What are the molecular structures of sulfonphthalein acid-base indicators?
Co-Authored by Iain A. Smellie* and Iain L. J. Patterson*
*University of St Andrews, School of Chemistry, North Haugh, St Andrews KY16 9ST, United Kingdom
Many educators working the schools, colleges and universities will be familiar with the use of sulfonphthalein compounds as acid/base indicators.1 Bromothymol blue is a well-known sulfonphthalein indicator. It is often used to determine the end point of acid/base titrations and it can be used to monitor the progress of reactions that produce acidic products.2,3 In aqueous conditions, acidified bromothymol indicator solutions should appear yellow to the human eye. In contrast, when the pH of an aqueous solution is greater than 7.6, the solution will appear blue.4
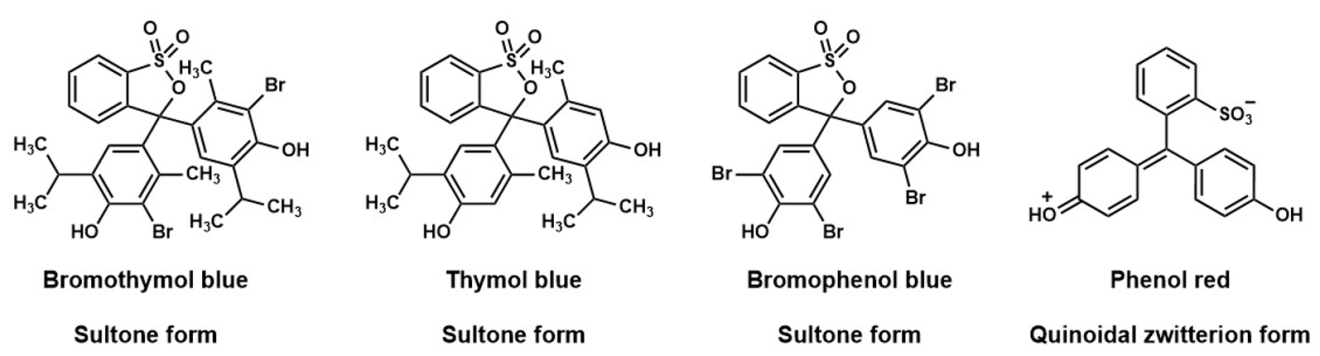
Figure 1: Example sulfonphthaleins used in teaching laboratory activities
Attempts to identify the molecular structures of bromothymol blue (and other sulfonphthaleins) associated with observed colour changes can be a confusing exercise. This is because there has been debate in the literature for many years about the structures of sulfonphthaleins, as a result, differing explanations are often provided in various sources.5-8 Fortunately, a better understanding of the structure of sulfonphthalein compounds has been reached in recent years by researchers who have investigated the pH responsive reactions of bromothymol blue (and other sulfonphthalein indicators) using a variety of analytical techniques.9-12 This short article aims to summarise the key findings from recent studies of the behaviour of sulfonphthalein compounds in aqueous solutions. We hope that the information provided will equip educators with up-to-date interpretations of very familiar experimental observations, such as colour changes in acid/base titrations. For a more detailed overview, readers may wish to consult the excellent review of the chemistry of sulfonphthalein compounds published recently by Magnaghi, Zanoni, Alberti, and Biesuz.5
Sulfonphthalein structures – Solid phase
A search of online sources will often show sulfonphthalein compounds, as a cyclic sulfonic ester, often referred to as a “sultone”, in acidic solutions5,7,8 (Figure 1 – bromothymol blue, thymol blue and bromophenol blue). This is not an unreasonable proposal, since the X-ray crystal structure of bromophenol blue has been published,11 and this compound is reported to exist as a sultone in the solid state (Figure 1). However, the same authors have also published an X-ray crystal structure of phenol red (Figure 1),12 in this case the molecule is reported to adopt a quinoidal zwitterion form in the solid state. The results from X-ray diffraction studies suggest that in the solid state, sultone or zwitterionic forms of sulfonphthaleins are possible, the isolated form is likely dependent on the substituents in each case. The X-ray studies also provide additional useful information, the solid bromophenol blue sample is described as “colorless crystal”,11 whereas the solid sample of phenol red is noted as being “bright red”.12 The vivid colour of the phenol sample is consistent with a fully delocalised π-system.6 In the case of bromophenol blue, the sultone ring disrupts the delocalised π-system, this structural change can shift the absorbed and emitted wavelengths out of the visible part of the electromagnetic spectrum. A similar effect can be seen in the solution state behaviour of phenolphthalein.13,14 In acidic aqueous solution, phenolphthalein indicator is colourless, this due to the presence of cyclic ester (lactone) structure that prevents a fully delocalised p-system being formed. The structure of phenolphthalein changes when the pH is adjusted to 8-9, the indicator then appears pink to the eye.4 The observed colour change is due to the formation of a planar quinoidal ion where the p electrons are fully delocalised.
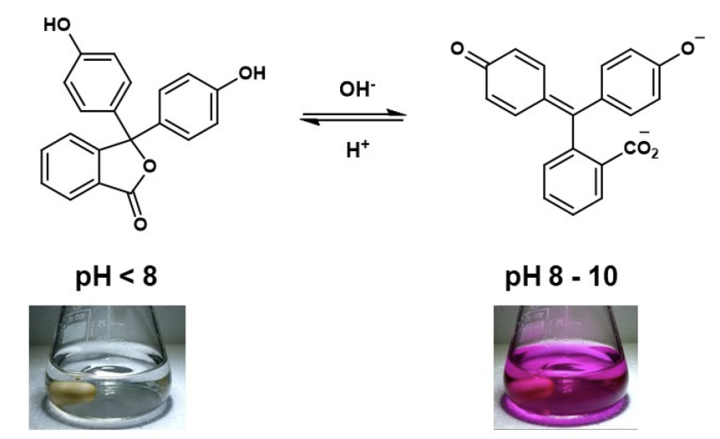
Equation 1: Phenolphthalein structures in aqueous solutions as pH is varied.
Video 1: Addition of dilute sodium hydroxide to acidified solutions of each indicator, phenolphthalein and thymolphthalein, GardenIndicators YouTube Channel, March 13, 2024.
Sulfonphthalein structures – Solution phase
In most educational settings, sulfonphthalein compounds are used in dilute solutions and the pH is usually varied. Under these conditions, spectroscopic techniques are more appropriate for investigating the molecular structures present. The structure of bromothymol blue has previously been investigated by Raman spectroscopy,5 more recently, a combined approach, using UV-visible spectroscopy and quantum mechanical simulations has been reported.9 The spectroscopic studies suggest that the bromothymol blue equilibrium shown in equation 2 is unlikely. These recent studies now suggest equation 3 aligns better with experimental results.9 Bromothymol blue appears green at the pH range 6.5-7.0, the spectroscopic data reported suggests that this color is not attributed to a specific structure, rather, it is likely a mixture of the yellow and blue forms.9
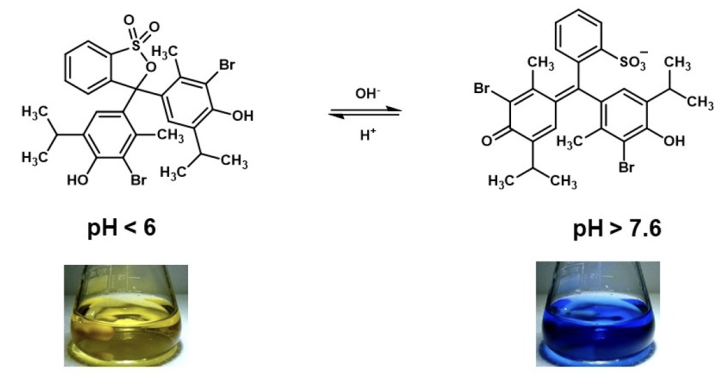
Equation 2: Previously proposed bromothymol blue structures in aqueous solutions as pH is varied.
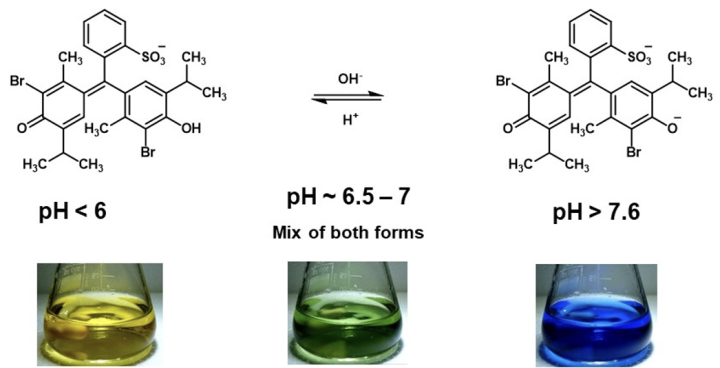
Equation 3: Recently proposed bromothymol blue structures in aqueous solutions as pH is varied.
The authors of the spectroscopic study of bromothymol blue have also used a similar approach to investigate the pH-dependent behaviour of thymol blue in aqueous solution.10 Thymol blue shows three colour changes: red (pH < 1.2), yellow (pH 2-8) and blue (pH > 8).4 Spectroscopic investigations suggest three structures exist in solution. All three structures are proposed to be quinoidal, with no indication that any of the sulfonphthalein molecules in solution contain a sultone ring (Equation 4).
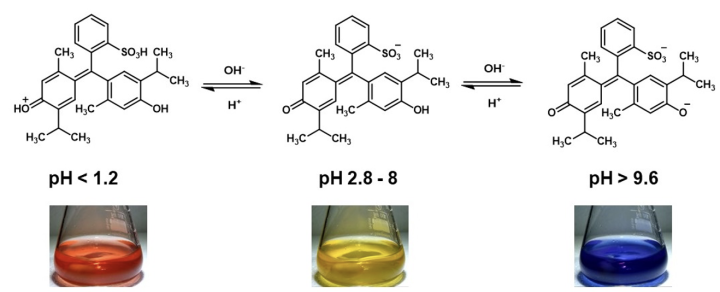
Equation 4: Recently proposed thymol blue structures in aqueous solutions as pH is varied.
Summary
In summary, current evidence suggests that in the solid state, sulfonphthalein indicators can potentially exist as cyclic or open structures. However, in contrast to phthalein acid/base indicators, recent experimental evidence suggests that sulfonphthaleins are unlikely to exist in cyclic forms in acidic aqueous solutions.
References
- Tucker, S. A; Acree, W. E, "A student-designed analytical laboratory method", J. Chem. Educ., 1994, 71, 71-74.
- Herbrandson, H. F., "The hydrolysis of t-butyl chloride", J. Chem. Educ., 1971, 48, 706-707.
- Owyeung, R. E.; Panzer, M. J.; Sonkusale, S. R. “Colorimetric gas sensing washable threads for smart textiles” Sci. Rep., 2019, 9, 5607.
- Vogel’s Textbook of Quantitative Chemical Analysis 6th Edition, Mendham, J.; Denney, R. C.; Barnes, J. D.; Thomas, M. Prentice Hall, 2000; page 38.
- Magnaghi, L. R.; Zanoni, C.; Alberti, G.; Biesuz, R. “The colorful world of sulfonephthaleins: Current applications in analytical chemistry for “old but gold” molecules” Anal. Chim. Acta, 2023, 1281, 341807.
- Heger, D.; Klánová, J.; Klán. P. “Enhanced protonation of cresol red in acidic aqueous solutios caused by freezing” J. Phys. Chem. B 2006, 110, 1277-1287.
- Klotz, E.; Doyle, R.; Gross, E.; Mattson, B. "The equilibrium constant for bromothymol blue: A general chemistry laboratory experiment using spectroscopy", J. Chem. Educ., 2011, 88, 637-639.
- Yimkosol, W.; Dangkulwanich, M. "Finding the pKa values of a double-range indicator thymol blue in a remote learning activity", J. Chem. Educ., 2021, 98, 3930-3934.
- Shimada, T.; Hasegawa, T. “Determination of pH structures of bromothymol blue revealed by using quantum chemistry with an aid of multivariate analysis of electronic absorption spectra” Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 2017, 185, 104-110.
- Shimada, T.; Tochinai, K.; Hasegawa, T. “Determination of pH structures of thymol blue revealed by cooperative analytical method of quantum chemistry and multivariate analysis of electronic spectra” Bull. Chem. Soc. Jpn., 2019, 92, 1759-1766.
- Yamaguchi, K.; Tamura, Z.; M. Maeda “Molecular structure of bromophenol blue having a g-sultone ring” Analytical Sciences, 1997, 1057-1058.
- Yamaguchi, K.; Tamura, Z.; M. Maeda “Molecular structure of the zwitterionic form of phenolsulfonphthalein” Analytical Sciences, 1997, 13, 521-522.
- Wittke, G. “Reactions of phenolphthalein at various pH values” J. Chem. Educ., 1983, 60, 239-240.
- Berger, S. “The pH dependence of phenolphthalein” Tetrahedron, 1981, 37, 1607-1611.