Formative Assessment via Redox Reactions
Three Dimensional Assessments
Next Generation Science Standards (NGSS) or some version of the standards have been adopted in many states across the country. The phenomenon based teaching style called for in the standards, have raised questions about the ways in which three dimensional student learning should be assessed. My district recently provided a professional development session focused on utilizing three dimensional formative assessments in the classroom. The ideas I learned in the session as well an an activity for students to engage in formative assessment are outlined below.
What are Formative Assessments?
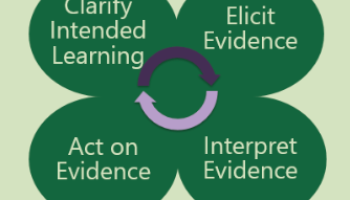
Formative assessments are utilized by educators to stay aware of what students actually know or do not know about a topic as well and provide evidence of student learning. When utilizing formative assessments with the end educational outcome in mind the teacher and student engage in a process of evaluating what learning has taken place and evaluate and fine-tune the next piece of learning that needs to occur. Formative assessments have no point values attached to them and are “low stakes” means for me to continually check in with my students. When designing these assessments, inclusivity and the equality of the various types of learners need to be kept in mind so that the interest and prior knowledge of all students are addressed. For example, a students lack of exposure or prior knowledge about a phenomena should not impede their ability to correctly answer the task at hand. Thus the assessments should be relevant to the learners and promote self- reflection, self- learning and social learning and are deliberate tools to measure what actions need to be taken to promote the process of learning science, as opposed to summative assessments which measure what students have learned with focus on a particular product. Formative assessments should be utilized throughout the unit and the following model can be used to guide the process. The teacher clarifies the intended learning goal and then provides a task and will elicit evidence of student learning. The teacher and student can then interpret that evidence and act on the evidence to clarify or modify the learning goal (see figure 1).
Figure 1: The Formative Assessment Process (Adapted by Simpson & McCulloch from Ruiz-Primo & Furak 2007)
Types of Formative Assessment
- Data Driven Decision Making- uses benchmark assessment to identify areas of improvement
- Strategy Focused- tools and practices that can be routinely used by the teacher to provide feedback and engage the students in learning
- Cognitive- examines the social nature of learning to assess how students knowledge is utilized as they engage in practices similar to those that a disciplinary expert in that subject would
- Cultural- the teacher elicits information that students bring with them from outside the classroom including their knowledge, interests and experiences and applies this way of knowing, being and learning in the natural world that is specific to science.
How to Provide Formative Assessments
- On the fly check ins
- Conversations with and between students
- Curriculum embedded
- Exit-tickets
- Do-Nowes
- Criterion based rubrics to evaluate student performance
Formative assessments should not be graded for points or should have a low stake value. For instance: points for effort in the grade. The purpose is to examine a student performance in a way that is manageable for the teacher.
Redox Formative Assessment
One of the tools I have found most beneficial for use as a means of formative assessment is the set of probes that Page Keeley has written for various topics. I collaborate with chemistry teachers in my area and recently, we wrote our own probes for oxidation and reduction (see figure 2). I decided to try them out for my oxidation and reduction unit as a formative assessment to find out what students initial conceptions were at the onset of the topic as well as to circle back to at the end of the unit as evidence of student learning.

Figure 2: Example redox probe
Students read the probe and had to choose one or more answers they agreed with and described their thinking as well as provided an explanation for their answer. Student then placed a yellow sticky note for the first choice, a blue sticky note for the second choice, and a green sticky note on the wall to show their prediction. The purpose of the post-its was to have a quick visual representation of students’ initial conceptions. The whiteboard was divided into three columns: 1, 2 and 3. The students then were broken up into groups, where they met in a designated area with other classmates that shared the same first choice. As a group, the students came up with an argument to support the group’s rationale for the best answer. A group member was chosen to write the group’s rationale on a large whiteboard.
In the reaction between aluminum and copper(II) chloride oxidation is occurring.
How would you describe oxidation?
- Only metals can be oxidized.
- Oxidation is the loss of electrons.
- Oxygen is required for oxidation to occur.
Student are asked to again describe their thinking and provide an explanation for their answer. Students revisited with their group, and added more points to support their argument. If students no longer agreed with their group’s argument, they could move to a group that better fit their view.
Students are then given one final probe.
During the Haber process, ammonia is synthesized from nitrogen gas and hydrogen gas, oxidation is occurring.
How would you describe oxidation?
- Only metals can be oxidized.
- Oxidation is the loss of electrons.
- Oxygen is required for oxidation to occur.
Student were asked to again describe their thinking and provide an explanation for their answer. Students revisited with their group, and added more points to support their argument. If students no longer agreed with their group’s argument, they could move to a group that better fit their view. A spokesperson from each group then shared each group’s points and the whiteboards were left until more information was learned and we could re-visit each phenomena as a class following additional instruction. At the end of the unit, students were required to write a reflection of how their learning and initial conceptions changed over the course of the unit. All students stated that the phenomena helped keep them engaged in the learning. One student explained that while the learning of oxidation numbers in the beginning of the unit was tedious and he did not enjoy it, at least he knew the intended outcome would help him understand something he did care about, uncovering the answers to the phenomena.
Log into your ChemEd X account to download the Supporting Information.
Resources
ACESSE Resource A - Introduction to Formative Assessment to Support Equitable 3D Instruction, OER Commons - Open Educational Resources (accessed 4/25/19)
Black, P., & Wiliam, D. (2009). Developing the theory of formative assessment. Educational Assessment, Evaluation and Accountability (formerly: Journal of Personnel Evaluation in Education), 21(1), 5.
Garrison, C., & Ehringhaus, M. (2007). Formative and summative assessments in the classroom.
Keeley, P. (2015). Science formative assessment, volume 1: 75 practical strategies for linking assessment, instruction, and learning. Corwin Press.
Keeley, P., Eberle, F., & Dorsey, C. (2005). Uncovering student ideas in science: Another 25 formative assessment probes(Vol. 3). NSTA press.
Ruiz‐Primo, M. A., & Furtak, E. M. (2007). Exploring teachers' informal formative assessment practices and students' understanding in the context of scientific inquiry. Journal of research in science teaching, 44(1), 57-84.
Shepard, L. A. (2000). The role of assessment in a learning culture. Educational researcher, 29(7), 4-14.