Plating Pennies with Tin

One of my favorite chemistry demonstrations is the “Copper to Silver to Gold” experiment1 (VIDEO 1), in which a penny is first plated with zinc and then heated to form brass, an alloy of copper and zinc. The heat causes zinc to diffuse into the copper in the penny. The zinc plate imparts a beautiful silver color to the penny. The brass that is formed upon heating has a stunning golden color.
Video 1: Copper to "Silver" to "Gold", Tommy Technicium YouTube Channel (accessed 2/1/2021)
My students and I have performed this experiment many times, and students often keep the golden colored pennies as keepsakes. Unfortunately, the zinc plated pennies lose their silver color over a few days, presumably because zinc diffuses into the copper even at room temperature.2 A new experiment published in the Journal of Chemical Education describes how copper can be coated with tin.2 In this case, the silver-colored coating imparted by the tin appears to last for quite some time – perhaps indefinitely! I tried to see if this tin-plating experiment would work on pennies (VIDEO 2).
Video 2: Plating pennies with tin, Tommy Technicium YouTube Channel (accessed 2/1/2021)
The pennies I plated with tin using this method have kept their silver color for weeks and show no sign of discoloring! The authors claim that the stability of the tin-plated copper as opposed to the zinc-plated copper can be understood by comparing the enthalpies of sublimation of tin (301 kJ mol-1) and zinc (130 kJ mol-1).2 Based on these values, the bonds between tin atoms are stronger than those between zinc atoms. As a result, zinc is more likely to diffuse into the copper than tin – and it does so even at room temperature. When zinc diffuses into the copper, it loses its silvery coat because brass is formed.
A few other features of the experiment are of note. The original “Copper to Silver to Gold” experiment requires the use of caustic sodium hydroxide solution, while the reagents required for this new tin-plating experiment are quite benign by comparison. Also, the tin-plated pennies can be heated to form bronze, which is a golden-colored alloy of copper and tin. I tried to form bronze on tin-plated pennies with mixed success. I could achieve a golden color by heating tin-plated pennies, but I had trouble getting a uniformly golden color. In my opinion, if you wish to plate pennies with a golden color, the formation of brass via the original “Copper to Silver to Gold” experiment is superior to this new experiment. However, if you wish to give pennies a nice silvery coat, this tin-plating experiment is the way to go!
Given that copper is higher up in the electrochemical series than tin, it might seem curious that the tin plates out on copper in this experiment. In fact, it is not the case that an electrochemical reaction between tin ions and copper metal occurs in this experiment. Rather, tin metal in granular tin is oxidized to Sn2+(aq):
Sn(s) à Sn2+(aq) + 2 e-
The electrons transfer into the surface of the copper, where dissolved tin ions are then reduced to tin metal. This forms the tin plate:
Sn2+(aq) + 2 e- à Sn(s)
The overall process involves granular tin being deposited on the surface of the copper. Figure 1 below summarizes this whole process. The authors state that this happens because the tin atoms are more stable when interacting with copper atoms than they are when interacting with each other.2
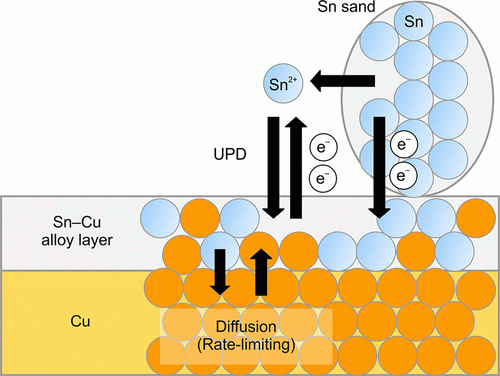
Figure 1: Principle of Sn Plating - Reprinted with permission from Suzuki, T.; Inoue, M., *Cu Plating with Sn and Subsequent Bronze Formation under Mild Conditions, J. Chem. Educ. Copyright 2021 American Chemical Society.
I will be interested to see how the pennies that I plated with tin retain their silver color – or not – over the next few months. I’m hoping that the silver color lasts for years. If you try out this experiment, let me know how the silver coat works for you. Also, if you get the bronze coating to form with a nice, uniform golden color, be sure to send me a few tips on how to do so.
Happy experimenting!
References
- Shakhashiri, B. Z. Chemical Demonstrations: A Handbook for Teachers of Chemistry; University of Wisconsin: Madison, WI, 1992; Vol. 4, pp 263−268.
- Suzuki, T.; Inoue, M., *Cu Plating with Sn and Subsequent Bronze Formation under Mild Conditions, J. Chem. Educ. 2021, DOI: 10.1021/acs.jchemed.0c00552.
*See Accessing Cited Articles.