hydrogen ions.
What is a buffer?
A buffer is described by three parameters:
1. the combined number of moles of the weak acid and its conjugate base -> The larger the amount (moles) of weak acid, the more of a strong base can react with it, and correspondingly, the larger the amount (moles) of weak base, the more of a strong acid can react with it. Thus, the higher the concentrations of weak acid and conjugate base, the stronger the buffer, i.e., the more resistant it is to pH change upon addition of strong acid or strong base.
2. the strength of the weak acid -> The larger the Ka, the more acidic the compound, and thus the easier it is for the acid to lose a proton. Correspondingly, the larger the Ka, the less basic the conjugate base of the compound. Ka is the state function that describes hydronium concentration space that the weak acid and its conjugate base can play.
3. the ratio between [conjugate acid]/[conjugate base] -> The conjugate base in a buffer is a place for protons to bind. As stated above, the lack of a proton on a base is not "nothing", but the potential for a reaction. So, as we combine weak acid and its conjugate base, we combine places with protons that can be donated (acid) and places where protons can go (base). When the ratio between acid and base forms of a weak acid/base pair is greater than 1, then there are more protons to donate than places for protons to go. With this fine tuning, we can use the Ka expression and the definition of pH (and analogously pKa) to obtain the Henderson-Hasselbach Equation defined below:
pH = pKa + log([base]/[acid])
A curiosity of history and electronics make the most commonly used parameter of hydrogen concentration, pH, decrease logarithmically with increasing [H3O+]. One consequence is that in deriving the above equation, the ratio is [base]/[acid], that is [empty of protons]/[filled with protons]. This will be useful to the following analogy because empty things are typically above full. Also, any buffer made will have a [H3O+] that is ⅒ - 10 times the Ka, and thus a pH ±1 of the pKa.
To sum up, the condition of acid/base co-existence in a buffer can be described by proton filled vs. proton empty.
- Proton-filled is the acid capacity.
- Proton-empty is the base capacity.
- Transferring from proton-filled to proton-empty is an acid-base reaction.
- The buffer capacity depends upon the total amount of filled and empty: conjugate acid and base.
- The more proton-filled (acid) you have, the more can be emptied (reacted), the higher the acid capacity.
- The more proton-empty (base) you have, the more can be filled (reacted), the higher the base capacity.
- A system with both a large empty and full capacities has more of an ability to respond to either of the stressors, emptying or filling
Buffer: A Reservoir Analogy
Extending this to a Reservoir Analogy (image below) uses the empty/filled concepts to help us comprehend the workings of a buffer. A reservoir is a structure to hold (i.e., reserve) water for future use. A lake behind a dam or a water tower would be two examples. A reservoir is defined by its: A. capacity, B. useful range and C. fill-factor. These three parameters for a water reservoir are directly analogous to the three given for a buffer, except a buffer holds protons, not water.
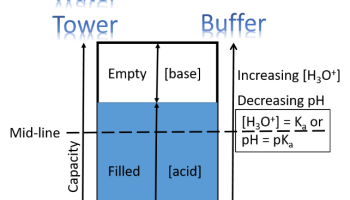
Figure below = Water Tower Analogy of a Buffer. The three factors 1) Capacity, 2) Mid-line and 3) Fill-Factor define both.
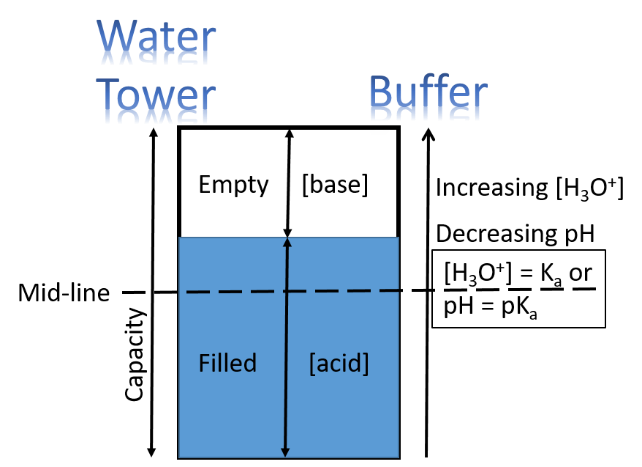
A. Capacity
In developing our analogy, we imagine a water tower as our reservoir because its size and height are obvious, even though the level it is filled to is less obvious than a lake reservoir (Perhaps we can imagine a glass-sided water tower.). The water tower capacity is set, just like a buffer. It can only be filled so far before it overflows and emptied so much before it is drained. It is the same with a buffer- only so many moles of strong acid can be added before it “overflows” with protons, and it has so many protons to be "emptied" by reaction of strong base.
|
|
Capacity |
Useful Range |
Fill-Factor |
|
weak acid/base buffer |
moles: acid + base |
⅒ * Ka < H3O+ < 10* Ka or log(Ka) ±1 |
[base]/[acid] |
|
water tower |
water tower volume Capacity = empty + full |
From nearly the bottom to nearly the top of the tower |
Ratio: Empty to Full |
B. Useful Range
Gravitational potential (height of the tank) is a common analogy tool for energetics and thermodynamics. Just as the ultimate water pressure of a system is mostly determined by the height of the tower above the user (see Note below), so the "proton pressure" of a reaction is mostly determined by how acidic the acid is. The altitude of the Mid-line of the water tower (see Water Tower figure) is inherent to the structure, and the difference in altitude to the user can give a useful estimate of the ultimate water pressure. For purposes of our analogy, we are going to have to assume that all users are at the same level.
Note: The typical municipal water supply should be at between 50-100 psi (pounds per square inch) (https://people.howstuffworks.com/water.htm). For each foot, water supplies 0.4278 psi. That would mean the water supply should be 117-234 feet above the user. A typical tower is placed on a hill which gives some height, and the tower itself provides the remaining height.
C. Fill-Factor
People, in general, are less concerned with the fine points of the pressure of the water coming out of their taps than that there should be water coming out of their taps. Analogously, people, in general are less concerned with the precise pH of a buffer and more concerned that the buffer keeps the pH within an allowed range. So, to continue the water tank analogy, as long as the tank has some water (but not overflowing) and is delivering it with sufficient pressure, though certainly the differences in pressure can be measured, no one much cares whether the pressure is a little higher or lower. However, knowing and predicting the precise pH is very important, and so the adjustment of pH by changes in [base]/[acid] ratio is subtle, yet important. For example, biological systems can be exquisitely sensitive to changes in pH, so the subtleties of fill factor and its relationship to pH need to be quantified, and our analogy breaks down a little in quantification.
Finally, changes in the proton Fill-Factor of buffers are a challenge to calculate. Typically, the Capacity (moles: acid + base) is fixed, and a strong acid or strong base is added to shift the Fill-Factor. The strong acid both increases the weak acid and decreases the conjugate base (strong acid addition) while the strong base does the reverse. If you keep in mind the water tank analogy, the change in concentration of the acid must equal the negative of the change of concentration of the base, just as you amount you fill the tank must equal the negative of the amount you reduce the empty portion.
The Ubiquitous PowerPoint Presentation
As is common in my posts, we have a PowerPoint presentation with a little bit of animation. I would encourage you to open the included PPT file, Reservoir Analogies-Acid-Base.pptx. The first three slides after the title deal with the general discussion of Brønsted acid-base chemistry, most of which is given above. The four animated slides illustrate:
1) a complete strong acid/weak base reaction,
2) a complete strong base/weak acid reaction,
3) a buffered, partial strong acid/weak base reaction, and finally
4) a buffered, partial strong base/weak acid reaction.
In the first PPT illustration, the unfilled acetic acid/acetate reservoir is unfilled and is the same size (1:1 mole ratio between the two reservoirs) as the proton-filled HCl/Cl- reservoir. The latter is higher on the page reflecting the greater "proton pressure" that the former system has, and in fact the system lies at the "height" of a hydronium ion. Like any strong acid (opportunity alert: this illustrates the leveling effect), in an HCl/Cl- aqueous system, any protons transferred from a weak acid the system will go directly to water, not Cl- because Cl- is has so little basicity, it has an unmeasurable hold on hydrogen ions in water. This is another way of saying both: 1) HCl is a strong acid, and 2) HCl/Cl- aqueous solution cannot be considered a buffer.
Clicking through the PPT slide will eventually cause the valve to turn and the protons to drain from the HCl to the acetate for the reaction. Note that as the reaction proceeds, as if we were watching a titration and monitoring the pH in the acetic acid/acetate beaker, the height in the acetic acid/acetate reservoir increases. This is an analogous, but by no means quantitative, view of decreasing pH in the acetic acid/acetate beaker.
The next slide does the complementary reaction with the filled acetic acid/acetate beaker (acetic acid) with an empty water/NaOH beaker. The "height" of the latter is for that of water, and though rarely discussed, there is a leveling effect for bases more basic than the hydroxide. Complementary to the HCl/Cl- system, the water/NaOH system is no buffer, but we can see the progress of the acetic acid/acetate buffer as the acid is drained of protons with the qualitative increase in pH.
The final two slides start with the acetate half-filled with protons (that is, 1:1 ratio of acetic acid:acetate). This would be a buffer that has pH = pKa. This is considered the "best buffer" for any particular buffer capacity because it has equal amount of filled and empty to lose and accept protons. At this point the effect of capacity (total acetate moles or "tank size") can be mentioned. This is perhaps the best vocal-visual of this blog.
The first of these final two slides adds a sub-stoichiometric amount of HCl to react with the acetate. Though somewhat redundant to the very first slide with its Wipe motion filling the fully empty beaker, it is nice to have an illustration that starts with the buffer and shows the modest increase in proton pressure upon addition of the strong acid. For those viewing this as part of a titration, this would be moving from the ½ titration point forward a bit.
The very last slide shows the addition of a sub-stoichiometric amount of NaOH to react with the acetic acid. Some of the protons with higher proton pressure drain to the unfilled hydroxide to make some water. With the Reservoir Analogy, it becomes obvious that the effect of strong base moves the buffer system modestly towards a more basic condition.
Wrap-up
As analogies go, this covers a lot of ground. And the concept of transfer can be seen at every point, and transfer implies the fundamental concept of chemistry—making and breaking bonds. Using a reservoir brings in both energetics and stoichiometry without using those particular words. I have used the reservoir analogy in the electron transfer of redox chemistry as well. This being said, I have been humbled in my attempt to complete the animation for a back titration with two redox reaction steps. In conclusion, I hope the Gentle Reader will have the capacity to transfer knowledge of acid/base chemistry to fill minds of their students.
(1) "Kinetic Classroom: Acid-Base and Redox Demonstrations with Student Movement", Joseph F. Lomax, J. Chem. Educ. 1994, 71, 5, 428-430.