JCE 93.12 December 2016 Issue Highlights
Improving Student Understanding
The December 2016 issue of the Journal of Chemical Education is now available online to subscribers. Topics featured in this issue include: synthesis in the laboratory, examining and using a flipped classroom, improving labs through multimedia-based and student-directed learning, using applied math for better understanding, improving student understanding of thermodynamics, inclusive chemistry teaching, using manuscript review for assessment, climate chemistry, spectroscopy experiments, performing safe demonstrations.
Cover: Synthesis in the Laboratory
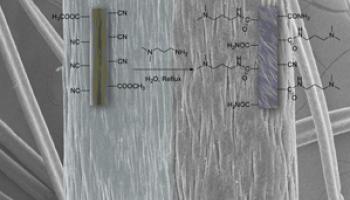
In Synthesizing Substituted 2-Amino-2-chromenes Catalyzed by Tertiaryamine-Functionalized Polyacrylonitrile Fiber for Students To Investigate Multicomponent Reactions and Heterogeneous Catalysis, Yujia Xie, Xiaoxing Liu, and Minli Tao discuss a multistep experiment for a synthesis laboratory course that incorporates organic synthesis, chemical analysis, and instrumental analysis. In the experiment, students characterize a tertiaryamine-functionalized polyacrylonitrile fiber (PANTF) synthesized from polyacrylonitrile fiber (PANF) and N,N-dimethyl-1,3-propanediamine (shown on the cover). Students then use PANTF as an immobilized catalyst in a three-component condensation reaction. At the end of the reaction, the fiber catalyst can be easily separated from the reaction system by simple filtration and used directly in the next cycle. SEM images of PANF (left half of cover, shown at two different levels of magnification) and PANTF (right half of the cover, shown at two different levels of magnification) reveal that the smooth surface of PANF becomes slightly rougher after reaction to form PANTF.
Other synthesis labs in this issue include:
Biocatalyzed Regioselective Synthesis in Undergraduate Organic Laboratories: Multistep Synthesis of 2-Arachidonoylglycerol ~ Meghan R. Johnston, Alexandros Makriyannis, Kyle M. Whitten, Olivia C. Drew, and Fiona A. Best
Drug Synthesis and Analysis on a Dime: A Capstone Medicinal Chemistry Experience for the Undergraduate Biochemistry Laboratory ~ Craig N. Streu, Randall D. Reif, Kelly Y. Neiles, Amanda J. Schech, and Pamela S. Mertz
Editorial
Norbert J. Pienta highlights Journal of Chemical Education content in 2016 and acknowledges contributors to the Journal in Volume 93 in Review.
Commentary
Harry E. Pence discusses the opportunities for chemical educators available through cloud computing. By Moving Chemical Education into the Cloud(s) every student in a class can have access to all of the resources necessary for the class, which encourages collaboration and the development of new educational models.
Examining and Using a Flipped Classroom
Evaluation of a Flipped, Large-Enrollment Organic Chemistry Course on Student Attitude and Achievement ~ Suazette R. Mooring, Chloe E. Mitchell, and Nikita L. Burrows
A Parallel Controlled Study of the Effectiveness of a Partially Flipped Organic Chemistry Course on Student Performance, Perceptions, and Course Completion ~ James C. Shattuck
Coordinated Implementation and Evaluation of Flipped Classes and Peer-Led Team Learning in General Chemistry ~ Jenay Robert, Scott E. Lewis, Razanne Oueini, and Andrea Mapugay
Scaffolded Semi-Flipped General Chemistry Designed To Support Rural Students’ Learning ~ Mary S. Lenczewski
Improving Labs through Multimedia-Based and Student-Directed Learning
Comparable Educational Benefits in Half the Time: An Alternating Organic Chemistry Laboratory Sequence Targeting Prehealth Students ~ Sherri C. Young, Keri L. Colabroy, and Marsha R. Baar
LabLessons: Effects of Electronic Prelabs on Student Engagement and Performance ~ Patrick Gryczka, Edward Klementowicz, Chappel Sharrock, MacRae Maxfield, and Jin Kim Montclare
The Effect of Procedural Guidance on Students’ Skill Enhancement in a Virtual Chemistry Laboratory ~ Sehat Ullah, Numan Ali, and Sami Ur Rahman
Using Applied Math for Better Understanding
Box-and-Whisker Plots Applied to Food Chemistry ~ João E. V. Ferreira, Ricardo M. Miranda, Antonio F. Figueiredo, Jardel P. Barbosa, and Edykarlos M. Brasil
Let Students Derive, by Themselves, Two-Dimensional Atomic and Molecular Quantum Chemistry from Scratch ~ Yingbin Ge
Teaching Reciprocal Space to Undergraduates via Theory and Code Components of an IPython Notebook ~ Matthew N. Srnec, Shiv Upadhyay, and Jeffry D. Madura
Improving Student Understanding of Thermodynamics
Improving Students’ Understanding of the Connections between the Concepts of Real-Gas Mixtures, Gas Ideal-Solutions, and Perfect-Gas Mixtures ~ Romain Privat, Jean-Noël Jaubert, and Edouard Moine
From Discrete to Continuous Process Simulation in Classical Thermodynamics: Irreversible Expansions of Ideal Monatomic Gases ~ Carmen Álvarez-Rúa and Javier Borge
Inclusive Chemistry Teaching
Promoting Inclusive Chemistry Teaching by Developing an Accessible Thermometer for Students with Visual Disabilities ~ Felipe A. Vitoriano, Vânia L. G. Teles, Ivanise M. Rizzatti, and Régia C. Pesssoa de Lima
Communicating Science Concepts to Individuals with Visual Impairments Using Short Learning Modules ~ Anthony S. Stender, Ryan Newell, Eduardo Villarreal, Dayne F. Swearer, Elisabeth Bianco, and Emilie Ringe
Using Manuscript Review for Assessment
The “pHunger Games”: Manuscript Review to Assess Graduating Chemistry Majors ~ David J. Gorin, Elizabeth R. Jamieson, K. T. Queeney, Kevin M. Shea, and Carrie G. Read Spray
Climate Chemistry
Using Demonstrations Involving Combustion and Acid–Base Chemistry To Show Hydration of Carbon Dioxide, Sulfur Dioxide, and Magnesium Oxide and Their Relevance for Environmental Climate Science ~ C. Frank Shaw III, James W. Webb, and Otis Rothenberger
Using Modern Solid-State Analytical Tools for Investigations of an Advanced Carbon Capture Material: Experiments for the Inorganic Chemistry Laboratory ~ Mario Wriedt, Julian P. Sculley, Darpandeep Aulakh, and Hong-Cai Zhou
Spectroscopy Experiments
Analyzing Exonuclease-Induced Hyperchromicity by UV Spectroscopy: An Undergraduate Biochemistry Laboratory Experiment ~ Megan M. Ackerman, Christopher Ricciardi, David Weiss, Alan Chant, and Christina M. Kraemer-Chant
Flash Photolysis Experiment of o-Methyl Red as a Function of pH: A Low-Cost Experiment for the Undergraduate Physical Chemistry Lab ~ Molly C. Larsen and Russell J. Perkins
Kinetics and Photochemistry of Ruthenium Bisbipyridine Diacetonitrile Complexes: An Interdisciplinary Inorganic and Physical Chemistry Laboratory Exercise ~ Teresa L. Rapp, Susan R. Phillips, and Ivan J. Dmochowski
From the Archives: Performing Safe Demonstrations
Demonstrations can be an engaging and effective way to show chemistry in action, but they need to be conducted in the safest manner possible. In this issue, John J. Dolhun discusses Peak Sound Pressure Levels and Associated Auditory Risk from an H2–Air “Egg-Splosion”. Some additional articles that address the noise level from exploding chemical demonstrations are:
Auditory Risk of Exploding Hydrogen−Oxygen Balloons ~ Kent L. Gee, Julia A. Vernon, and Jeffrey H. Macedone
Managing Auditory Risk from Acoustically Impulsive Chemical Demonstrations ~ Jeffrey H. Macedone, Kent L. Gee, and Julia A. Vernon
Additional articles that improve on the safety of popular demonstrations include:
Observations on Manganese Dioxide As a Catalyst in the Decomposition of Hydrogen Peroxide: A Safer Demonstration ~ John J. Dolhun
Variations on the "Whoosh" Bottle Alcohol Explosion Demonstration Including Safety Notes ~ John J. Fortman, Andrea C. Rush, Jennifer E. Stamper
Whoosh Bottle Safety, Again: What About What Is Inside? ~ Robert B. Gregory and Matthew Lauber
A very safe way to experience a demonstration is with a video, such as the Chemistry Comes Alive! collection available to subscribers of ChemEdX.
Improving Understanding with JCE
With 93 volumes of the Journal of Chemical Education to explore, you will always find something to improve understanding—including the articles mentioned above, and many more, in the Journal of Chemical Education. Articles that are edited and published online ahead of print (ASAP—As Soon As Publishable) are also available.
Do you have something to share? Write it up for the Journal! For some advice on becoming an author, read Erica Jacobsen’s Commentary. In addition, numerous author resources are available on JCE’s ACS Web site, including: Author Guidelines, Document Templates, and Reference Guidelines.