Especially JCE: February 2018

I love the periodic table. Proclaiming this feels appropriate for Valentine’s Day week, with its modern-day opportunity to declare one’s love and affection (although typically not for a chemistry tool, no matter how elegant and engaging). The curriculum in the K–6 science classrooms where I volunteer had a periodic table focus this week, so part of my method of familiarizing them with the table itself was to share an “Elemental Valentine’s Day Advice” puzzle gleaned from the February 2018 issue of Chem13 News. I like fun, short activities like these where students get to know element names and symbols, along with their atomic numbers, in order to reveal words. There have been many throughout the years in the Journal of Chemical Education as well.
But, of course, there’s much more to the table than that. The February 2018 issue of the JCE offers one way to help students understand the table more in depth, particularly its repeating trends and how one can use the information the table contains. Hoffman and Hennessy discuss the method they used with high school and college chemistry students in The People Periodic Table: A Framework for Engaging Introductory Chemistry Students (available to JCE subscribers).
The instructor arranges seating before students enter the room, limiting where students may choose to sit. As students select their seats, they are not aware of their literally elemental role in the arrangement (see figure 1).
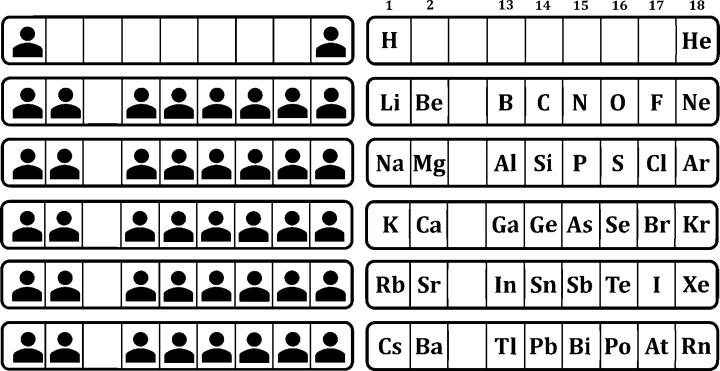
Figure 1 – Reprinted with permission from The People Periodic Table: A Framework for Engaging Introductory Chemistry Students, Hoffman and Hennessy. Journal of Chemical Education, 95 (2), 281–285. Copyright 2018 American Chemical Society.
Their place in the People Periodic Table is soon revealed, when students are assigned to identify their personal element. From that point, the authors’ classes “engaged in predictions regarding the … periodic properties of atomic size, electronegativity, ionization energy, electron affinity, and metallic character.” The classroom-sized scale of the table and visual aspect of the questions/answers (e.g., point in the direction of the person with the largest ionization in their period) help to illustrate the idea of a periodic trend.
The article has online supporting information that outlines the activity and questions to use. Although this formula can get one started, it is not a formulaic activity—instructors should be ready for teachable moments to crop up, in the authors’ experience. When discrepancies in student answers arise, they suggest using the opportunity for the student elements themselves to discuss with their surrounding elements which is correct and why. The supporting information also has suggestions for different seating arrangements for groups from 8 students to more than 45.
The Periodic Table and ChemEd XChange
Can’t get enough of the periodic table? Check in with a recent XChange post that generated a lot of discussion about memorizing element names and symbols. Where do you fall on the memorizing (or not) spectrum? See Jordan Smith’s post I Have My Students Learn All of the Elements...6 Strategies I Use.
More from the February 2018 Issue
Mary Saecker’s post JCE 95.02 February 2018 Issue Highlights summarizes the issue for you. The glowing cover caught my eye—she briefly describes how the images were obtained, along with a link to the related demonstration.
Do you have a past JCE activity or article on the periodic table that you love? Please share! Start by submitting a contribution form, explaining you’d like to contribute to the Especially JCE column. Then, put your thoughts together in a blog post. Questions? Contact us using the ChemEd X contact form.