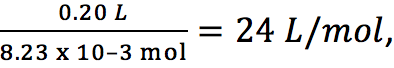A Super-Engaging Way to Start 11 Chemistry

I still love the first day of a course, even after almost 36 years in this business.
But I don’t love wasting student-energy and excitement droning through course expectations, mark-allocations and the like. Now, more than ever, face-time cannot be taken for granted or frittered away.
Yes . . . teacher-demonstrations can be good, but student-wise they can be passive, and have the potential to reduce Chemistry to entertainment.
Here’s what I do on day one: After taking attendance, stressing the importance of respectful communication (read: “Yes, Sir; no Sir”); mobile phones on the side bench; and making it clear that asking questions—anything (!)—is encouraged, I get’em started on a lab—real minds-on and hands-on work.
Bonus: This lets me introduce my favorite words: EMPIRICAL EVIDENCE.
Materials
Magnesium, 0.20 g per trial
Zinc, 0.43g per trial
100 mL of 1 mol∙L–1 HCl(aq) à to react with Mg
100 mL of 6 mol∙L–1 HCl(aq) à to react with Zn
Each student station requires:
pneumatic trough filled with water; two - 250-mL flasks, stopper with glass tube and hose; 100 mL graduated cylinder; grease pencil for marking the amount of gas produced; beaker (see figure 1)
Background
Procedure
The primary goal of the lab is the determination of the relative atomic mass of Mg to Zn, but I keep this to myself . . . for the time being. Students accomplish this by quantitatively reacting a small amount of each metal—separately—with excess HCl(aq) according to the reactions:
Mg(s) + 2 HCl(aq) à MgCl2(aq) + H2(g) Equation 1
Zn(s) + 2 HCl(aq) à ZnCl2(aq) + H2(g) Equation 2
Students collect the H2(g) by downward displacement of water as illustrated.

Figure 1: Collection of gas by downward displacement of water.1 We use a rubber hose instead of a glass tube—just sayin’.
Students collect the gas in an inverted 250 mL Erlenmeyer flask, and mark the level of gas collected with a grease pencil. They subsequently determine the volume of H2(g) collected using a 100 mL graduated cylinder and tap water.
I use this totally imprecise method for a reason; it allows me to introduce the concept of precision—or lack of it—and confidence level, well in advance of formally teaching significant figures.
Students react magnesium with ca. 100 mL of 1 mol∙L–1 HCl(aq); they react the zinc with 100 mL of 6 mol∙L–1 HCl(aq).2,3 In both cases, the HCl(aq) is in excess. These acids are measured and provided to students in advance. To save money, I use muriatic acid purchased at Canadian Tire.4
Table 1: Sample student data, collected by a bunch of rookies (25 groups):
|
Metal |
Class average of mass used per trial (g) |
Class average of H2 collected per trial (L) |
Mass of metal per 0.20 L H2 produced (g) |
Empirically—(student) determined ratio of atomic mass |
Actual (Periodic Table) ratio of atomic masses (amu or g∙mol–1) |
Actual (Periodic Table) ratio of atomic masses |
|
Mg |
0.20 |
0.20 |
0.20 |
1 |
24.3 |
1 |
|
Zn |
0.43 |
0.16 |
0.54 |
2.7 |
65.3 |
2.7 |
The results of this experiment, shown in Table 1, are nothing short of spectacular. On the first day of their first course dedicated to chemistry, students determine—with enviable accuracy—the relative atomic mass of Mg : Zn.
Incredible!
This lays the foundation for chemistry as an empirical science; it shows that atomic masses from the Periodic Table are not mysterious or arbitrary.
Along the way, I foreshadow—or soft intro—a number of topics that will be taught later in the course:
- the mole (unit of quantity)
- concentration, or quantity per liter (mol∙L–1)
- reaction rate
- solubility of gas in a liquid
- measurement, precision, significant figures
Later in the course, I reference these data to determine the molar volume of a gas under classroom conditions of temperature and pressure—ca SATP (Standard Atmospheric Temperature and Pressure, 25C, 100 kPa).
Referencing equation 1 and Table 1 we have
0.20 g Mg = 8.23 x 10–3 mol Mg, which produces 8.23 x 10–3 mol H2(g), which occupies 0.20 L.
This gives, for H2(g), a molar volume of
which is, to two significant figures, equal to the accepted value of 24.8 L∙mol–1, or 25 L∙mol–1.
The data for Zn give similar results.
At the risk of repeating myself: Incredible!!
From this initial student investigation, we get two labs-worth of data, and two fabulous empirically determined values, both of which are, to the precision level of the experiment, right on the money.
A student handout in the Supporting Information below.
May peace be with you,
Michael
Questions
Concepts
Time Required
I have no problem carrying out the lab in a 75 minute period, which included time for a demonstration and explanation of the procedure (collection of gas by downward displacement of water). Shorter class periods could have one-half the students reacting Mg with 1 mol/L HCl(aq), the other half of the class reacting Zn with 6 mol/L HCl(aq).
Preparation time is about 30 minutes.
Preparation
Set up student lab stations. Each station has the pneumatic trough, filled with water, two 250-mL flasks, stopper with glass tube, hose and grease pencil.
Attribution
- https://www.bestchoice.net.nz/html/sa1/main/s1219/p19281.htm
- Zn reacts much more slowly with HCl(aq) than Mg. This explains the higher [HCl(aq)] that is reacted with Zn.
- If you—or the safety wonks at your school district—feel that 6 mol∙L–1 HCl is too concentrated for student use, this part of the experiment can be done as a teacher demonstration.
- US residents will have to come to Canada, or simply go to a local hardware or building supply store to purchase Muriatic Acid, which is ca 10 mol∙L–1 HCl(aq).