Q, K, ∆G and ∆G°: The Darlings of Thermodynamics
Q, K, ∆G and ∆G°. In two connected ways, thermodynamics describes how both physical or chemical processes spontaneously trend towards equilibrium. In the first, the reaction quotient, Q, trends towards the equilibrium constant, K. In the other, the free energy of reaction, ΔGrxn, trends towards a value of zero. These two are connected by the equation: ΔGrxn = ΔG°rxn + RT ln(Q). Connecting this equation to the defining a condition at equilibrium, that when K = Q, and ΔGrxn = 0, one derives: ΔG°rxn = -RT ln(K).
My, what a mess of terms and definitions.
- ΔGrxn is the free energy of a reaction and describes whether the reaction spontaneously trends towards the products or reactants
- ΔG°rxn is ΔGrxn under standard state conditions
- R is the familiar Universal Ideal Gas Constant from PV = nRT
- T is absolute temperature from the same equation, and
- Q is the mass action reaction quotient
These definitions have not gotten us, or perhaps the student being introduced to them, much further towards understanding because we would now need to define "spontaneous" and "mass action" among other things. But, as an you are an instructor, I am figuring you can do that at your leisure. What this blog post is concerned with is showing how free energy (ΔGrxn) changes as the reaction quotient (Q) changes as both trend towards the special condition, equilibrium, described by the constant K.
To begin,...
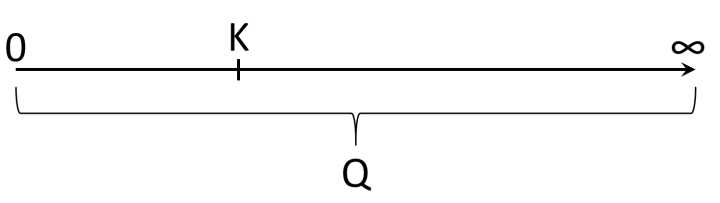
The reaction quotient, Q, is a value derived from the Law of Mass Action, and is often summarized as "Products over Reactants". This discussion assumes you have taken the time to define Q, and I thank you for doing that. The Law of Mass Action is derived from kinetics at equilibrium, but Q can be used away from equilibrium. As shown in the image above, Q can have any value on a number ray starting at zero and going to positive infinity. K is a special point on this line which is at equilibrium.
The reaction condition as described by Q is attracted to the equilibrium condition as described by K. Conventionally, reactions proceed on the page from left to right, and the reaction going from right to left is the 'reverse reaction'. It isn't so much going backwards in space or time, but the compounds written on the left are transformed into the compounds written on the right. K has a particular value and if Q is lower, the reaction causes Q to increase; if Q is larger than K, the reaction causes Q to decrease. Get that?
How does one instruct students to keep the information in the paragraph above straight? It is simple- put K before Q. With K written before Q, put the inequality or equal sign in between. Now, follow the (in)equality.
- K > Q follows the pointy part of the inequality from left to right towards the products.
- K < Q follows the pointy part of the inequality from right to left towards the reactants.
- K = Q is balanced at equilibrium.
It is that simple, yet textbooks almost pathologically put Q before K and then explain in words the direction of the reaction which is against the flow of the inequality. For example, when Q > K, the reaction trends towards the left, towards the reactants.1 There are only two ways to do it and the one that follows the chemistry is "K before Q". As a goofy pneumonic I call K the AttraKtor, and in the animated pptx file have a TraKtor follow the direction. If this is too kitschy for you, I understand, and you can edit out the image when using the pptx.

Why does this work? What attracts from each of two directions? As an instructor, it is always useful to think about what would make a good analogy. In this example, for reaction spontaneity towards equilibrium, what works? A magnet spontaneously aligns and attracts, but this is a poor analogy because describing the direction of the reaction as poles on a magnet get very confusing as the frame of reference changes as the magnet flips around to become North-South aligned. There are similar issues with using charge or dipole as the basis for an analogy of Q and K. The analogy that is most common with chemists, and for good reason, is with gravitational potential.
Chemists will describe a reaction as downhill for spontaneous and uphill for non-spontaneous. Heaven help us from the literalist who might want to create a lab with a sloping floor to induce desired reactions to occur in one physical direction. But downhill and uphill are good analogies because the basis behind gravity and chemical reactions both deal with potential energy. And potential energy trends to a minimum from whichever direction you come from. With gravity, it is the bottom of a hill and with chemical potential it is at equilibrium.
How does this play out with the Q number line? As we know, a picture paints a thousand words, and as I want to reduce the words and increase understanding. To do so see image below (= slide 4 in PowerPoint file, Supporting Information).
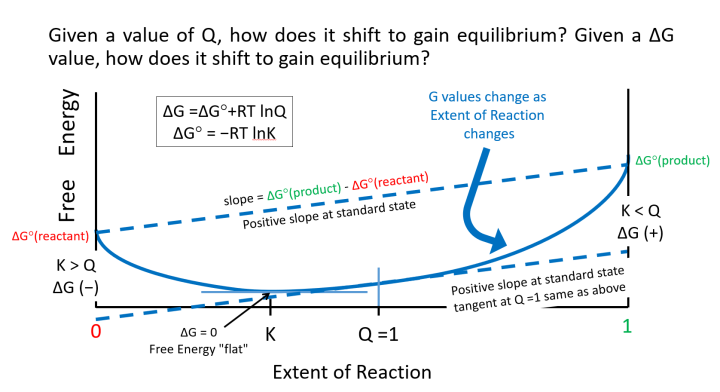
As a graphical convenience (who can graph infinity?), we change the x-axis to Extent of Reaction which goes from 0 for the beginning (or no products) to 1 for the end (or no reactants). The y-axis is going to be in Free Energy, G, which is chemical potential energy. When you get to electrochemistry, you will find that it is directly related to the electromotive force, ℰ, or electrochemical potential. The free energy surface is sort of a sagged line being held at:
1) 1 on the x-axis (Extent of Reaction: all products) and at ΔG°(products) on the free energy/y-axis, and
2) 0 on the x-axis (Extent of Reaction: all reactants) and at ΔG°(reactants) on the free energy/y-axis.
The math describing this free energy function does not lend itself easily to a spreadsheet, except for something like an isomerization that has a small ΔG°rxn. So, as I am using PowerPoint with its limited curved line shaping tools, I find it necessary to describe the surface as being described by a "magic rope" held by the "posts" of the Extent of Reaction, 0 and 1 respectively.
You can use a physical rope in class to show much of the behavior of equilibrium constants. Particularly, how the surface always slopes towards a minimum from either Extent of Reaction = 0 or 1, and how the minimum shifts as you change the relative heights (potential energies) of reactants and products. We see in the image above, that much like a rope that is tethered at two uneven heights, it sags down from the posts and a one point between the extremes; it is level. This level point is the equilibrium and there is a value of K that corresponds to this. If the reaction is uphill, the level point is towards the reactants, and if it is downhill, it is towards the products.
The other special point on the free energy surface is where Q = 1, that is at standard state. If the "magic rope" works correctly, the slope of the tangent at Q = 1 will be the ΔG°rxn. Again, PowerPoint lines do not quite curve like our "magic rope", so though I have Q = 1 at the point where the tangent on the surface is the same as ΔG°rxn, Q = 1 is not precisely in the middle between Extent of Reaction = 0 and 1.
Finally, we can follow the sign on ΔG by following the slope of the free energy surface. Starting with all reactants, the slope is negative and you move forward on the graph, getting shallower and shallower until it goes flat at the equilibrium. Correspondingly, starting at all products, the slope is negative and you move backward on the graph, getting shallower and shallower until it, again, goes flat at the equilibrium. At equilibrium, towards reactants or products, is uphill, so there is no driving force to change the extent of reaction, so the extent changes not.
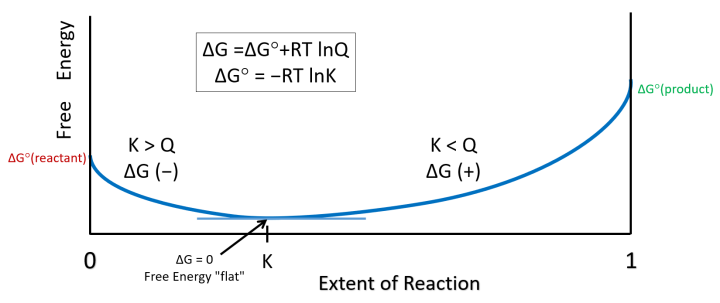
As a little added feature for those who find the image above (= slide 4 in the PowerPoint) a bit busy, a cleaner version is below (= slide 5 in PowerPoint).
For those of you keeping score, the next blog will be on Acid/Base equilibria and will try to connect proton transfer to our friend from today, Free Energy. I hope to see you there.
Added feature/supporting information: The static images in this post are animated in the PowerPoint. If you find the discussion above useful, it makes a good chalk talk, or if PowerPoint is your style, I encourage you to copy/paste the into your PowerPoint presentation. The heading of the first content slide is restating one of the Learning Objectives we have on this topic, as is the heading on the 3rd content slide.
1 I have a dozen textbooks on my shelf, and if an inequality is shown, all are set in the order of Q before K. There may be a good mathematical cultural explanation for this, but I contend that good chemical pedagogy override mathematical convention. If you can find a textbook that has it in the other order, please put it in the Comments of this blog.