Determination of Citric Acid in Candies

Citric acid is a weak tricarboxylic organic acid. It is highly soluble in water and, once it dissolves in that, it shows weak acidity but a strongly acidic taste which affects sweetness and provides a fruity tartness for which it is widely used to complement fruit flavors in the food and beverage industry. It is also used to mask the unpleasant taste of pharmaceuticals.1
However, a high content of citric acid in foods and drinks could damage your teeth. Chemical erosion of the teeth occurs either by the hydrogen ion derived from the acids or by anions which can bind or complex calcium. Citric acid seems to be quite aggressive since it can release three moles of H+ ions for every one mole of acid; each ion is able to attack the tooth mineral crystal leading to direct surface etching.2
Since various acids are added to food products to boost flavor, reading the food label may be the best way to determine the acid content yet this information is 99% absent. Usually it is indicated that some acids are present but the concentration of them is not. You can determine those values with some chemistry though! Compound Interest created a cool infographic about the nature and properties of acid contained in foods and drink.
Oftentimes, acid-base titration labs are carried out using vinegar as the acid to be titrated. Sometimes this activity is also used to conduct an IA (Internal assessment) for the IB (International Baccalaureate) program. A common activity is to titrate several different vinegars to find the concentration of acetic acid in each brand. The data collected is analyzed and compared. The activity is not that impressive but requires accuracy and the elaboration of data.
My colleague, William, and I were thinking about creating an original activity to engage students in a titration lab. Actually, a lot of different acids beside vinegar can be found at home. If the acid is contained in a solid matrix, it is necessary to get the acid out of that matrix in order to obtain a solution which can be quickly titrated. We considered many of those other acids before asking “Why don’t we extract the acid out of candy?” In general, citric acid is used as a preservative in food, drinks and, of course, candy. It is also denoted by E number (code for substances that are permitted to be used as a food additive in the EU) E3303. A more detailed description of this compound is available in the Journal of Chemical Education4 and on PubChem5. In a chemistry lab full of teenage students, it is not difficult to find a pack of candies and most of them contain citric acid! We started out with Mentos Now which we used to illustrate how you can titrate citric acid from candy.

Figure 1 - Equipment used for the titration (dark bottle contains Thymol blue indicator.
NOTE: See the Student and Teacher Documents provided below as Supporting Information.
1 - Chemistry Central Journal (2017) 11:22
2 - Monogr Oral Sci. Basel, Karger, 2006, vol 20, pp 66–76
3 - https://en.wikipedia.org/wiki/E_number
4 - J. Chem. Educ., CLIP (Chemical Laboratory Information Profile, 2003, 80 (5), p 480 (accessed 3/25/2018)
5 - https://pubchem.ncbi.nlm.nih.gov/compound/citric_acid#section=MeSH-Entr… (accessed 3/25/2018)
Materials
Procedure 1:
- Distilled water
- NaOH pellets
- Potassium hydrogen phthalate
- 100 ml volumetric flask
- 250 ml Erlenmeyer flask
- Phenolphthalein indicator
- Burette
Procedure 2:
- Candy (they must contain citric acid)
- Balance Mortar and pestle
- Magnetic stirrer
- 250 ml Erlenmeyer flasks
- Buchner funnel and filter paper
- Burette
- Thymol blue indicator
- 0.01 M standardized NaOH solution
Background
Procedure
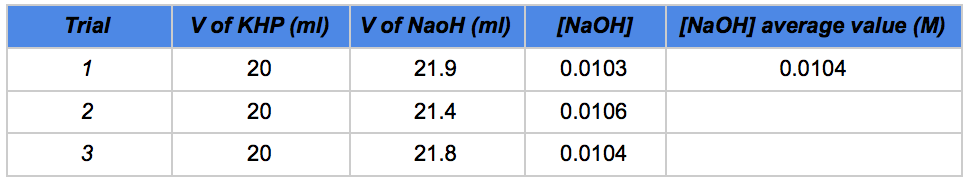
In order to have an adequate amount of citric acid which can also be titrated in a reasonable amount of time, we had to experiment a little bit. We decided that 5 g of Mentos Now tablets was a good option to start with. Candies are crushed in a mortar in order to maximize the possibility to extract the citric acid out of them (Fig. 2). The powder is dissolved in 100 ml of distilled water and the mixture is vigorously stirred for 30 minutes. A standardized solution of 0.01 M sodium hydroxide (made using the classic standardization method: 0.01 M KHP is prepared to determine the exact concentration of the NaOH solution). A 500 ml solution of NaOH is prepared by massing the required amount of base which gives a concentration of 0.01 M. Phenolphthalein is the indicator used to carry out the titration.
Figure 2 - Mortar and pestle is used to crunch up the Mentos Now candies.
Once the solution of NaOH is prepared, it is possible to move on to the filtration of the previously prepared Mentos Now candies. In order to maximize the speed of the filtration step, a vacuum filtration is recommended. Most of the particles are small enough to pass through the filter paper though, so it is a good idea to filter the solution with two filter papers. Filtration is obviously slower but the solution will be less cloudy. The small amount of cloudiness remaining after filtering with two papers is not an issue. The final result is shown in Fig. 3 below (as you can see, solution is not totally transparent).

Figure 3 - Solution of Citric Acid from Mentos Now candy.
Set up a burette holder on a stand; wash the burette with a small amount of standardized NaOH (at least for 3 times) and you are almost ready to start the titration! Pour 20 ml of the citric acid solution into an Erlenmeyer flask and add a couple of drops of thymol blue indicator. We found out that thymol blue is the best option as an indicator for this activity. (See the teacher handout for more detail.) In fact, when the titration is almost complete the solution turns green and, after, blue which represents the actual endpoint (Fig. 4).

Figure 4 - From left to right thymol blue at the beginning of the titration (yellow), immediately before (green) and at the endpoint (blue).
The reaction goes to completion and is as follows: C6H8O7 + 3NaOH --> C6H5O7Na3 + 3H2O.(Some teachers use the organic formula C3H5O(COO)3H3rather than C6H8O7.)
See the "student document" included as supporting information below for complete procedure.
Questions
Concepts
laboratory experiments, titration, candies, general chemistry, thymol blue, citric acid, analytical chemistry
Time Required
One 50 minute class period if the instructor prepares the standardized NaOH.
Preparation
The teacher may want to standardize the NaOH instead of having students do it. A standardized solution of 0.01 M sodium hydroxide (made using the classic standardization method: 0.01 M KHP) is prepared to determine the exact concentration of the NaOH solution). A 500 ml solution of NaOH is prepared by massing the required amount of base which gives a concentration of 0.01 M. See the supporting information below for complete procedure.
Table 1 - Standardization of sodium hydroxide solution
Attribution
Co-authored by William Weisberger.
Credits
General
Items tagged with the safety:general tag have some safety concerns that may be also described more specifically by additional safety terms
General Safety
For Laboratory Work: Please refer to the ACS Guidelines for Chemical Laboratory Safety in Secondary Schools (2016).
For Demonstrations: Please refer to the ACS Division of Chemical Education Safety Guidelines for Chemical Demonstrations.
Other Safety resources
RAMP: Recognize hazards; Assess the risks of hazards; Minimize the risks of hazards; Prepare for emergencies
Comments
8Query
Hello! This was quite helpful!! I want to find the concentration of citric acid on Sour Patch Kids. How do you suggest I do that?
In reply to Query by Megha Seri
Sour Patch kids
Hi there Megha!
Thanks for your comment; I've been quite lucky this summer...I went to the States and I bought a pack of Sour Patch Kids (they're not available here in Italy).
Well, the ingredients list states that they contain citric acid and tartaric acid. Mentos candies only contain citric acid so the acidity of the candies is related to that chemical; since Sour Patches contain two acids, you are getting the concentration of acid which is the sum of the two (citric and tartaric) - it's a sort of "total acidity".
As far as the method you can use, I would go for the procedure I used in this article; of course it's not easy to use mortar and pestle with Sour Patches, so I would use a knife or something like that to get small pieces of the candies. Put them in water and stir for 30 minutes - citric acid and tartaric acid are very soluble in water so they will be extracted easily. In any case, the more you stir the better the extraction will be. Filter the solution and you are ready to carry out the titration!
I hope I answered to your question!
Let me know if that works.
Indicator
Does anyone have a recommendation for another possible indicator to be used with this titration?
Indicator? It is the dose that makes the poison
In the UK we would have no issue with using phenolphthalein (0.5%) as the concentration used is less than 1% above which it has to have a toxic warning. It has been used in medicines to relieve constipation and bowel cancer could be an issue. So you have to drink it. But then if you did you would be drunk from the ethanol being used. You can use thymolphthalein. But if you want to use no indicator you could a pH probe and meter. But countries have own rules so please check.
The final step
Hi if your reading this I am a bit confused on the last step. How do we actually determine the amount of citric acid in the very end after the thymol blue indicator is placed? Do we just measure the mass of the liquid or is there another step? sorry im a bit lost !
Share Your Thoughts