Three-dimensional figure to improve the didactics of the Periodic Table
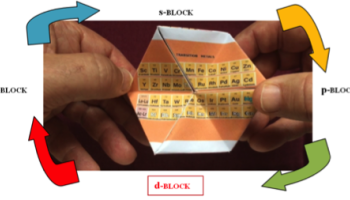
In 2019, the 150th anniversary of the creation of the first Periodic Table established by Dimitri Mendeleiev was commemorated. As high school teachers, we decided to make a figure of the Periodic Table that would be attractive and catching for our chemistry students. For this purpose, we came up with the idea of using a kaleidocycle, which is a three-dimensional figure obtained by folding a flat template.
A kaleidocycle has four different faces, each one made of a juxtaposition of rhombuses. By turning it you can easily choose one of the four faces. On these, four different pictures can be displayed. In this three-dimensional figure of the periodic table are the elements organized in four blocks according to their final electronic structure. It is intended that students with this playful figure actively participate in classes by rotating their kaleidocycle looking for the groups or elements that are being studied. The entire periodic table fits in one palm of their hands. It is also a didactic device because students only focus their attention on one block or group of elements from the entire Periodic Table. It can be a more entertaining, motivating and exciting way of learning about the subject of the Periodic Table. See figure 1.

Figure 1: The four faces of the kaleidocycle
The Kaleidocycle was invented in Princeton University by an English mathematician named Arthur Stone in the 1930s, and later was disclosed to everyone by the famous author Martin Gadner1.
The didactic criterion for this organization was the electronic structure, leaving the elements arranged in the four classic blocks that are obtained when the elements are divided following this feature. As we wanted the figure to be didactic, names of the groups of the representative elements were added and also the most common oxidation number of each of these groups. Also, a clear distinction is made between metallic, semi-metallic and non-metallic elements. Video 1 demonstrates how the kaleidocycle is used.
Video 1: Kaleidocycle of the Periodic Table
Who is the kaleidocycle for?
This device is mainly destined for students in high school who are studying chemistry. It is intended that students make their own figure from the template with the help of the teacher according to the instructions that we will develop in detail below. Each student can use it not only in class but also at home with friends or family members. It is a playful study tool that belongs to each student.
How the figure is made
STEP 1. Print the template seen in figure 2 on thick paper. (You will find both Word and PDF documents of the figure in the Supporting Information below.)
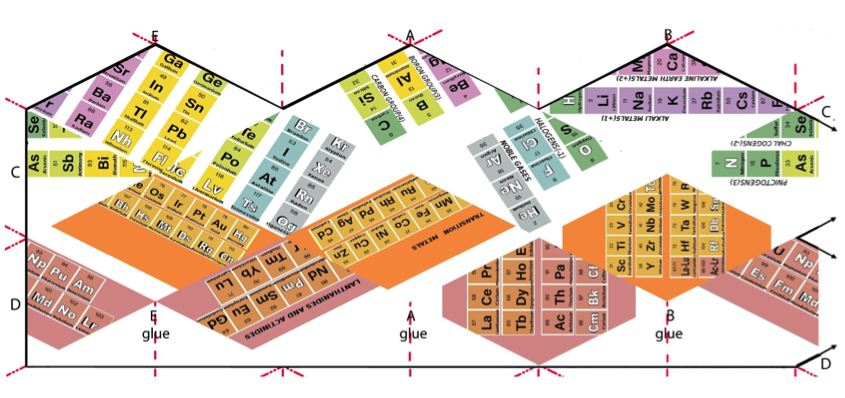
Figure 2: The template of the Kaleidocycle.
STEP 2. It is placed horizontally. Mark (unpainted) vertical and oblique lines (the ends of which are indicated by dashed red lines on the template). The best way to mark the oblique ones is tracing these lines with a pencil from behind, placing the printed figure upside down in a lighted window. See figure 3.
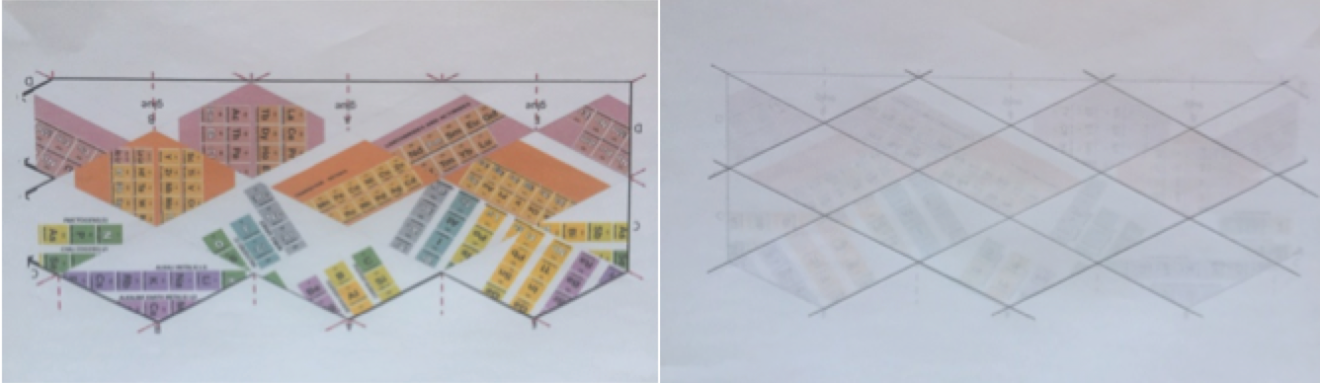
Figure 3: Mark oblique lines on back of template.
STEP 3. The marked vertical lines are bent inwards with the help of a ruler. See figure 4. The figure is cut out where there are bold lines, avoiding as far as possible those lines from being seen.

Figure 4: Bend vertical lines inward.
STEP 4. The diagonals should be bent outward (see figure 5). The best way is turning the figure over and folding the oblique lines that we had drawn in pencil on the window inward. It is better to use a ruler, as in the picture.

Figure 5: Bend diagonal outward.
STEP 5. The figure is forced to have a tubular shape by pushing the two parts marked with the letter A, one towards the other, achieving the shape of a rhombus and the part that says “glue” is glued under the other printed part. See figure 6.

Figure 6: Force to have a tubular shape and glue the two parts marked A.
STEP 6. Tab C moves to edge C, forming a ring. All tab C is glued, making sure that one part is well matched to the other. The same is done with the tab and end D. See figure 7.

Figure 7: Forming the ring.
STEP 7. Glue half of the rhombus marked E to the white triangle E. And finally, do the same with the rhombus B with the white triangle B. See figure 8.

Figure 8: Final connections.
Finally, the letters of the liquid elements are painted with the same color, for example blue (they are only Br and Hg) and those of the gases with another color. We suggest red color (they are only the noble gases, H, N, O, F and Cl).This step can also be done before assembling the figure. The letters of the solid elements are already painted black and the artificial ones already have the letters painted white.
If printing in black and white, most common if the class is large, students are encouraged to paint the boxes for the non-metals with a green fluorescent marker (H, halogens, C, O, S, Se , N, P) and to paint the boxes of the semimetals in yellow with a marker or fluorescent highlighter of that color (B, Si, Ge, As, Sb, Te, Po).
All other boxes left unpainted correspond to metals or noble gases. In the references the video that can be consulted to see the result is cited2.
What is intended to show and learn
The Periodic Table used is freely provided by the Foro Química y Sociedad from Spain3 with their typography and design of chemical symbols.
This original Periodic Table from the Forum has been radically transformed. All names have been translated into English. Its didactic character has been increased by adding the main or most common oxidation numbers of the representative elements, which we consider very useful and didactic. The names of these groups have also been added. In addition, the difference between nonmetals, semimetals and metals4 has been highlighted and the peculiarity of a very stable group of elements which is the group of noble gases has been emphasized out. In the original Table of the Foro Química y Sociedad (FQS), nonmetals in general are not differentiated by color from those that form the exclusive group of noble gases. Therefore, to distinguish them, we change the color of this group of noble gases (from blue we color it to gray). In this way, it is possible to distinguish all the rest of non-metallic elements (blue or green color) from noble gases in the table.
If the kaleidocycle template that we propose in black and white is used, the students themselves have to paint the grids of the semi-metals and those of the nonmetals. We propose to color those of semimetals in yellow and those of nonmetals in green, excluding noble gases.
In addition, the original Table did not indicate the physical state of the elements. To achieve this we had to change the color of many of the symbols in the original FQS Table. The black color was changed to white of all the symbols of all those elements that were not solid. We leave to the students the task of searching in books or on the internet for all liquid, gaseous and synthetic or artificial elements. To unify criteria, we advise them to paint, the letters of the gaseous elements in red, those of the liquids in blue and to leave the synthetics in white color. Thus, it is also achieved that the student, with his contribution, considers this figure as his own. Although this work may seem arduous or tedious, we consider that it is very didactic and it contributes to the kaleidocycle being considered with greater intensity as his own, partly elaborated by himself.
As mentioned above, all the elements were divided into four groups, taking into account the typical division of the elements according to their electronic cortex5. It is insisted that this electronic structure is very relevant because the properties of the chemical elements depend on the end of the electronic configuration6. For this reason, it shows all the elements of the periodic table on each of the faces of the Kaleidocycle which have a similar final electronic structure.
On the first side, the first 4 columns of the representative elements correspond to a final electronic structure with an "s" orbital or with a "p" orbital occupied, as a whole, only by one or two electrons. The four groups are alkali metals with oxidation number +1, alkaline earth metals with oxidation number +2, boron group with common number 3 and carbon group with oxidation number ± 4 (some also have +2).
On a second face of the kaleidocycle are collected all the representative elements that have at the end of their atomic structure more than one "p" orbital with more than two electrons in total (the 4 columns on the right). They are the pnictogens (-3); chalcogens (-2); halogens (-1) and the most stable: noble gases.
On the next side are the transition metals that have a "d" orbital at the end of their electronic configuration.
Finally, on the last side, the lanthanides and actinides (also called earth rare metals) are shown which, as is known, have an electron in an "f" orbital in their final electronic structure.
So, all the 118 elements that make up the periodic table are offered, in an interactive, structured, and manipulation way, but it is such a small figure that it fits in the student's case where they have all their pencils and pens.
To achieve this arrangement, many unsuccessful attempts were made first because we encountered with the difficulty of including all the transition elements on one face.
In addition, we had to be able to show on another side rows as long as those of the internal transition elements (lanthanides and actinides). It was decided to distribute each row of elements, either the lanthanides or the actinides, into two rows of 7 and 8 elements.
We also had to ensure that letters of the symbols were not distorted because they were in the middle of the separations of the three-dimensional figure. Therefore, we had to slightly shift some symbols or some boxes. It must be said that the final result is not a periodic table designed to know details that do not fit such as atomic mass or all oxidation numbers. For that purpose there are other periodic tables with profuse information.
Use and motivation in class
This figure is intended to neutralize or end a passive attitude that students sometimes have in front of the Periodic Table. Everything that contributes to energizing the classes will facilitate both the teacher's approach to the students and the understanding of the subject.
One aspect that increases motivation is that the completion of the table is considered a challenge, since the student has to be able to build it correctly by himself. When the students manage to put it together successfully, they show it and proudly explain it to classmates and family. It should be noted that they have not only carried out this kaleidocycle by themselves, but have also had to complete it previously on their own since they have had to investigate the physical state of the elements to paint the letters of liquids and gases with the appropriate colors. They have also been asked to look for synthetic or artificial elements (that do not have any added color).
With this kaleidocycle, the classes are more didactic. Indeed, since only one of the faces of the figure can be chosen, it is easier to focus or direct students' attention to one of the blocks or to one of the groups, isolating it from all the other elements of the periodic table. The figure helps to focus study and attention on a specific block or group of elements.
Classes in which all students have their kaleidocycle are more active and participatory. Students are not mere passive listeners, but rather have to actively participate by manually moving their kaleidocycle to find the right face in their figure and thus follow the analysis that is carried out of some group or block of the periodic system.
In the classroom, educational and entertaining exercises can be proposed in which the student has to rotate his Kaleidocycle to find the appropriate answer. This actively engages them, highly motivates them to a great extent, and they learn important things about the periodic table in a playful way7. Exercises can be carried out in which they have to find out those elements that meet some characteristic that we consider relevant. To do so, they manipulate or move their kaleidocycle with enthusiasm and high interest. For example, we can ask them to look for elements with similar characteristics to a selected one, or to count the total number of gaseous or liquid elements in each block, or we can ask them questions such as: how many elements are synthetic among the representative elements? Likewise, we can propose quick search exercises for a single element. When they find it, they have to point out on their kaleidocycle, showing the correct face. The name can be said directly or clues can be given such as that it has a certain oxidation number or that its name begins with a certain letter and belongs to this or that group or block.
We have found that certain characteristics of chemical elements are better learned. For example, it helps to better remember what the final electronic structure of each element is like according to the block to which it belongs.
It should be noted that students can carry the figure in a small box or in their cases. They don't need a large sheet to have the entire Periodic Table.
Teachers may consider another possibility which consists in establishing a constructive and open dialogue with the students in which is discussed with them the best criteria of dividing the elements in the kaleidocycle. They are divided or organized, in this figure, according to their electronic configuration. Could it be done according to another criterion in such a way that the elements are divided in an organized way?
Conclusions
This project, “Three-dimensional figure to improve the didactics of the periodic table” “, can help in the didactics of Chemistry in reference to the periodic system. With this kaleidocycle, there is an attractive, motivating and interactive approach to this subject. In addition it can contribute to the study of the Periodic Table of the elements in an organized way.
References
(1) Gadner, M.. Hexaflexagons and Other Mathematical Diversions, Scientific American, MAA press 1988
(2) Cassinello, P. Video of Kaleidocycle and the Periodic System. https://youtu.be/nChlEMFifN8, 11/23/2021.
(3) Foro Química y Sociedad (FQS). Tabla periódica https://www.quimicaysociedad.org/tabla-periodica-de-los-elementos-quimica/
(4) Zumdahl, S.S.; DeCoste D.J.. Introductory Chemistry. Brooks cole Cenguaje learning Boston.. 2011. 345-347
(5) Chang, R; Overby,J. Chemistry, 13th ed.; McGraw-Hill: New York, NY, USA, 2018
(6) Ebbing, D.D; Gammon,S.D. General Chemistry, 11th ed.; Cengage: Boston, MA, USA, 2017.
(7) Holstermann, N., Grube, D. & Bögeholz, S. Hands-on Activities and Their Influence on Students’ Interest. Res Sci Educ 40, 743–757 (2010).