Solutions Interactive Notebook

This is a relatively short unit in my course and as a result, I only use two spreads for it. I have two fully developed spreads to share, with optional bonus material you could add on to the first and an alternative layout for the second that includes colligative properties. This unit in my notebook changes from year to year, depending on how much time we have and resulting depth of coverage. I’ll discuss the details within the description for each spread. The next unit to come is acids and bases – one of my favorites!
Materials
Composition notebooks, printed copies of each page, colored paper or construction paper for making flipbooks/foldables, scissors, tape or glue
**A note on my manipulatives: If you see a dashed line, that is where the item should be cut. If you see dotted lines, that means fold.
Background
For backstory about how Nora uses interactive notebooks, view a recording of her ChemEd X Talk: Integrating Interactive Notebooks into Chemistry Courses with Nora Walsh and read her previous post, Interactive Notebook Unit on Stoichiometry. Nora wrote the following in response to questions she received after her ChemEd X Talk: A Classroom View of Using Interactive Notebook Pages.
See all of the interactive notebook units Nora has published: INTERACTIVE NOTEBOOK COLLECTION
Procedure
Spread 1: Solutions Vocab

Rightside - All About Solutions
This is a vocabulary-heavy page. As I’ve discussed in other INB units, I’m moving toward a class progression that follows Activity Before Concept and Concept Before Vocabulary. To this end, I will be using this spread as a summary page after we have done several activities that go over the concepts of solvation and types of mixtures. My plan this year is to have the students deduce the meaning of the vocabulary words by using a gallery walk.
You can choose to add or remove terms on this page as your curriculum map, state test or standards demand. I also strongly encourage you to consider vertical alignment and next-class progression when you are deciding what elements to keep, remove or add to this page.
The top-left flap lifts to provide extra space to show the relationships (with particle diagrams) between miscible, immiscible, electrolyte, nonelectrolyte, solution, solute and solvent. Keep in mind that while we as teachers may be fluent in transitioning between these terms, students struggle to distinguish them. These words are all related and even interchangeable, but students need support in synthesizing these connections. The particle view diagrams help illustrate these relationships.
A familiar example of a cup of tea shows the real-world concepts of concentrated and dilute, while an image depicting the dissolution of a crystal relates saturated, unsaturated and supersaturated. Underneath the image flap are definitions for those terms, with blanks for specifics. Factors affecting solvation rate (a bite-size dip into kinetics) are listed below. Finally, particle view diagrams comparing size of particles allows connections to be made between solution, colloid and suspension.
I have provided a Bonus Material document that has a large table (that I fold into an accordian foldable) for solutions, suspensions and colloids. This table could be pasted in the “Types of Mixtures” box on the bottom right area of the spread, if you wish to get more into the specifics of these mixtures.
Regardless of what information I include on this page, I use a 4-square layout and have lots of layers and foldables to represent a large amount of solution terms.
Leftside: Solutions True-False Pocket Sort
The output for this page was designed to help students begin to do some error analysis with solutions, leading them to pay attention to details and also to disrupt some misconceptions. This activity can easily be lengthened or shortened in your classroom in several ways:
- add some word or model-based prompts
- have students correct false statements
- have students create additional true and false statements
True and False pockets are created by cutting out the rectangles and taping them along three sides. The prompts are cut out, and each is inserted into its corresponding pocket.
You may wish to encourage students to write the correct answer on the back of each prompt so that they can redo and check this activity later.
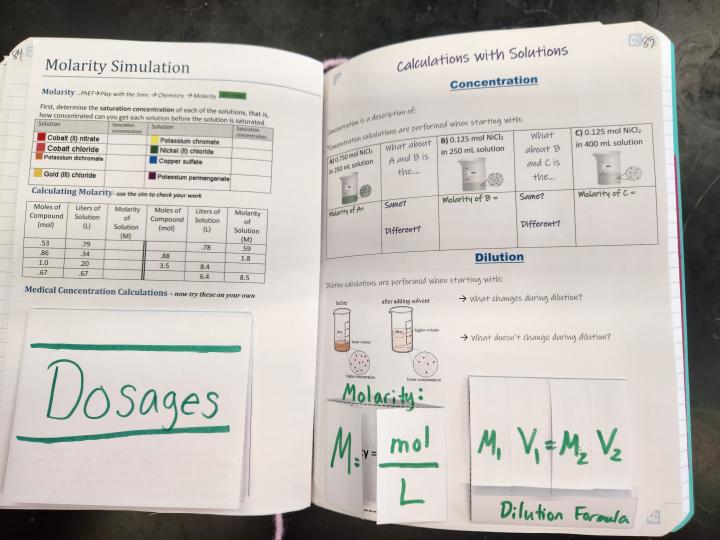
Spread 2: Calculations with Solutions
This spread focuses on the quantitative side of solutions, but still integrates particle view diagrams to help students relate the various levels of representations. These two pages, maybe more than any previous spread I’ve shared, really work together. In fact, I would suggest that students have the PhET - Molarity simulation open as they work through the right side. The concepts of molarity and dilution are made more accessible and real-world to students by integrating them with the simulation on the left page. It helps students to visually make connections between concentration and macroscopic observations. By incorporating particle view diagrams, molarity and dilution calculations and the PhET sim, this spread hits all 3 scales on Johnstone’s Triangle. For more on Johnstone’s Triangle and its importance in chemical education, see these three ChemEd X posts:
- Why Do You Make Us Draw so Many Particle Diagrams?
- Tribute to Alex Johnstone
- Integrating Three Types of Chemical Representation
Rightside: Molarity Simulation
This page is the guiding document to go along with the PhET sim. At the top of the page, students find the saturation point for each solution in the sim that shows one. Then, they calculate molarity using the formula from the leftside. The sim allows them to check their work by using the sliders to see if their math matches up. Finally, at the bottom of the page are a handful of questions to apply their new knowledge to real-world medical problems.
Leftside: Calculations with Solutions
This page mostly focuses on the math of concentration, but seeks to have students see the meaning behind the numbers (instead of rotely plugging and chugging values). The particle view diagrams at the top with the comparison prompts help students to see that molarity is influenced by both amount of solute AND amount of solution. Additionally, by focusing on concentration on the top part of the page and dilutions on the bottom part, students can see the difference between adding a solute to a solvent and adding more solvent to an existing solution.
The bottom of the page has mini foldables with flaps to show variables. Students who struggle with manipulating variables in algebraic equations find these foldables helpful when substituting into equations.
Alternative page layout for Spread 2: If you cover colligative properties in your solutions unit, I have an alternative layout for spread 2 that covers this topic. It is also attached. It lacks the particle view diagrams because of space constrictions.
It might be helpful to view video 1 to help visualize what these pages will look like in action.
Video 1: Solutions INB pages, ChemEd X Vimeo Channel, 4/15/2023.
Questions
Concepts
Time Required
Preparation
Provide copies of the following for each student. Note that I have grouped the components by page. Please preview them before printing as some pages have multiple copies to minimize paper waste.
Spread 1:
All About Solutions Printing notes: All students get all of page 1 – the 4 squares are cut out as 1 big page, and then the saturation test tubes are cut out to be a flap. Page 2 has 4 sets of miscible/immiscible vocabulary flaps, so print that on a separate job and separate the sets for students.,
Spread 2:
Calculations with Solutions (*Printing note: Page 1 contains two sets of each foldable. You should print and separate these into sets for the students. The second page is a full-page glue down, that only needs to be trimmed to notebook page size.),
Molarity Simulation (This is a 2 page document that needs to be printed one-sided. The first page is trimmed to notebook-size, and then the second page is cut out as 1 big rectangle, and folded in quarters to attach at the bottom of the page.)
Alternative Spread 2 - Solution Behavior (with colligative props)
Attribution
PhET Molarity simulation (accessed 4/13/2023)

