Naming and Formula Writing for Ionic Compounds Virtual Activity

Writing formulas is one of those concepts in chemistry that requires much practice and repetition for students to gain confidence. Some issues students find challenging when learning to write formulas are identifying when a roman number is needed and what the value of that numeral should be, how and when to use prefixes, when are parentheses needed and what the suffix of a compound name should be.
In a non COVID world, to help students practice writing formulas, I would have students complete an activity where they roll two dice, one with a variety of cations on it and another with various anions on it. The goal was to combine the two dice to create an ionic compound. Students would then write the formula for the ionic compound and the corresponding name. It was a fun way for students to practice formula writing yet wasn’t daunting or seem as boring to a student as a worksheet full of practice questions. This year due to restrictions in sharing lab materials a digital alternative was needed. Jessica Mintz (@mintzchemistry) has created a version of the activity that will work in a remote world. The lesson should take about forty minutes.
Download a copy of the following procedure to your Google Drive: Names and Formulas Activity Google Doc
If you use Google Classroom, create a copy for every student when you assign the activity. If you do not use Google Classroom, you can force a copy for students.
Materials
Share the Google Doc with students.
Background
Procedure
Figure 1: TeacherLed.com's dice option
Students will roll dice virtually using a website for digital dice. They need to choose the pink and blue dice from the menu at the top of the screen to match the colors in the Dice Key. After each roll they will match the values of the dice to the key and record the cation and anion in the table provided (see figure 4). They will consider the ratio of ions and write the chemical formula. (They can use SymbolSalad.com for subscripts and superscripts.) Then students will record the name of the compound. An example is provided in the Google Doc. Students need to record 10 different chemical formulas using the dice.
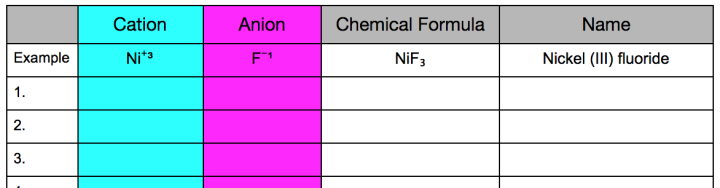
Figure 3: Data table for recording results
Questions
The following questions are included in Google Doc of the activity as referenced above.
- Explain, in terms of electrons, what a positive ion indicates.
- Explain, in terms of electrons, what a negative ion indicates.
- Explain why ionic compounds are “fixed” in their chemical composition.
- Why do some ionic compounds contain a roman numeral?
- If element X combines with oxygen to form an ionic compound X2O , what is the charge of element X? What Group does X belong to on the Periodic Table?
Concepts
Time Required
40 minutes
Preparation
Provide students access to the activity Google Doc noted above.
Attribution
Thanks to Jessica Mintz (@mintzchemistry) for revising my Names and Formulas dice activity into a virtual activity.
Credits
This activity uses the TeacherLed.com Dice option (https://www.teacherled.com/iresources/tools/dice/). It also uses the SymbolSalad.com created by @KevPluck (https://symbolsalad.com/).
Comments
5Thank you!
I'm getting ready to teach nomenclature and formulas right after Christmas break and this is perfect for what I need. I have usually done a "Bond with a Classmate" activity where each student is an ion and they have to wander the room to find an ion they can bond with...obviousy can't do that this year, so this is a perfect substitute. Thank you!
Subscripts
This is a great addition. I actually just did something similar with my class yesterday since we are back F2F, but this would have been helpful when we were all virtual. I'm going to still incorporate it as a review activity. I just wanted to add that you can make subscripts & superscripts in Google docs. The keyboard shortcuts are [ctrl+,] and [ctrl+.] Thank you!
Share Your Thoughts