3-Dimensional Assessments and Helping Students Develop the Necessary Skills

Recent efforts have recognized the Framework for K-12 Science Education and the Next Generation Science Standards as the most current research regarding what we know about teaching and learning of science, and have suggested that 3-dimensional (3D) instruction should guide science instruction at not only the K-12 level, but also at the college level.
Of course, if we want students to truly value 3D thinking and learning, viewing science as a means of investigating and making sense of our natural world through asking questions, analyzing data, developing and using models, and constructing arguments and explanations as opposed to a set of disconnected facts and skills to be memorized and repeated, then we need to design assessments that go beyond testing factoids and skills to ones that focus on students abilities to use their knowledge. Though we still need to assess skills as it is important for students to be able to calculate a molar mass, convert from grams to moles, balance chemical equations, and draw appropriate Lewis structures for molecules, we are all familiar with many ways to do this, including using multiple choice questions. There have been several articles published regarding the development of high-quality multiple choice questions, including one that is freely available as Editors' Choice in the Journal of Chemical Education (JCE) 1.
In addition to these types of assessments, however, we also need to develop assessments that require students to analyze data, use and interpret models, demonstrate mathematical and computational thinking and construct explanations. These assessments are in general much more difficult to write and moreover, more difficult for students to answer as they first need to develop the skills to think like a scientist. In this brief article I hope to provide you with some strategies and resources to help both in the writing of 3D assessment items and in developing student skills to answer these types of questions.
If we want students to truly value 3D thinking and learning, viewing science as a means of investigating and making sense of our natural world through asking questions, analyzing data, developing and using models, and constructing arguments and explanations as opposed to a set of disconnected facts and skills to be memorized and repeated, then we need to design assessments that go beyond testing factoids and skills to ones that focus on students abilities to use their knowledge.
Writing 3D Assessment Items
Admittedly it is easier to write free or constructed response 3D items than it is multiple choice questions of this form, but that doesn’t mean it is impossible to write 3D multiple choice questions. One way you can start to move in this direction is instead of asking about a correct statement, ask students which choice provides evidence for a particular claim. Consider the two questions below.
1. Which of the following is a true statement about Thomson’s cathode ray experiment?
A. The particles were attracted to the + electrode.
B. The particles were different depending on the identity of the cathode.
C. Most of the particles passed straight through the gold foil.
D. The particles had a positive charge.
2. What is the evidence from Thomson’s experiment that supported his claim that "all atoms contain electrons"?
A. The particles were attracted to the + electrode.
B. The particles were deflected by magnetic fields.
C. The particles were deflected by electrical fields.
D. The particles were identical regardless of the identity of the cathode (where they were emitted from).
The first question only presents one correct statement about Thomson’s experiment (A). The other statements are either incorrect or do not pertain to Thomson’s experiment. Accordingly, to answer this question correctly, students just have to memorize the important elements of Thomson’s experiment. The second question is considerably more difficult for students since all of the statements are true regarding Thomson’s experiment, but only D supports the claim that all atoms contain electrons. With this question we are then testing both students’ knowledge about atomic structure as well as their abilities to support claims with appropriate evidence.
Another relatively easy thing you can start to do is ask students to make a choice about something, which can easily be done using multiple choice, and then justify their choice. Consider the following example involving gases:
1. In the following 2-D illustrations, assume that the gas molecules are in motion and that a larger box indicates a larger volume for the container holding the molecules. If the molecules are at the same pressure, what can you say about the relative temperatures of the two samples?
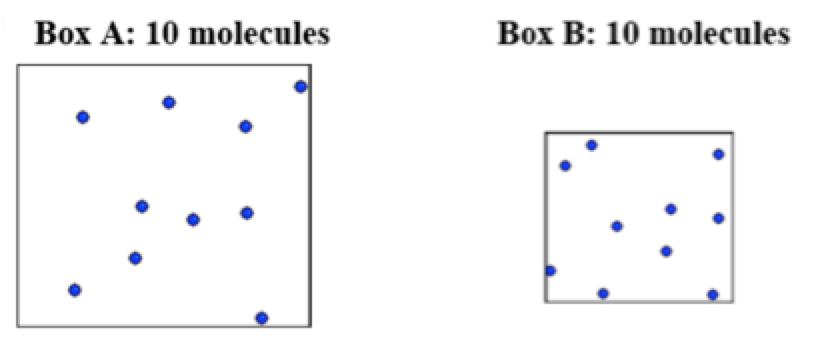
A. The sample in Box A is at a higher temperature
B. The sample in Box B is at a higher temperature
C. Both samples are at the same temperature
D. There is not enough information provided to determine the relative temperatures
2. Provide reasoning to support your answer to the question above. Be sure to include particle motion in your reasoning.
It is often the case that when you require students to justify their choices to multiple choice questions you find that they are not necessarily applying the correct scientific reasoning in making their choices. In this case, we would want them to choose A as the answer to question 1 and then provide reasoning that indicates that pressure is the result of particles colliding with the walls of the container and for the particles in box A to collide with the walls at approximately the same overall frequency/force as those in box B, they must be moving faster (at a higher temperature) as they have more space to move around in.
You may notice that instead of just telling students to justify their answer or provide reasoning to support their answer, a specific clue as to what concept they should include in their answer is provided. This has been found to be very important to ensure that students are accessing the correct knowledge structures to provide you with the response you are looking for. If you do not provide this cue, then you cannot be sure if students did not provide the answer you were looking for because they didn’t know the answer or because they didn’t know exactly what you were looking for. Though the objective is ultimately that students would be able to activate these knowledge structures without prompting, we need to remember that they are still largely novices with respect to chemistry concepts. A recent Editors' Choice JCE article by Underwood and co-workers describes in more detail the importance of scaffolding your question prompts so as to elicit the information you want from students without giving the answer away as well as provides examples of taking traditional chemistry assessment items and transforming them into 3D items.2
Developing Students 3D Skills
These are not things that come naturally to students. Rather they need to be developed through both in class and out of class practice. Providing practice in class requires some rethinking of how you present material. For example, instead of telling students that the strength of attractive forces between ions in ionic compounds depends on both the size of the ions and the charges on the ions, it is possible to provide them with some data and guiding questions to help them construct this understanding on their own through a combination of analyzing data and constructing an explanation. The following is an example of what that could look like:
Thinking About Ionic Bonding
Use the data in the following table in answering the questions below.
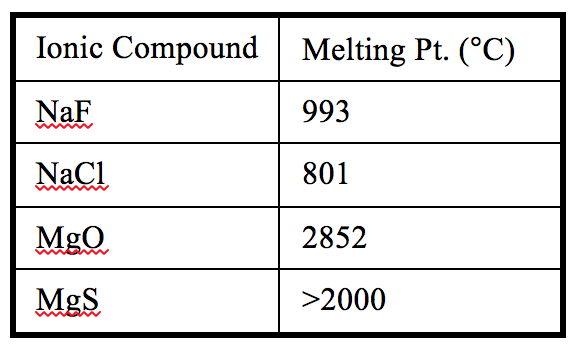
1. How does the strength of the ionic bonds relate to melting point?
2. Which would be expected to have stronger ionic bonds: NaCl or NaF?
A. What evidence did you base this on?
B. How might you explain this? (Hint: think about size of the ions and Coulomb’s law)
3. Which would be expected to have stronger ionic bonds: NaCl or MgO?
A. What evidence did you base this on?
B. How might you explain this? (Hint: think about the charges of the ions and Coulomb’s law)
4. Which factor, the size of the ions or charge, has the larger effect on the melting point? Explain (provide evidence from table and reasoning).
Here students are generally able to identify that the higher the melting point (mp) the stronger the attraction between the ions (#1) and then accordingly identify NaF (#2) and MgO (#3) as the ionic compound in each pair that has the stronger attractive forces between ions. However, the explanation part is always more difficult. In my class at this point we have already used Coulomb’s law (shown below) in talking about relative atomic size and electrostatic attractive and repulsive forces, so they have had some experience with this kind of reasoning already. Yet, it often still takes some prompting to get students to realize that for NaCl and NaF, the only difference is the size of the anion. Once they recognize that, most can explain that because Cl- is larger than F- the ions in NaF are closer together and thus have a stronger force of attraction between them. For NaCl and MgO, they have to realize that though there are some size differences in the ions, the mp for MgO is so much higher that there must be something else going on and that in this case the ion charges for MgO (2+ and 2-) would result in a force of attraction that is about four times larger than that for NaCl (1+ and 1-). Based on this most students can then rationalize that in general charge of the ions has a larger effect on the attractive force between them, and hence the mp, than does the size of the ions.
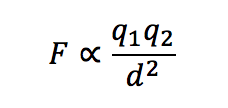
Another simple activity that I did in class the other day was instead of telling students about VSEPR and showing them the shapes, I gave each group of 4 students 12 pieces of modeling clay rolled into balls (3 large and 9 small) and 9 toothpicks and told them to do the following:
1. Using the large ball as the middle “atom” and two small balls as peripheral “atoms” use the toothpicks to connect the atoms so that the peripheral atoms were as far apart as possible in 3D space.
2. Using the large ball as the middle “atom” and three small balls as peripheral “atoms” use the toothpicks to connect the atoms so that the peripheral atoms were as far apart as possible in 3D space.
3. Using the large ball as the middle “atom” and four small balls as peripheral “atoms” use the toothpicks to connect the atoms so that the peripheral atoms were as far apart as possible in 3D space.
Before we begin the activity, we review that covalent bonds are an electrostatic attraction composed of electrons that are attracted to the nuclei of the two atoms involved in the bond, and that when electrons get close together they repel each other. This provides the context for the bonds being as far apart as possible in 3D space. This activity helps them quickly (as it only takes about 10 minutes) construct an understanding of molecular shape while at the same time constructing a model. The first two (linear and trigonal planar) are really easy because all of the atoms are in the same plane so they are really just 2D structures. For number 3, groups almost always make a square planar structure to start. As I walk by I point to it and tell them that they can get the atoms further apart than that in 3D space, and then walk away. It doesn’t take long before groups start to figure it out and make a tetrahedron, and then it goes around the room really fast!
Out of class practice tends to be a little more difficult. One method that has worked well for me, not just in terms of providing students with practice using this kind of thinking but also for providing formative assessment that I can then use to inform my instruction to help students further develop these skills, combines a text messaging app (Remind) with Google forms. This system is free for students and teachers and is easy to use, probably more so for most high school teachers who more regularly use many of the Google products than college faculty! We recently published an article about this in JCE that describes the system, some best practices for using it that we identified during our pilot test last year, and how questions were used both for student self-assessment and as formative assessment to drive subsequent instruction.3 Google forms is a great platform because it allows for a large variety of question types, including the option of having students draw and upload pictures.
One question format that I particularly like with this system is requiring students to do something “traditional” like perform a calculation or choose the compound they would predict to have the higher boiling point and then require them to explain or justify their answer. I also appreciate the ability to help scaffold student answers. The following is an example of how this was done using the Claim, Evidence, Reasoning framework. (Bolded are desired student responses. Italics are common student answers.)
A. Consider a reaction between a metal and a non-metal represented by the following spheres. Which sphere represents the metal (blue or white)? (This is your claim) [Blue]

B. What feature of the spheres did you look at to answer the question above? (This is your evidence.) [Change in size. Many say just size.]
C. How does your evidence support your claim? (This is your reasoning.) [Metals react by losing electrons from the outermost shell, which means their outermost electrons and now closer to the nucleus (one shell lower) and thus they get smaller when you go from metal atom to metal ion. Many students just say they lose electrons so they get smaller.]
You can also think about reversing this order, particularly if you want to address a common issue that you know students have. The following is a question regarding stoichiometry that first gets students to focus on the mathematical thinking, and then asks them to actually do the calculation. Here we are forcing students to recognize the fact that you cannot directly compare masses because each compound/particle has a different mass, but rather must first convert to what I call a “counting unit,” which in chemistry is most often moles. Students then confirm this when they calculate the mass and find that for 1.0g of baking soda they need 0.66g of CaCl2.
A. You are in the lab using baking soda (NaHCO3) and calcium chloride (CaCl2) to try and "launch" a film canister rocket and you know that you will get the most height if you have correct reacting ratio of baking soda and calcium chloride. The balanced chemical equation for this reaction is: 2NaHCO3 + CaCl2 → CaCO3 + CO2 + 2NaCl + H2O. Your lab partner says "It's a 2:1 ratio so we should mix 1.0g of baking soda and 0.5g of CaCl2. You know this will not give you the correct reacting ratio. Explain to your lab partner why this won't work.
B. Calculate how much CaCl2 you need to react with 1.0g of baking soda to get the correct reacting ratio. Show your work, take a picture, and upload it.
The value of this system from a formative assessment perspective is that it can provide instructors with a quick snapshot of student understanding and allow them to use student responses to guide discussion.
The following is an example of a question that follows the first format of requiring students to do a calculation followed by an interpretation of their results and an explanation. I have included data output from student responses as well.
Isomers are molecules that contain the same atoms, but the atoms are connected differently. The reaction below shows an isomerization process, ethanol rearranges to form dimethyl ether (it is not important for you to understand exactly how this occurs).
A. Using the bond energies provided, calculate the enthalpy (delta H) for this isomerization process. Make sure if the process is endothermic your value is positive, and if it is exothermic your enthalpy value is negative. (Short answer where students enter value they calculated)
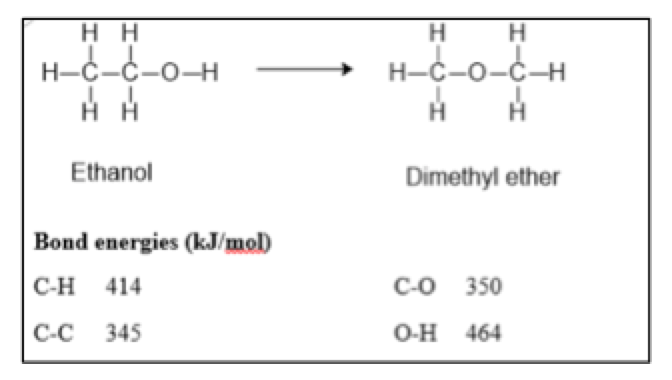
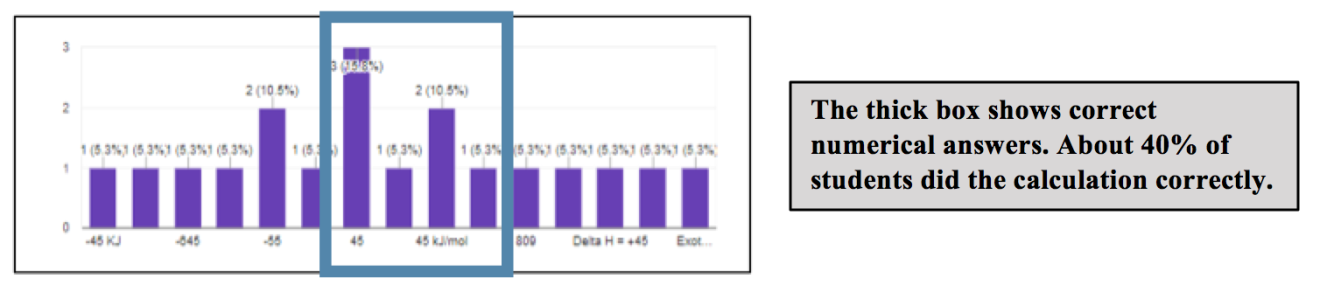
B. Which isomer is more stable?

C. How did you decide on your answer to the question above?
Sample Student Explanations
- Not sure, formal charges are all the same
- Dimethyl ether is more stable due to its symmetrical compared to ethanol, which is asymmetrical
- There is more energy required to break C-H bonds than the others, and dimethyl ether has more C-H bonds than ethanol
- O-H is the most stable bond because it has the highest energy
- Since the delta H is positive, the reaction is endothermic, which means that more energy is required to break the bonds of the reactant than to form the bonds in the product. More energy required means that the bonds are stronger in ethanol, and stronger bonds mean more stability for the molecule.
In terms of using this information to drive instruction, when such a small percentage could actually perform the calculation correctly, I would typically provide students with the correct numerical value and have them make sure that in their group they could figure out how to get that number. In this particular case, I asked the groups to use this to determine which was more stable, and suggested that an energy profile diagram might be helpful. The groups also had to be able to explain their prediction. I then presented them with the list of incorrect explanations (the first 4 bullet points above) and asked them to identify what was incorrect about each one. In this case, if the group came up with one of these as their explanation, they were forced to try and confront it before discussing the correct explanation (the last bullet point which is an actual student response verbatim).
For these types of questions, it was pretty typical that only about half of the students who did the calculation correctly were able to interpret their result correctly. This is no doubt something that we want students to be able to do, so it really emphasizes the need to help students develop this skill and to assess this. Further, though we certainly did continue to see incorrect explanations over the course of the semester, we also noted that we did begin to see the frequency of correct and complete explanations, like the last one in the list, increase. This supports the idea that students can get better at 3D thinking with appropriate scaffolding, practice, and focus on skill development.
I am sure that a number of people have a lot of other great ideas and resources around 3D teaching and assessment that they have found. I would love to hear about them!
References
(1) Towns, M. H., Guide To Developing High-Quality, Reliable, and Valid Multiple-Choice Assessments. J. Chem. Educ. 2014, 91 (9), 1426–1431. (This is an Editors' Choice article. It is open access to all.)
(2) Underwood, S. M.; Posey, L. A.; Herrington, D. G.; Carmel, J. H.; Cooper, M. M., Adapting Assessment Tasks To Support Three-Dimensional Learning. J. Chem. Educ. 2018, 95 (2), 207–217. (This is an Editors' Choice article. It is open access to all.)
(3) Herrington, D. G.; Sweeder, R. D., Using Text Messages To Encourage Meaningful Self-Assessment Outside of the Classroom. J. Chem. Educ. 2018. (Available to subscribers of JCE. Members of ACS & AACT can use their complimentary downloads to access.)