Will a Watched Pot Ever Boil? A Simple, but Effective, Chemistry Lab: Constructing a Heating Curve of Water
Heating and cooling curves are important visuals for chemistry students to understand when they are studying phase changes and thermal energy. While most textbooks and other traditional resources present heating and cooling curves and explain what the graphs show, it is impactful when students discover the shape of the curves through their own data collection. The process of collecting and plotting their own data can also help dispel the common misconceptions that substances continue to rise in temperature when undergoing a phase change. The process of collecting and graphing their own data will help students understand how and why substances stay at a certain temperature while going through a phase change.

Do you know the expression “a watched pot never boils”? Well, this lab activity guides students through the process of collecting temperature data as they watch water boil. Don’t worry, the water will still boil even when they are making careful measurements and observations! When they graph their data, they will see the shape of a heating curve. The advantages of the lab include its relatively safe nature, the low teacher prep required, the simplicity of equipment and materials, and the fact that it can even be done at home.
The most impactful part of this activity is the possible “aha!” moment when students connect their hypotheses to their resulting graphs. It is recommended to do this lab before showing or assigning reading about heating / cooling curves so that students can truly discover it themselves. This is also a great opportunity to have students create a hypothesis by sketching a graph of what they think their data will look like in the end. While written hypotheses, especially in the “if…then…” format, are vital for high school science courses, thinking ahead about graphs and relationships between variables is a high-order skill crucial for scientists. If students are struggling with where to start when making a hypothesis-graph, the teacher can provide the x- and y-axes and can show several options of graph ideas to get them started.
Materials
- Hot plate
- Boiling flask or small cooking pot
- Thermometer
- Heat proof glove / tongs
- Tap water
- Stopwatch
Background
Procedure
An example student procedure and teacher’s guide are provided in the Supporting Information below, but the main steps of the lab are fairly simple. Many different combinations of equipment can work. The most important parts are a heat source, such as a hot plate or stove; a heat-proof container for the boiling water, such as a heat-proof beaker or small kitchen pot; a thermometer; and a stopwatch.
Students begin by responding to the following question with a prediction.
How will the temperature of water change over time as you apply heat to bring it to a boil?
Students are then directed to sketch a graph of that hypothesis. Developing a hypothesis in the form of a graph is most likely new for students, so they might need extra guidance to do this. One idea is to show example graphs that they can choose from or use as a starting point (examples are offered in the Supporting Information).
The students turn on the heat source and begin to take temperature readings at regular time intervals. Qualitative observations should also be recorded. When the water reaches a rolling boil, students take temperature readings for two additional minutes. Then, they can graph and analyze their data.
Figure 1 is an example of a student graph made using spreadsheet software.
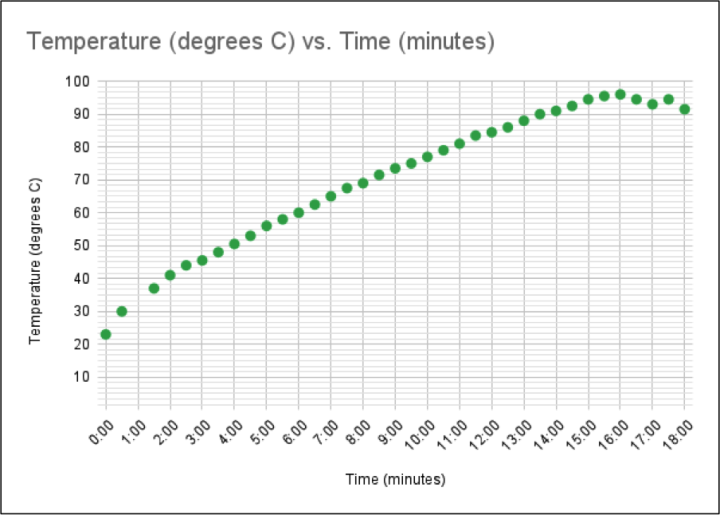
Figure 1: Temperature vs Time graph in spreadsheet software
Students then analyze their graphs, compare them to heating curves from their textbook and answer conclusion questions.
As far as safety goes, boiling water and steam can both cause burns. Students should use heat-safe gloves or potholders when handling equipment, should tie long hair and clothing back, and should not put their hands or faces over boiling water or steam.
This lab has successfully been done at home with a kitchen stove, pot, and meat thermometer. Distance learning or homeschool courses could take advantage of this simple set up to deliver a rigorous lab environment at home.
Overall, this lab activity packs a lot of punch for a relatively simple set up. As chemistry teachers, we want to maximize the time in class where students are doing hands-on and high-order thinking tasks. However, we teachers are usually short on prep time and resources. This kind of lab activity can strike a good balance between these desires and constraints.
SUPPORTING INFORMATION:
There is a full-length student lab handout with procedure and post-lab questions, as well as a teacher guide with NGSS standards, helpful tips, and detailed procedure notes. The Supporting Information can be found below when you are logged into your ChemEd X account. Not a member? Register for FREE!
Questions
Concepts
Time Required
If working in the lab, students work in groups of 2-3 students. It is helpful to have at least 2 people working together, as one can keep an eye on the stopwatch and record data while the other looks at the thermometer and the water for observations.
The procedure and data collection would take a 45-minute class period. Post-lab graphing and the conclusion can be completed as homework if the students don't finish it in class.
Preparation
Prepare materials for groups within the classroom or advise students how to set up the heating curve materials at home. It is helpful to test the equipment to be used and determine a rough idea of how many minutes the water will take to boil with your specific equipment. The heat source and volume of container will determine this and it is useful to give students a rough idea of how long they will need to record data.
Attribution
Thank you to my chemistry students who tested this lab for me and helped to refine it!
Credits
General
Items tagged with the safety:general tag have some safety concerns that may be also described more specifically by additional safety terms
General Safety
For Laboratory Work: Please refer to the ACS Guidelines for Chemical Laboratory Safety in Secondary Schools (2016).
For Demonstrations: Please refer to the ACS Division of Chemical Education Safety Guidelines for Chemical Demonstrations.
Other Safety resources
RAMP: Recognize hazards; Assess the risks of hazards; Minimize the risks of hazards; Prepare for emergencies
