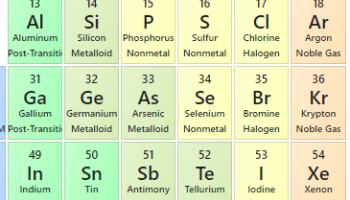
Years ago, I put on a pair of snowshoes during a winter cold snap and found that the plastic binder straps broke fairly easily. At first I thought the plastic straps had simply become more brittle with time, but I found that the straps became less brittle when brought inside to warmer temperatures.